+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
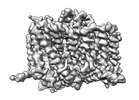
| Title | Complex of human ASCT2 with Syncytin-1 | |||||||||
 Map data Map data | 2.62 Ang map of ASCT2-Syncytin-1. | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Small neutral amino acid transporter / ASCT2 / Syncytin-1 /  Receptor binding domain / Receptor binding domain /  PROTEIN TRANSPORT PROTEIN TRANSPORT | |||||||||
| Function / homology |  Function and homology information Function and homology informationsyncytium formation by plasma membrane fusion / glutamine secretion / syncytium formation / L-glutamine import across plasma membrane / glutamine transport / L-glutamine transmembrane transporter activity / L-serine transmembrane transporter activity /  ligand-gated channel activity / neutral amino acid transport / amino acid transmembrane transporter activity ...syncytium formation by plasma membrane fusion / glutamine secretion / syncytium formation / L-glutamine import across plasma membrane / glutamine transport / L-glutamine transmembrane transporter activity / L-serine transmembrane transporter activity / ligand-gated channel activity / neutral amino acid transport / amino acid transmembrane transporter activity ...syncytium formation by plasma membrane fusion / glutamine secretion / syncytium formation / L-glutamine import across plasma membrane / glutamine transport / L-glutamine transmembrane transporter activity / L-serine transmembrane transporter activity /  ligand-gated channel activity / neutral amino acid transport / amino acid transmembrane transporter activity / Amino acid transport across the plasma membrane / L-aspartate transmembrane transporter activity / L-aspartate import across plasma membrane / neutral L-amino acid transmembrane transporter activity / ligand-gated channel activity / neutral amino acid transport / amino acid transmembrane transporter activity / Amino acid transport across the plasma membrane / L-aspartate transmembrane transporter activity / L-aspartate import across plasma membrane / neutral L-amino acid transmembrane transporter activity /  symporter activity / myoblast fusion / symporter activity / myoblast fusion /  antiporter activity / amino acid transport / RHOJ GTPase cycle / RHOQ GTPase cycle / protein homotrimerization / RHOH GTPase cycle / transport across blood-brain barrier / anatomical structure morphogenesis / RAC3 GTPase cycle / RAC1 GTPase cycle / antiporter activity / amino acid transport / RHOJ GTPase cycle / RHOQ GTPase cycle / protein homotrimerization / RHOH GTPase cycle / transport across blood-brain barrier / anatomical structure morphogenesis / RAC3 GTPase cycle / RAC1 GTPase cycle /  erythrocyte differentiation / basal plasma membrane / erythrocyte differentiation / basal plasma membrane /  melanosome / virus receptor activity / melanosome / virus receptor activity /  signaling receptor activity / extracellular exosome / signaling receptor activity / extracellular exosome /  membrane / membrane /  metal ion binding / metal ion binding /  plasma membrane plasma membraneSimilarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 2.62 Å cryo EM / Resolution: 2.62 Å | |||||||||
 Authors Authors | Khare S / Reyes N | |||||||||
