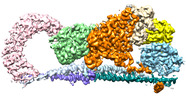
登録情報 データベース : EMDB / ID : EMD-22650タイトル Cryo-EM structure of STRIPAK complex SK7 primary map 複合体 : SK7 complexタンパク質・ペプチド : Serine/threonine-protein phosphatase 2A 65 kDa regulatory subunit A alpha isoformタンパク質・ペプチド : Striatin-3タンパク質・ペプチド : Serine/threonine-protein phosphatase 2A catalytic subunit alpha isoformタンパク質・ペプチド : MOB-like protein phoceinタンパク質・ペプチド : Striatin-interacting protein 1リガンド : MANGANESE (II) IONリガンド : ZINC IONリガンド : INOSITOL HEXAKISPHOSPHATE / / / 機能・相同性 分子機能 ドメイン・相同性 構成要素
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Homo sapiens (ヒト)手法 / / 解像度 : 3.3 Å Jeong B-C / Bai XC 資金援助 Organization Grant number 国 National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) GM132275 National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) CA220283 Welch Foundation I-1932 Welch Foundation I-1944 Welch Foundation I-1702
ジャーナル : Nat Struct Mol Biol / 年 : 2021タイトル : Cryo-EM structure of the Hippo signaling integrator human STRIPAK.著者 : Byung-Cheon Jeong / Sung Jun Bae / Lisheng Ni / Xuewu Zhang / Xiao-Chen Bai / Xuelian Luo / 要旨 : The striatin-interacting phosphatase and kinase (STRIPAK) complex is a large, multisubunit protein phosphatase 2A (PP2A) assembly that integrates diverse cellular signals in the Hippo pathway to ... The striatin-interacting phosphatase and kinase (STRIPAK) complex is a large, multisubunit protein phosphatase 2A (PP2A) assembly that integrates diverse cellular signals in the Hippo pathway to regulate cell proliferation and survival. The architecture and assembly mechanism of this critical complex are poorly understood. Using cryo-EM, we determine the structure of the human STRIPAK core comprising PP2AA, PP2AC, STRN3, STRIP1, and MOB4 at 3.2-Å resolution. Unlike the canonical trimeric PP2A holoenzyme, STRIPAK contains four copies of STRN3 and one copy of each the PP2AA-C heterodimer, STRIP1, and MOB4. The STRN3 coiled-coil domains form an elongated homotetrameric scaffold that links the complex together. An inositol hexakisphosphate (IP) is identified as a structural cofactor of STRIP1. Mutations of key residues at subunit interfaces disrupt the integrity of STRIPAK, causing aberrant Hippo pathway activation. Thus, STRIPAK is established as a noncanonical PP2A complex with four copies of regulatory STRN3 for enhanced signal integration. 履歴 登録 2020年9月10日 - ヘッダ(付随情報) 公開 2021年3月10日 - マップ公開 2021年3月10日 - 更新 2024年3月6日 - 現状 2024年3月6日 処理サイト : RCSB / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報 マップデータ
マップデータ 試料
試料 キーワード
キーワード phosphorylation (リン酸化) /
phosphorylation (リン酸化) /  complex /
complex /  PP2A (プロテインホスファターゼ2) /
PP2A (プロテインホスファターゼ2) /  SIGNALING PROTEIN
SIGNALING PROTEIN 機能・相同性情報
機能・相同性情報 armadillo repeat domain binding / meiotic spindle elongation / Integration of energy metabolism / PP2A-mediated dephosphorylation of key metabolic factors /
armadillo repeat domain binding / meiotic spindle elongation / Integration of energy metabolism / PP2A-mediated dephosphorylation of key metabolic factors /  regulation of microtubule binding / MASTL Facilitates Mitotic Progression / mitotic sister chromatid separation / regulation of meiotic cell cycle process involved in oocyte maturation / protein phosphatase type 2A complex ...FAR/SIN/STRIPAK complex /
regulation of microtubule binding / MASTL Facilitates Mitotic Progression / mitotic sister chromatid separation / regulation of meiotic cell cycle process involved in oocyte maturation / protein phosphatase type 2A complex ...FAR/SIN/STRIPAK complex /  armadillo repeat domain binding / meiotic spindle elongation / Integration of energy metabolism / PP2A-mediated dephosphorylation of key metabolic factors /
armadillo repeat domain binding / meiotic spindle elongation / Integration of energy metabolism / PP2A-mediated dephosphorylation of key metabolic factors /  regulation of microtubule binding / MASTL Facilitates Mitotic Progression / mitotic sister chromatid separation / regulation of meiotic cell cycle process involved in oocyte maturation / protein phosphatase type 2A complex / meiotic sister chromatid cohesion, centromeric / peptidyl-serine dephosphorylation / negative regulation of intracellular estrogen receptor signaling pathway / peptidyl-threonine dephosphorylation / positive regulation of microtubule binding / Regulation of glycolysis by fructose 2,6-bisphosphate metabolism / Inhibition of replication initiation of damaged DNA by RB1/E2F1 / female meiotic nuclear division / protein antigen binding / protein phosphatase regulator activity /
