+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-10204 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Abeta fibril (Morphology I) | |||||||||
 Map data Map data | ABeta(1-40) Morphology I | |||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationregulation of epidermal growth factor-activated receptor activity / signaling receptor activator activity / collateral sprouting in absence of injury / cytosolic mRNA polyadenylation / microglia development / regulation of synapse structure or activity / Formyl peptide receptors bind formyl peptides and many other ligands / axo-dendritic transport / synaptic assembly at neuromuscular junction / smooth endoplasmic reticulum calcium ion homeostasis ...regulation of epidermal growth factor-activated receptor activity / signaling receptor activator activity / collateral sprouting in absence of injury / cytosolic mRNA polyadenylation / microglia development / regulation of synapse structure or activity / Formyl peptide receptors bind formyl peptides and many other ligands / axo-dendritic transport / synaptic assembly at neuromuscular junction / smooth endoplasmic reticulum calcium ion homeostasis / axon midline choice point recognition / astrocyte activation involved in immune response / regulation of spontaneous synaptic transmission /  regulation of Wnt signaling pathway / mating behavior / regulation of Wnt signaling pathway / mating behavior /  ciliary rootlet / Lysosome Vesicle Biogenesis / ciliary rootlet / Lysosome Vesicle Biogenesis /  PTB domain binding / Golgi-associated vesicle / positive regulation of amyloid fibril formation / neuron remodeling / Insertion of tail-anchored proteins into the endoplasmic reticulum membrane / : / Deregulated CDK5 triggers multiple neurodegenerative pathways in Alzheimer's disease models / presynaptic active zone / nuclear envelope lumen / modulation of excitatory postsynaptic potential / suckling behavior / COPII-coated ER to Golgi transport vesicle / dendrite development / PTB domain binding / Golgi-associated vesicle / positive regulation of amyloid fibril formation / neuron remodeling / Insertion of tail-anchored proteins into the endoplasmic reticulum membrane / : / Deregulated CDK5 triggers multiple neurodegenerative pathways in Alzheimer's disease models / presynaptic active zone / nuclear envelope lumen / modulation of excitatory postsynaptic potential / suckling behavior / COPII-coated ER to Golgi transport vesicle / dendrite development /  smooth endoplasmic reticulum / smooth endoplasmic reticulum /  regulation of NMDA receptor activity / TRAF6 mediated NF-kB activation / Advanced glycosylation endproduct receptor signaling / neuromuscular process controlling balance / regulation of presynapse assembly / The NLRP3 inflammasome / intracellular copper ion homeostasis / transition metal ion binding / regulation of multicellular organism growth / negative regulation of long-term synaptic potentiation / negative regulation of neuron differentiation / ECM proteoglycans / spindle midzone / positive regulation of T cell migration / regulation of NMDA receptor activity / TRAF6 mediated NF-kB activation / Advanced glycosylation endproduct receptor signaling / neuromuscular process controlling balance / regulation of presynapse assembly / The NLRP3 inflammasome / intracellular copper ion homeostasis / transition metal ion binding / regulation of multicellular organism growth / negative regulation of long-term synaptic potentiation / negative regulation of neuron differentiation / ECM proteoglycans / spindle midzone / positive regulation of T cell migration /  Purinergic signaling in leishmaniasis infection / positive regulation of calcium-mediated signaling / forebrain development / regulation of peptidyl-tyrosine phosphorylation / positive regulation of chemokine production / Purinergic signaling in leishmaniasis infection / positive regulation of calcium-mediated signaling / forebrain development / regulation of peptidyl-tyrosine phosphorylation / positive regulation of chemokine production /  clathrin-coated pit / clathrin-coated pit /  Notch signaling pathway / positive regulation of G2/M transition of mitotic cell cycle / ionotropic glutamate receptor signaling pathway / positive regulation of protein metabolic process / neuron projection maintenance / cholesterol metabolic process / extracellular matrix organization / positive regulation of glycolytic process / positive regulation of mitotic cell cycle / response to interleukin-1 / Notch signaling pathway / positive regulation of G2/M transition of mitotic cell cycle / ionotropic glutamate receptor signaling pathway / positive regulation of protein metabolic process / neuron projection maintenance / cholesterol metabolic process / extracellular matrix organization / positive regulation of glycolytic process / positive regulation of mitotic cell cycle / response to interleukin-1 /  axonogenesis / adult locomotory behavior / trans-Golgi network membrane / dendritic shaft / locomotory behavior / platelet alpha granule lumen / positive regulation of peptidyl-threonine phosphorylation / axonogenesis / adult locomotory behavior / trans-Golgi network membrane / dendritic shaft / locomotory behavior / platelet alpha granule lumen / positive regulation of peptidyl-threonine phosphorylation /  learning / learning /  central nervous system development / positive regulation of interleukin-1 beta production / positive regulation of long-term synaptic potentiation / astrocyte activation / endosome lumen / synapse organization / central nervous system development / positive regulation of interleukin-1 beta production / positive regulation of long-term synaptic potentiation / astrocyte activation / endosome lumen / synapse organization /  Post-translational protein phosphorylation / regulation of long-term neuronal synaptic plasticity / positive regulation of JNK cascade / microglial cell activation / TAK1-dependent IKK and NF-kappa-B activation / Post-translational protein phosphorylation / regulation