+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8e9i | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Mycobacterial respiratory complex I, semi-inserted quinone | |||||||||||||||
 Components Components |
| |||||||||||||||
 Keywords Keywords |  MEMBRANE PROTEIN / MEMBRANE PROTEIN /  Complex / Complex /  Oxidative Phosphorylation / Oxidative Phosphorylation /  NADH-quinone oxidoreductase / NADH-quinone oxidoreductase /  Iron-sulfur Protein Iron-sulfur Protein | |||||||||||||||
| Function / homology |  Function and homology information Function and homology information NADH dehydrogenase (quinone) / Translocases; Catalysing the translocation of protons; Linked to oxidoreductase reactions / NADH:ubiquinone reductase (non-electrogenic) activity / respiratory chain complex I / molybdopterin cofactor binding / oxidoreductase activity, acting on NAD(P)H, quinone or similar compound as acceptor / phosphorelay signal transduction system / NADH dehydrogenase (quinone) / Translocases; Catalysing the translocation of protons; Linked to oxidoreductase reactions / NADH:ubiquinone reductase (non-electrogenic) activity / respiratory chain complex I / molybdopterin cofactor binding / oxidoreductase activity, acting on NAD(P)H, quinone or similar compound as acceptor / phosphorelay signal transduction system /  NADH dehydrogenase (ubiquinone) activity / NADH dehydrogenase (ubiquinone) activity /  quinone binding / ATP synthesis coupled electron transport ... quinone binding / ATP synthesis coupled electron transport ... NADH dehydrogenase (quinone) / Translocases; Catalysing the translocation of protons; Linked to oxidoreductase reactions / NADH:ubiquinone reductase (non-electrogenic) activity / respiratory chain complex I / molybdopterin cofactor binding / oxidoreductase activity, acting on NAD(P)H, quinone or similar compound as acceptor / phosphorelay signal transduction system / NADH dehydrogenase (quinone) / Translocases; Catalysing the translocation of protons; Linked to oxidoreductase reactions / NADH:ubiquinone reductase (non-electrogenic) activity / respiratory chain complex I / molybdopterin cofactor binding / oxidoreductase activity, acting on NAD(P)H, quinone or similar compound as acceptor / phosphorelay signal transduction system /  NADH dehydrogenase (ubiquinone) activity / NADH dehydrogenase (ubiquinone) activity /  quinone binding / ATP synthesis coupled electron transport / 2 iron, 2 sulfur cluster binding / NAD binding / FMN binding / 4 iron, 4 sulfur cluster binding / quinone binding / ATP synthesis coupled electron transport / 2 iron, 2 sulfur cluster binding / NAD binding / FMN binding / 4 iron, 4 sulfur cluster binding /  oxidoreductase activity / iron ion binding / oxidoreductase activity / iron ion binding /  membrane / membrane /  metal ion binding / metal ion binding /  plasma membrane plasma membraneSimilarity search - Function | |||||||||||||||
| Biological species |  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria) | |||||||||||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 2.8 Å cryo EM / Resolution: 2.8 Å | |||||||||||||||
 Authors Authors | Liang, Y. / Rubinstein, J.L. | |||||||||||||||
| Funding support |  Canada, 4items Canada, 4items
| |||||||||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2023 Journal: Proc Natl Acad Sci U S A / Year: 2023Title: Structure of mycobacterial respiratory complex I. Authors: Yingke Liang / Alicia Plourde / Stephanie A Bueler / Jun Liu / Peter Brzezinski / Siavash Vahidi / John L Rubinstein /   Abstract: Oxidative phosphorylation, the combined activity of the electron transport chain (ETC) and adenosine triphosphate synthase, has emerged as a valuable target for the treatment of infection by and ...Oxidative phosphorylation, the combined activity of the electron transport chain (ETC) and adenosine triphosphate synthase, has emerged as a valuable target for the treatment of infection by and other mycobacteria. The mycobacterial ETC is highly branched with multiple dehydrogenases transferring electrons to a membrane-bound pool of menaquinone and multiple oxidases transferring electrons from the pool. The proton-pumping type I nicotinamide adenine dinucleotide (NADH) dehydrogenase (Complex I) is found in low abundance in the plasma membranes of mycobacteria in typical in vitro culture conditions and is often considered dispensable. We found that growth of in carbon-limited conditions greatly increased the abundance of Complex I and allowed isolation of a rotenone-sensitive preparation of the enzyme. Determination of the structure of the complex by cryoEM revealed the "orphan" two-component