regulation of microtubule binding / MASTL Facilitates Mitotic Progression / mitotic sister chromatid separation / regulation of meiotic cell cycle process involved in oocyte maturation / protein phosphatase type 2A complex / meiotic sister chromatid cohesion, centromeric / peptidyl-serine dephosphorylation / negative regulation of intracellular estrogen receptor signaling pathway / peptidyl-threonine dephosphorylation / positive regulation of microtubule binding / Regulation of glycolysis by fructose 2,6-bisphosphate metabolism / Inhibition of replication initiation of damaged DNA by RB1/E2F1 / female meiotic nuclear division / protein antigen binding / protein phosphatase regulator activity /  GABA receptor binding / APC truncation mutants have impaired AXIN binding / AXIN missense mutants destabilize the destruction complex / Truncations of AMER1 destabilize the destruction complex / Initiation of Nuclear Envelope (NE) Reformation / ERKs are inactivated / negative regulation of epithelial to mesenchymal transition / positive regulation of extrinsic apoptotic signaling pathway in absence of ligand / Beta-catenin phosphorylation cascade / Signaling by GSK3beta mutants / CTNNB1 S33 mutants aren't phosphorylated / CTNNB1 S37 mutants aren't phosphorylated / CTNNB1 S45 mutants aren't phosphorylated / CTNNB1 T41 mutants aren't phosphorylated / regulation of cell morphogenesis / regulation of growth / Disassembly of the destruction complex and recruitment of AXIN to the membrane / cortical actin cytoskeleton organization / negative regulation of glycolytic process through fructose-6-phosphate / positive regulation of NLRP3 inflammasome complex assembly / Golgi cisterna membrane / myosin phosphatase activity / CTLA4 inhibitory signaling /
GABA receptor binding / APC truncation mutants have impaired AXIN binding / AXIN missense mutants destabilize the destruction complex / Truncations of AMER1 destabilize the destruction complex / Initiation of Nuclear Envelope (NE) Reformation / ERKs are inactivated / negative regulation of epithelial to mesenchymal transition / positive regulation of extrinsic apoptotic signaling pathway in absence of ligand / Beta-catenin phosphorylation cascade / Signaling by GSK3beta mutants / CTNNB1 S33 mutants aren't phosphorylated / CTNNB1 S37 mutants aren't phosphorylated / CTNNB1 S45 mutants aren't phosphorylated / CTNNB1 T41 mutants aren't phosphorylated / regulation of cell morphogenesis / regulation of growth / Disassembly of the destruction complex and recruitment of AXIN to the membrane / cortical actin cytoskeleton organization / negative regulation of glycolytic process through fructose-6-phosphate / positive regulation of NLRP3 inflammasome complex assembly / Golgi cisterna membrane / myosin phosphatase activity / CTLA4 inhibitory signaling /  protein serine/threonine phosphatase activity / Platelet sensitization by LDL / protein-serine/threonine phosphatase /
protein serine/threonine phosphatase activity / Platelet sensitization by LDL / protein-serine/threonine phosphatase /  regulation of cell differentiation / ERK/MAPK targets / T cell homeostasis / regulation of G1/S transition of mitotic cell cycle /
regulation of cell differentiation / ERK/MAPK targets / T cell homeostasis / regulation of G1/S transition of mitotic cell cycle /  phosphoprotein phosphatase activity / mesoderm development /
phosphoprotein phosphatase activity / mesoderm development /  chromosome, centromeric region / DARPP-32 events / lateral plasma membrane / negative regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction / Nonsense Mediated Decay (NMD) enhanced by the Exon Junction Complex (EJC) / Amplification of signal from unattached kinetochores via a MAD2 inhibitory signal / Cyclin A/B1/B2 associated events during G2/M transition / Mitotic Prometaphase / EML4 and NUDC in mitotic spindle formation / Loss of Nlp from mitotic centrosomes / Loss of proteins required for interphase microtubule organization from the centrosome / Recruitment of mitotic centrosome proteins and complexes / Resolution of Sister Chromatid Cohesion / cytoskeleton organization / Recruitment of NuMA to mitotic centrosomes / Anchoring of the basal body to the plasma membrane / AURKA Activation by TPX2 / protein dephosphorylation / protein phosphatase 2A binding / meiotic cell cycle /
