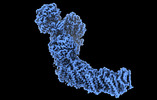
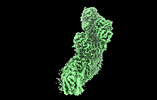
登録情報 データベース : EMDB / ID : EMD-27964タイトル Mycobacterial respiratory complex I, fully-inserted quinone 複合体 : Mycobacterial respiratory complex I, fully-inserted quinoneリガンド : x 7種機能・相同性 分子機能 ドメイン・相同性 構成要素
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Mycolicibacterium smegmatis MC2 155 (バクテリア)手法 / / 解像度 : 2.7 Å Liang Y / Rubinstein JL 資金援助 Organization Grant number 国 Canadian Institutes of Health Research (CIHR) PJT162186 Canadian Institutes of Health Research (CIHR) PJT451412 Canadian Institutes of Health Research (CIHR) PJT173353 Canada Research Chairs
ジャーナル : Proc Natl Acad Sci U S A / 年 : 2023タイトル : Structure of mycobacterial respiratory complex I.
著者 :
Yingke Liang / Alicia Plourde / Stephanie A Bueler / Jun Liu / Peter Brzezinski / Siavash Vahidi / John L Rubinstein / 要旨 :
Oxidative phosphorylation, the combined activity of the electron transport chain (ETC) and adenosine triphosphate synthase, has emerged as a valuable target for the treatment of infection by and ... Oxidative phosphorylation, the combined activity of the electron transport chain (ETC) and adenosine triphosphate synthase, has emerged as a valuable target for the treatment of infection by and other mycobacteria. The mycobacterial ETC is highly branched with multiple dehydrogenases transferring electrons to a membrane-bound pool of menaquinone and multiple oxidases transferring electrons from the pool. The proton-pumping type I nicotinamide adenine dinucleotide (NADH) dehydrogenase (Complex I) is found in low abundance in the plasma membranes of mycobacteria in typical in vitro culture conditions and is often considered dispensable. We found that growth of in carbon-limited conditions greatly increased the abundance of Complex I and allowed isolation of a rotenone-sensitive preparation of the enzyme. Determination of the structure of the complex by cryoEM revealed the "orphan" two-component response regulator protein MSMEG_2064 as a subunit of the assembly. MSMEG_2064 in the complex occupies a site similar to the proposed redox-sensing subunit NDUFA9 in eukaryotic Complex I. An apparent purine nucleoside triphosphate within the NuoG subunit resembles the GTP-derived molybdenum cofactor in homologous formate dehydrogenase enzymes. The membrane region of the complex binds acyl phosphatidylinositol dimannoside, a characteristic three-tailed lipid from the mycobacterial membrane. The structure also shows menaquinone, which is preferentially used over ubiquinone by gram-positive bacteria, in two different positions along the quinone channel, comparable to ubiquinone in other structures and suggesting a conserved quinone binding mechanism. 履歴 登録 2022年8月26日 - ヘッダ(付随情報) 公開 2022年10月12日 - マップ公開 2022年10月12日 - 更新 2023年4月5日 - 現状 2023年4月5日 処理サイト : RCSB / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報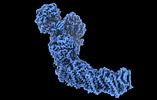
 マップデータ
マップデータ 試料
試料 機能・相同性情報
機能・相同性情報 NADHデヒドロゲナーゼ (キノン) / トランスロカーゼ; ヒドロンの輸送の触媒; 酸化還元酵素反応を伴う / NADH:ubiquinone reductase (non-electrogenic) activity / molybdopterin cofactor binding / oxidoreductase activity, acting on NAD(P)H, quinone or similar compound as acceptor / respiratory chain complex I / phosphorelay signal transduction system /
NADHデヒドロゲナーゼ (キノン) / トランスロカーゼ; ヒドロンの輸送の触媒; 酸化還元酵素反応を伴う / NADH:ubiquinone reductase (non-electrogenic) activity / molybdopterin cofactor binding / oxidoreductase activity, acting on NAD(P)H, quinone or similar compound as acceptor / respiratory chain complex I / phosphorelay signal transduction system /  NADH dehydrogenase (ubiquinone) activity /
NADH dehydrogenase (ubiquinone) activity /  quinone binding / ATP synthesis coupled electron transport ...