| Funding support | European Union, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2024 Journal: Nat Struct Mol Biol / Year: 2024Title: Receptor-recognition and antiviral mechanisms of retrovirus-derived human proteins. Authors: Shashank Khare / Miryam I Villalba / Juan C Canul-Tec / Arantza Balsebre Cajiao / Anand Kumar / Marija Backovic / Felix A Rey / Els Pardon / Jan Steyaert / Camilo Perez / Nicolas Reyes /     Abstract: Human syncytin-1 and suppressyn are cellular proteins of retroviral origin involved in cell-cell fusion events to establish the maternal-fetal interface in the placenta. In cell culture, they ...Human syncytin-1 and suppressyn are cellular proteins of retroviral origin involved in cell-cell fusion events to establish the maternal-fetal interface in the placenta. In cell culture, they restrict infections from members of the largest interference group of vertebrate retroviruses, and are regarded as host immunity factors expressed during development. At the core of the syncytin-1 and suppressyn functions are poorly understood mechanisms to recognize a common cellular receptor, the membrane transporter ASCT2. Here, we present cryo-electron microscopy structures of human ASCT2 in complexes with the receptor-binding domains of syncytin-1 and suppressyn. Despite their evolutionary divergence, the two placental proteins occupy similar positions in ASCT2, and are stabilized by the formation of a hybrid β-sheet or 'clamp' with the receptor. Structural predictions of the receptor-binding domains of extant retroviruses indicate overlapping binding interfaces and clamping sites with ASCT2, revealing a competition mechanism between the placental proteins and the retroviruses. Our work uncovers a common ASCT2 recognition mechanism by a large group of endogenous and disease-causing retroviruses, and provides high-resolution views on how placental human proteins exert morphological and immunological functions. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_17192.map.gz emd_17192.map.gz | 122.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-17192-v30.xml emd-17192-v30.xml emd-17192.xml emd-17192.xml | 22.1 KB 22.1 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_17192_fsc.xml emd_17192_fsc.xml | 13.2 KB | Display |  FSC data file FSC data file |
| Images |  emd_17192.png emd_17192.png | 116.8 KB | ||
| Filedesc metadata |  emd-17192.cif.gz emd-17192.cif.gz | 7 KB | ||
| Others |  emd_17192_half_map_1.map.gz emd_17192_half_map_1.map.gz emd_17192_half_map_2.map.gz emd_17192_half_map_2.map.gz | 226.8 MB 226.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-17192 http://ftp.pdbj.org/pub/emdb/structures/EMD-17192 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17192 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17192 | HTTPS FTP |
-Related structure data
| Related structure data |  8ouhMC  8oudC  8ouiC  8oujC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_17192.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_17192.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | 2.62 Ang map of ASCT2-Syncytin-1. | ||||||||||||||||||||
| Voxel size | X=Y=Z: 0.731 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_17192_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_17192_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Complex of ASCT2 with Synctin-1
| Entire | Name: Complex of ASCT2 with Synctin-1 |
|---|---|
| Components |
|
-Supramolecule #1: Complex of ASCT2 with Synctin-1
| Supramolecule | Name: Complex of ASCT2 with Synctin-1 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|---|
| Molecular weight | Theoretical: 180 kDa/nm |
-Supramolecule #2: Alanine Serine Cysteine Transporter 2
| Supramolecule | Name: Alanine Serine Cysteine Transporter 2 / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
-Supramolecule #3: Syncytin-1
| Supramolecule | Name: Syncytin-1 / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #2 |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Neutral amino acid transporter B(0)