chromosome, centromeric region / DARPP-32 events / lateral plasma membrane / negative regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction / Nonsense Mediated Decay (NMD) enhanced by the Exon Junction Complex (EJC) / Amplification of signal from unattached kinetochores via a MAD2 inhibitory signal / Cyclin A/B1/B2 associated events during G2/M transition / Mitotic Prometaphase / EML4 and NUDC in mitotic spindle formation / Loss of Nlp from mitotic centrosomes / Loss of proteins required for interphase microtubule organization from the centrosome / Recruitment of mitotic centrosome proteins and complexes / Resolution of Sister Chromatid Cohesion / cytoskeleton organization / Recruitment of NuMA to mitotic centrosomes / Anchoring of the basal body to the plasma membrane / AURKA Activation by TPX2 / protein dephosphorylation / protein phosphatase 2A binding / meiotic cell cycle /  protein tyrosine phosphatase activity /
protein tyrosine phosphatase activity /  chromosome segregation / RHO GTPases Activate Formins / response to lead ion /
chromosome segregation / RHO GTPases Activate Formins / response to lead ion /  regulation of protein phosphorylation / Spry regulation of FGF signaling / RAF activation / PKR-mediated signaling / Degradation of beta-catenin by the destruction complex / tau protein binding / positive regulation of protein serine/threonine kinase activity /
regulation of protein phosphorylation / Spry regulation of FGF signaling / RAF activation / PKR-mediated signaling / Degradation of beta-catenin by the destruction complex / tau protein binding / positive regulation of protein serine/threonine kinase activity /  small GTPase binding /
small GTPase binding /  kinase binding /
kinase binding /  紡錘体 / Negative regulation of MAPK pathway / Separation of Sister Chromatids / Cyclin D associated events in G1 / microtubule cytoskeleton /
紡錘体 / Negative regulation of MAPK pathway / Separation of Sister Chromatids / Cyclin D associated events in G1 / microtubule cytoskeleton /  Regulation of PLK1 Activity at G2/M Transition / Regulation of TP53 Degradation / response to estradiol / mitotic cell cycle / PI5P, PP2A and IER3 Regulate PI3K/AKT Signaling / protein-containing complex assembly /
Regulation of PLK1 Activity at G2/M Transition / Regulation of TP53 Degradation / response to estradiol / mitotic cell cycle / PI5P, PP2A and IER3 Regulate PI3K/AKT Signaling / protein-containing complex assembly /  樹状突起スパイン /
樹状突起スパイン /  calmodulin binding / intracellular signal transduction / neuron projection
calmodulin binding / intracellular signal transduction / neuron projection
 Homo sapiens (ヒト)
Homo sapiens (ヒト) 単粒子再構成法 /
単粒子再構成法 /  クライオ電子顕微鏡法 / 解像度: 3.3 Å
クライオ電子顕微鏡法 / 解像度: 3.3 Å  データ登録者
データ登録者 米国, 5件
米国, 5件  引用
引用 ジャーナル: Nat Struct Mol Biol / 年: 2021
ジャーナル: Nat Struct Mol Biol / 年: 2021
 構造の表示
構造の表示 ムービービューア
ムービービューア SurfView
SurfView Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク emd_22650.map.gz
emd_22650.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-22650-v30.xml
emd-22650-v30.xml emd-22650.xml
emd-22650.xml EMDBヘッダ
EMDBヘッダ emd_22650.png
emd_22650.png emd-22650.cif.gz
emd-22650.cif.gz emd_22650_additional_1.map.gz
emd_22650_additional_1.map.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-22650
http://ftp.pdbj.org/pub/emdb/structures/EMD-22650 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-22650
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-22650 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
マップ ダウンロード / ファイル: emd_22650.map.gz / 形式: CCP4 / 大きさ: 178 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_22650.map.gz / 形式: CCP4 / 大きさ: 178 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) 試料の構成要素
試料の構成要素
 Homo sapiens (ヒト)
Homo sapiens (ヒト)
 Homo sapiens (ヒト)
Homo sapiens (ヒト)
 Baculovirus expression vector pFastBac1-HM (バキュロウイルス科)
Baculovirus expression vector pFastBac1-HM (バキュロウイルス科)
 Homo sapiens (ヒト)
Homo sapiens (ヒト)
 Baculovirus expression vector pFastBac1-HM (バキュロウイルス科)
Baculovirus expression vector pFastBac1-HM (バキュロウイルス科)
 Homo sapiens (ヒト)
Homo sapiens (ヒト)
 Baculovirus expression vector pFastBac1-HM (バキュロウイルス科)
Baculovirus expression vector pFastBac1-HM (バキュロウイルス科)
 Homo sapiens (ヒト)
Homo sapiens (ヒト)
 Baculovirus expression vector pFastBac1-HM (バキュロウイルス科)
Baculovirus expression vector pFastBac1-HM (バキュロウイルス科)
 Homo sapiens (ヒト)
Homo sapiens (ヒト)
 Baculovirus expression vector pFastBac1-HM (バキュロウイルス科)
Baculovirus expression vector pFastBac1-HM (バキュロウイルス科)
 クライオ電子顕微鏡法
クライオ電子顕微鏡法 解析
解析 単粒子再構成法
単粒子再構成法 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN Bright-field microscopy / Cs: 2.7 mm
Bright-field microscopy / Cs: 2.7 mm
 画像解析
画像解析
 ムービー
ムービー コントローラー
コントローラー









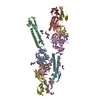



















 Z
Z Y
Y X
X