quinone binding / ATP synthesis coupled electron transport ... NADHデヒドロゲナーゼ (キノン) / トランスロカーゼ; ヒドロンの輸送の触媒; 酸化還元酵素反応を伴う / NADH:ubiquinone reductase (non-electrogenic) activity / molybdopterin cofactor binding / oxidoreductase activity, acting on NAD(P)H, quinone or similar compound as acceptor / respiratory chain complex I / phosphorelay signal transduction system /
NADHデヒドロゲナーゼ (キノン) / トランスロカーゼ; ヒドロンの輸送の触媒; 酸化還元酵素反応を伴う / NADH:ubiquinone reductase (non-electrogenic) activity / molybdopterin cofactor binding / oxidoreductase activity, acting on NAD(P)H, quinone or similar compound as acceptor / respiratory chain complex I / phosphorelay signal transduction system /  NADH dehydrogenase (ubiquinone) activity /
NADH dehydrogenase (ubiquinone) activity /  quinone binding / ATP synthesis coupled electron transport / 2 iron, 2 sulfur cluster binding / NAD binding / FMN binding / 4 iron, 4 sulfur cluster binding /
quinone binding / ATP synthesis coupled electron transport / 2 iron, 2 sulfur cluster binding / NAD binding / FMN binding / 4 iron, 4 sulfur cluster binding /  oxidoreductase activity / iron ion binding /
oxidoreductase activity / iron ion binding /  生体膜 /
生体膜 /  metal ion binding /
metal ion binding /  細胞膜
細胞膜 Mycolicibacterium smegmatis MC2 155 (バクテリア)
Mycolicibacterium smegmatis MC2 155 (バクテリア) 単粒子再構成法 /
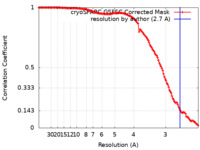
単粒子再構成法 /  クライオ電子顕微鏡法 / 解像度: 2.7 Å
クライオ電子顕微鏡法 / 解像度: 2.7 Å  データ登録者
データ登録者 カナダ, 4件
カナダ, 4件  引用
引用 ジャーナル: Proc Natl Acad Sci U S A / 年: 2023
ジャーナル: Proc Natl Acad Sci U S A / 年: 2023

 ジャーナル: Biorxiv / 年: 2022
ジャーナル: Biorxiv / 年: 2022 構造の表示
構造の表示 ダウンロードとリンク
ダウンロードとリンク emd_27964.map.gz
emd_27964.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-27964-v30.xml
emd-27964-v30.xml emd-27964.xml
emd-27964.xml EMDBヘッダ
EMDBヘッダ emd_27964_fsc.xml
emd_27964_fsc.xml FSCデータファイル
FSCデータファイル emd_27964.png
emd_27964.png emd_27964_half_map_1.map.gz
emd_27964_half_map_1.map.gz emd_27964_half_map_2.map.gz
emd_27964_half_map_2.map.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-27964
http://ftp.pdbj.org/pub/emdb/structures/EMD-27964 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27964
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27964 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
マップ ダウンロード / ファイル: emd_27964.map.gz / 形式: CCP4 / 大きさ: 216 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_27964.map.gz / 形式: CCP4 / 大きさ: 216 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) 試料の構成要素
試料の構成要素 クライオ電子顕微鏡法
クライオ電子顕微鏡法 解析
解析 単粒子再構成法
単粒子再構成法 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN Bright-field microscopy / 最大 デフォーカス(公称値): 2.2 µm / 最小 デフォーカス(公称値): 0.8 µm / 倍率(公称値): 75000
Bright-field microscopy / 最大 デフォーカス(公称値): 2.2 µm / 最小 デフォーカス(公称値): 0.8 µm / 倍率(公称値): 75000 
 ムービー
ムービー コントローラー
コントローラー





















 Z
Z Y
Y X
X