response regulator protein MSMEG_2064 as a subunit of the assembly. MSMEG_2064 in the complex occupies a site similar to the proposed redox-sensing subunit NDUFA9 in eukaryotic Complex I. An apparent purine nucleoside triphosphate within the NuoG subunit resembles the GTP-derived molybdenum cofactor in homologous formate dehydrogenase enzymes. The membrane region of the complex binds acyl phosphatidylinositol dimannoside, a characteristic three-tailed lipid from the mycobacterial membrane. The structure also shows menaquinone, which is preferentially used over ubiquinone by gram-positive bacteria, in two different positions along the quinone channel, comparable to ubiquinone in other structures and suggesting a conserved quinone binding mechanism. #1:  Journal: Biorxiv / Year: 2022 Journal: Biorxiv / Year: 2022Title: Structure of mycobacterial respiratory Complex I Authors: Liang, Y. / Plourde, A. / Bueler, S.A. / Liu, J. / Brzezinski, P. / Vahidi, S. / Rubinstein, J.L. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8e9i.cif.gz 8e9i.cif.gz | 817.9 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8e9i.ent.gz pdb8e9i.ent.gz | 648.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8e9i.json.gz 8e9i.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/e9/8e9i https://data.pdbj.org/pub/pdb/validation_reports/e9/8e9i ftp://data.pdbj.org/pub/pdb/validation_reports/e9/8e9i ftp://data.pdbj.org/pub/pdb/validation_reports/e9/8e9i | HTTPS FTP |
|---|
-Related structure data
| Related structure data | 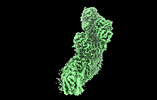 27965MC  8e9gC  8e9hC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Protein , 1 types, 1 molecules O
| #1: Protein |  Two-component regulatory system Two-component regulatory systemMass: 14119.161 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria)Strain: ATCC 700084 / mc(2)155 / References: UniProt: A0QU37 |
|---|
-NADH-quinone oxidoreductase subunit ... , 12 types, 12 molecules BACDEGFIHJKN
| #2: Protein |  NADH dehydrogenase (quinone) / NADH dehydrogenase I subunit B / NDH-1 subunit B NADH dehydrogenase (quinone) / NADH dehydrogenase I subunit B / NDH-1 subunit BMass: 20123.703 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria)Strain: ATCC 700084 / mc(2)155 References: UniProt: A0QU35, Translocases; Catalysing the translocation of protons; Linked to oxidoreductase reactions |
|---|---|
| #3: Protein |  NADH dehydrogenase (quinone) / NADH dehydrogenase I subunit A / NDH-1 subunit A / NUO1 NADH dehydrogenase (quinone) / NADH dehydrogenase I subunit A / NDH-1 subunit A / NUO1Mass: 13628.287 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria)Strain: ATCC 700084 / mc(2)155 References: UniProt: A0QU36, Translocases; Catalysing the translocation of protons; Linked to oxidoreductase reactions |
| #4: Protein |  NADH dehydrogenase (quinone) / NADH dehydrogenase I subunit C / NDH-1 subunit C NADH dehydrogenase (quinone) / NADH dehydrogenase I subunit C / NDH-1 subunit CMass: 26610.689 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria)Strain: ATCC 700084 / mc(2)155 References: UniProt: A0QU34, Translocases; Catalysing the translocation of protons; Linked to oxidoreductase reactions |
| #5: Protein |  NADH dehydrogenase (quinone) / NADH dehydrogenase I subunit D / NDH-1 subunit D NADH dehydrogenase (quinone) / NADH dehydrogenase I subunit D / NDH-1 subunit DMass: 48556.750 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria)Strain: ATCC 700084 / mc(2)155 References: UniProt: A0QU33, Translocases; Catalysing the translocation of protons; Linked to oxidoreductase reactions |
| #6: Protein |  NADH dehydrogenase (quinone) NADH dehydrogenase (quinone)Mass: 25746.746 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria)Strain: ATCC 700084 / mc(2)155 / References: UniProt: A0QU32,  NADH dehydrogenase (quinone) NADH dehydrogenase (quinone) |
| #7: Protein |  NADH dehydrogenase (quinone) NADH dehydrogenase (quinone)Mass: 83894.773 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria)Strain: ATCC 700084 / mc(2)155 References: UniProt: A0QU30, Translocases; Catalysing the translocation of protons; Linked to oxidoreductase reactions |
| #8: Protein |  NADH dehydrogenase (quinone) NADH dehydrogenase (quinone)Mass: 47831.078 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria)Strain: ATCC 700084 / mc(2)155 References: UniProt: A0QU31, Translocases; Catalysing the translocation of protons; Linked to oxidoreductase reactions |