of long-term neuronal synaptic plasticity / positive regulation of JNK cascade / microglial cell activation / TAK1-dependent IKK and NF-kappa-B activation /  visual learning / visual learning /  neuromuscular junction / serine-type endopeptidase inhibitor activity / recycling endosome / neuromuscular junction / serine-type endopeptidase inhibitor activity / recycling endosome /  cognition / positive regulation of inflammatory response / Golgi lumen / neuron cellular homeostasis / cognition / positive regulation of inflammatory response / Golgi lumen / neuron cellular homeostasis /  endocytosis / positive regulation of interleukin-6 production / positive regulation of non-canonical NF-kappaB signal transduction / cellular response to amyloid-beta / Regulation of Insulin-like Growth Factor (IGF) transport and uptake by Insulin-like Growth Factor Binding Proteins (IGFBPs) / neuron projection development / positive regulation of DNA-binding transcription factor activity / G2/M transition of mitotic cell cycle / cell-cell junction / endocytosis / positive regulation of interleukin-6 production / positive regulation of non-canonical NF-kappaB signal transduction / cellular response to amyloid-beta / Regulation of Insulin-like Growth Factor (IGF) transport and uptake by Insulin-like Growth Factor Binding Proteins (IGFBPs) / neuron projection development / positive regulation of DNA-binding transcription factor activity / G2/M transition of mitotic cell cycle / cell-cell junction /  synaptic vesicle / positive regulation of tumor necrosis factor production / synaptic vesicle / positive regulation of tumor necrosis factor production /  regulation of translation regulation of translationSimilarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) / Homo sapiens (human) /   Human (human) Human (human) | |||||||||
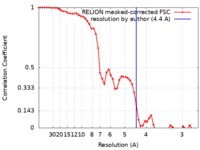
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 4.4 Å cryo EM / Resolution: 4.4 Å | |||||||||
 Authors Authors | Kollmer M / Fandrich M | |||||||||
| Funding support |  Germany, 2 items Germany, 2 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2019 Journal: Nat Commun / Year: 2019Title: Cryo-EM structure and polymorphism of Aβ amyloid fibrils purified from Alzheimer's brain tissue. Authors: Marius Kollmer / William Close / Leonie Funk / Jay Rasmussen / Aref Bsoul / Angelika Schierhorn / Matthias Schmidt / Christina J Sigurdson / Mathias Jucker / Marcus Fändrich /    Abstract: The formation of Aβ amyloid fibrils is a neuropathological hallmark of Alzheimer's disease and cerebral amyloid angiopathy. However, the structure of Aβ amyloid fibrils from brain tissue is poorly ...The formation of Aβ amyloid fibrils is a neuropathological hallmark of Alzheimer's disease and cerebral amyloid angiopathy. However, the structure of Aβ amyloid fibrils from brain tissue is poorly understood. Here we report the purification of Aβ amyloid fibrils from meningeal Alzheimer's brain tissue and their structural analysis with cryo-electron microscopy. We show that these fibrils are polymorphic but consist of similarly structured protofilaments. Brain derived Aβ amyloid fibrils are right-hand twisted and their peptide fold differs sharply from previously analyzed Aβ fibrils that were formed in vitro. These data underscore the importance to use patient-derived amyloid fibrils when investigating the structural basis of the disease. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_10204.map.gz emd_10204.map.gz | 1.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-10204-v30.xml emd-10204-v30.xml emd-10204.xml emd-10204.xml | 11.8 KB 11.8 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_10204_fsc.xml emd_10204_fsc.xml | 5.4 KB | Display |  FSC data file FSC data file |
| Images |  emd_10204.png emd_10204.png | 60 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-10204 http://ftp.pdbj.org/pub/emdb/structures/EMD-10204 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-10204 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-10204 | HTTPS FTP |
-Related structure data
| Related structure data |  6shsMC  4864C  4866C C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_10204.map.gz / Format: CCP4 / Size: 12.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_10204.map.gz / Format: CCP4 / Size: 12.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | ABeta(1-40) Morphology I | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.35 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : meninges
| Entire | Name: meninges |
|---|---|
| Components |
|
-Supramolecule #1: meninges
| Supramolecule | Name: meninges / type: tissue / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) / Organ: brain / Tissue: meninges Homo sapiens (human) / Organ: brain / Tissue: meninges |
-Macromolecule #1: Amyloid-beta precursor protein
| Macromolecule | Name: Amyloid-beta precursor protein / type: protein_or_peptide / ID: 1 / Number of copies: 12 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Human (human) / Organ: BRAIN / Tissue: meninges Human (human) / Organ: BRAIN / Tissue: meninges |
| Molecular weight | Theoretical: 4.335852 KDa |
| Sequence | String: DAEFRHDSGY EVHHQKLVFF AEDVGSNKGA IIGLMVGGVV |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | helical array |
- Sample preparation
Sample preparation
| Buffer | pH: 7 |
|---|---|
| Grid | Model: C-flat-1.2/1.3 / Material: COPPER / Mesh: 200 / Pretreatment - Type: GLOW DISCHARGE |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 90 % / Chamber temperature: 294 K / Instrument: GATAN CRYOPLUNGE 3 |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 105000 Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 105000 |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Digitization - Frames/image: 1-40 / Number real images: 3027 / Average exposure time: 24.0 sec. / Average electron dose: 40.9 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: AB INITIO MODEL |
|---|---|
| Output model |  PDB-6shs: |
 Movie
Movie Controller
Controller