| Macromolecule | Name: Neutral amino acid transporter B(0) / type: protein_or_peptide / ID: 1 / Number of copies: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 58.984566 KDa |
| Recombinant expression | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MWSHPQFEKS SGGLEVLFQG PMVADPPRDS KGLAAAEPTA NGGLALASIE DQGAAAGGYC GSRDQVRRCL RANLLVLLTV VAVVAGVAL GLGVSGAGGA LALGPERLSA FVFPGELLLR LLRMIILPLV VCSLIGGAAS LDPGALGRLG AWALLFFLVT T LLASALGV ...String: MWSHPQFEKS SGGLEVLFQG PMVADPPRDS KGLAAAEPTA NGGLALASIE DQGAAAGGYC GSRDQVRRCL RANLLVLLTV VAVVAGVAL GLGVSGAGGA LALGPERLSA FVFPGELLLR LLRMIILPLV VCSLIGGAAS LDPGALGRLG AWALLFFLVT T LLASALGV GLALALQPGA ASAAINASVG AAGSAENAPS KEVLDSFLDL ARNIFPSNLV SAAFRSYSTT YEERNITGTR VK VPVGQEV EGMNILGLVV FAIVFGVALR KLGPEGELLI RFFNSFNEAT MVLVSWIMWY APVGIMFLVA GKIVEMEDVG LLF ARLGKY ILCCLLGHAI HGLLVLPLIY FLFTRKNPYR FLWGIVTPLA TAFGTSSSSA TLPLMMKCVE ENNGVAKHIS RFIL PIGAT VNMDGAALFQ CVAAVFIAQL SQQSLDFVKI ITILVTATAS SVGAAGIPAG GVLTLAIILE AVNLPVDHIS LILAV DWLV DRSCTVLNVE GDALGAGLLQ NYVDRTESRS TEPELIQVKS ELPLDPLPVP TEEGNPLLKH YRGPAGDATV ASEKES VM UniProtKB: Neutral amino acid transporter B(0) |
-Macromolecule #2: Syncytin-1
| Macromolecule | Name: Syncytin-1 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 49.24593 KDa |
| Recombinant expression | Organism:   Drosophila melanogaster (fruit fly) Drosophila melanogaster (fruit fly) |
| Sequence | String: AVVAFVGLSL GAPPPCRCMT SSSPYQEFLW RMQRPGNIDA PSYRSLSKGT PTFTAHTHMP RNCYHSATLC MHANTHYWTG KMINPSCPG GLGVTVCWTY FTQTGMSDGG GVQDQAREKH VKEVISQLTR VHGTSSPYKG LDLSKLHETL RTHTRLVSLF N TTLTGLHE ...String: AVVAFVGLSL GAPPPCRCMT SSSPYQEFLW RMQRPGNIDA PSYRSLSKGT PTFTAHTHMP RNCYHSATLC MHANTHYWTG KMINPSCPG GLGVTVCWTY FTQTGMSDGG GVQDQAREKH VKEVISQLTR VHGTSSPYKG LDLSKLHETL RTHTRLVSLF N TTLTGLHE VSAQNPTNSW ICLPLNFRPY VSIPVPEQWN NFSTEINTTS VLVGPLVSNL EITHTSNLTC VKFSNTTYTT NS QCIRWVT PPTQIVCLPS GIFFVCGTSA YRCLNGSSES MCFLSFLVPP MTIYTEQDLY NYVISKPRNK RVPILPFVIG AGV LGALGT GIGGITTSTQ FYYKLSQELN GDMERVADSL VTLQDQLNSL AAVVLQNRRA LDLLTAERGG TCLFLGEECC YYVN QSGIV TEKVKEIRDR IQRRAEELRN TGPWGSGLEV LFQGPGPEPE A UniProtKB: Syncytin-1 |
-Macromolecule #3: ALANINE
| Macromolecule | Name: ALANINE / type: ligand / ID: 3 / Number of copies: 2 / Formula: ALA |
|---|---|
| Molecular weight | Theoretical: 89.093 Da |
| Chemical component information |  ChemComp-ALA: |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 9 mg/mL | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.4 Component:
| ||||||||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Support film - Material: CARBON / Support film - topology: CONTINUOUS / Support film - Film thickness: 10 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 40 sec. / Pretreatment - Atmosphere: AIR / Pretreatment - Pressure: 0.003 kPa | ||||||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.4 µm / Nominal magnification: 165000 Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.4 µm / Nominal magnification: 165000 |
| Specialist optics | Phase plate: VOLTA PHASE PLATE / Energy filter - Name: TFS Selectris X / Energy filter - Slit width: 10 eV |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Software | Name: EPU (ver. 2) |
| Image recording | Film or detector model: FEI FALCON IV (4k x 4k) / Number grids imaged: 1 / Number real images: 15376 / Average exposure time: 6.0 sec. / Average electron dose: 53.4 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
-Atomic model buiding 1
| Initial model |
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Software | Name:  Coot (ver. 0.9.8.3) Coot (ver. 0.9.8.3) | |||||||||
| Refinement | Space: REAL / Protocol: FLEXIBLE FIT / Overall B value: 60.1 | |||||||||
| Output model |  PDB-8ouh: |
 Movie
Movie Controller
Controller












 Z
Z Y
Y X
X