| #9: Protein |  NADH dehydrogenase (quinone) / NADH dehydrogenase I subunit I / NDH-1 subunit I NADH dehydrogenase (quinone) / NADH dehydrogenase I subunit I / NDH-1 subunit IMass: 19860.510 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria)Strain: ATCC 700084 / mc(2)155 References: UniProt: A0QU28, Translocases; Catalysing the translocation of protons; Linked to oxidoreductase reactions |
| #10: Protein |  NADH dehydrogenase (quinone) / NADH dehydrogenase I subunit H / NDH-1 subunit H NADH dehydrogenase (quinone) / NADH dehydrogenase I subunit H / NDH-1 subunit HMass: 44477.293 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria)Strain: ATCC 700084 / mc(2)155 References: UniProt: A0QU29, Translocases; Catalysing the translocation of protons; Linked to oxidoreductase reactions |
| #11: Protein |  NADH dehydrogenase (quinone) NADH dehydrogenase (quinone)Mass: 26398.791 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria)Strain: ATCC 700084 / mc(2)155 References: UniProt: A0QU27, Translocases; Catalysing the translocation of protons; Linked to oxidoreductase reactions |
| #12: Protein |  NADH dehydrogenase (quinone) / NADH dehydrogenase I subunit K / NDH-1 subunit K NADH dehydrogenase (quinone) / NADH dehydrogenase I subunit K / NDH-1 subunit KMass: 10876.923 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria)Strain: ATCC 700084 / mc(2)155 References: UniProt: A0QU26, Translocases; Catalysing the translocation of protons; Linked to oxidoreductase reactions |
| #14: Protein |  NADH dehydrogenase (quinone) / NADH dehydrogenase I subunit N / NDH-1 subunit N NADH dehydrogenase (quinone) / NADH dehydrogenase I subunit N / NDH-1 subunit NMass: 54013.266 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria)Strain: ATCC 700084 / mc(2)155 References: UniProt: A0QU23, Translocases; Catalysing the translocation of protons; Linked to oxidoreductase reactions |
-NADH-quinone oxidoreductase, ... , 2 types, 2 molecules LM
| #13: Protein |  NADH dehydrogenase (quinone) NADH dehydrogenase (quinone)Mass: 65446.609 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria)Strain: ATCC 700084 / mc(2)155 / References: UniProt: A0QU25,  NADH dehydrogenase (quinone) NADH dehydrogenase (quinone) |
|---|---|
| #15: Protein |  NADH dehydrogenase (quinone) NADH dehydrogenase (quinone)Mass: 56338.316 Da / Num. of mol.: 1 / Source method: isolated from a natural source Source: (natural)  Mycolicibacterium smegmatis MC2 155 (bacteria) Mycolicibacterium smegmatis MC2 155 (bacteria)Strain: ATCC 700084 / mc(2)155 / References: UniProt: A0QU24,  NADH dehydrogenase (quinone) NADH dehydrogenase (quinone) |
-Non-polymers , 7 types, 14 molecules 




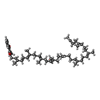







| #16: Chemical | ChemComp-SF4 /  Iron–sulfur cluster Iron–sulfur cluster#17: Chemical |  Iron–sulfur cluster Iron–sulfur cluster#18: Chemical | ChemComp-GTP / |  Guanosine triphosphate Guanosine triphosphate#19: Chemical | ChemComp-FMN / |  Flavin mononucleotide Flavin mononucleotide#20: Chemical | ChemComp-ZN / | #21: Chemical | ChemComp-MQ9 / |  Vitamin K2 Vitamin K2#22: Chemical | ChemComp-XP2 / ( | |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Mycobacterial respiratory complex I, semi-inserted quinone Type: COMPLEX / Entity ID: #1-#15 / Source: NATURAL |
|---|---|
| Molecular weight | Value: 0.56 MDa / Experimental value: NO |
| Source (natural) | Organism:  Mycolicibacterium smegmatis MC2 155 (bacteria) / Cellular location: Inner membrane Mycolicibacterium smegmatis MC2 155 (bacteria) / Cellular location: Inner membrane |
| Buffer solution | pH: 6 |
| Specimen | Conc.: 2 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES : YES |
| Specimen support | Grid material: COPPER/RHODIUM / Grid mesh size: 400 divisions/in. / Grid type: Homemade |
Vitrification | Instrument: LEICA EM GP / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 277 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Nominal magnification: 75000 X / Nominal defocus max: 2200 nm / Nominal defocus min: 800 nm Bright-field microscopy / Nominal magnification: 75000 X / Nominal defocus max: 2200 nm / Nominal defocus min: 800 nm |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Electron dose: 47 e/Å2 / Film or detector model: FEI FALCON IV (4k x 4k) |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.19.2_4158: / Classification: refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| ||||||||||||||||||||||||
CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||
| Symmetry | Point symmetry : C1 (asymmetric) : C1 (asymmetric) | ||||||||||||||||||||||||
3D reconstruction | Resolution: 2.8 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 14385 / Symmetry type: POINT | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller




 PDBj
PDBj






















