[English] 日本語
 Yorodumi
Yorodumi- PDB-2ygd: Molecular architectures of the 24meric eye lens chaperone alphaB-... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2ygd | ||||||
|---|---|---|---|---|---|---|---|
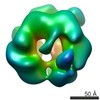
| Title | Molecular architectures of the 24meric eye lens chaperone alphaB- crystallin elucidated by a triple hybrid approach | ||||||
 Components Components | ALPHA-CRYSTALLIN B CHAIN | ||||||
 Keywords Keywords | CHAPERONE / PROTEIN AGGREGATION / HYBRID METHOD | ||||||
| Function / homology |  Function and homology information Function and homology informationmicrotubule polymerization or depolymerization / negative regulation of intracellular transport / apoptotic process involved in morphogenesis / cardiac myofibril / regulation of programmed cell death / tubulin complex assembly / structural constituent of eye lens / negative regulation of amyloid fibril formation / M band / lens development in camera-type eye ...microtubule polymerization or depolymerization / negative regulation of intracellular transport / apoptotic process involved in morphogenesis / cardiac myofibril / regulation of programmed cell death / tubulin complex assembly / structural constituent of eye lens / negative regulation of amyloid fibril formation / M band / lens development in camera-type eye / muscle organ development / actin filament bundle / HSF1-dependent transactivation / negative regulation of reactive oxygen species metabolic process / negative regulation of protein-containing complex assembly / muscle contraction / stress-activated MAPK cascade / synaptic membrane / response to hydrogen peroxide / negative regulation of cell growth / cellular response to gamma radiation / Z disc / unfolded protein binding / protein folding / response to estradiol / amyloid-beta binding / protein refolding / response to heat / microtubule binding / perikaryon / dendritic spine / lysosome / response to hypoxia / protein stabilization / axon / negative regulation of gene expression / negative regulation of DNA-templated transcription / negative regulation of apoptotic process / protein-containing complex binding / structural molecule activity / cell surface / protein homodimerization activity / protein-containing complex / mitochondrion / extracellular exosome / nucleoplasm / identical protein binding / nucleus / metal ion binding / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  HOMO SAPIENS (human) HOMO SAPIENS (human) | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 9.4 Å | ||||||
 Authors Authors | Braun, N. / Zacharias, M. / Peschek, J. / Kastenmueller, A. / Zou, J. / Hanzlik, M. / Haslbeck, M. / Rappsilber, J. / Buchner, J. / Weinkauf, S. | ||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2011 Journal: Proc Natl Acad Sci U S A / Year: 2011Title: Multiple molecular architectures of the eye lens chaperone αB-crystallin elucidated by a triple hybrid approach. Authors: Nathalie Braun / Martin Zacharias / Jirka Peschek / Andreas Kastenmüller / Juan Zou / Marianne Hanzlik / Martin Haslbeck / Juri Rappsilber / Johannes Buchner / Sevil Weinkauf /  Abstract: The molecular chaperone αB-crystallin, the major player in maintaining the transparency of the eye lens, prevents stress-damaged and aging lens proteins from aggregation. In nonlenticular cells, it ...The molecular chaperone αB-crystallin, the major player in maintaining the transparency of the eye lens, prevents stress-damaged and aging lens proteins from aggregation. In nonlenticular cells, it is involved in various neurological diseases, diabetes, and cancer. Given its structural plasticity and dynamics, structure analysis of αB-crystallin presented hitherto a formidable challenge. Here we present a pseudoatomic model of a 24-meric αB-crystallin assembly obtained by a triple hybrid approach combining data from cryoelectron microscopy, NMR spectroscopy, and structural modeling. The model, confirmed by cross-linking and mass spectrometry, shows that the subunits interact within the oligomer in different, defined conformations. We further present the molecular architectures of additional well-defined αB-crystallin assemblies with larger or smaller numbers of subunits, provide the mechanism how "heterogeneity" is achieved by a small set of defined structural variations, and analyze the factors modulating the oligomer equilibrium of αB-crystallin and thus its chaperone activity. | ||||||
| History |
| ||||||
| Remark 650 | HELIX DETERMINATION METHOD: AUTHOR PROVIDED. | ||||||
| Remark 700 | SHEET DETERMINATION METHOD: DSSP THE SHEETS PRESENTED AS "AC" IN EACH CHAIN ON SHEET RECORDS BELOW ... SHEET DETERMINATION METHOD: DSSP THE SHEETS PRESENTED AS "AC" IN EACH CHAIN ON SHEET RECORDS BELOW IS ACTUALLY AN 18-STRANDED BARREL THIS IS REPRESENTED BY A 19-STRANDED SHEET IN WHICH THE FIRST AND LAST STRANDS ARE IDENTICAL. THE SHEETS PRESENTED AS "GC" IN EACH CHAIN ON SHEET RECORDS BELOW IS ACTUALLY AN 18-STRANDED BARREL THIS IS REPRESENTED BY A 19-STRANDED SHEET IN WHICH THE FIRST AND LAST STRANDS ARE IDENTICAL. THE SHEETS PRESENTED AS "MC" IN EACH CHAIN ON SHEET RECORDS BELOW IS ACTUALLY AN 18-STRANDED BARREL THIS IS REPRESENTED BY A 19-STRANDED SHEET IN WHICH THE FIRST AND LAST STRANDS ARE IDENTICAL. THE SHEETS PRESENTED AS "SC" IN EACH CHAIN ON SHEET RECORDS BELOW IS ACTUALLY AN 18-STRANDED BARREL THIS IS REPRESENTED BY A 19-STRANDED SHEET IN WHICH THE FIRST AND LAST STRANDS ARE IDENTICAL. |
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2ygd.cif.gz 2ygd.cif.gz | 712.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2ygd.ent.gz pdb2ygd.ent.gz | 576.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2ygd.json.gz 2ygd.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  2ygd_validation.pdf.gz 2ygd_validation.pdf.gz | 952.7 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  2ygd_full_validation.pdf.gz 2ygd_full_validation.pdf.gz | 1 MB | Display | |
| Data in XML |  2ygd_validation.xml.gz 2ygd_validation.xml.gz | 103.4 KB | Display | |
| Data in CIF |  2ygd_validation.cif.gz 2ygd_validation.cif.gz | 148.8 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/yg/2ygd https://data.pdbj.org/pub/pdb/validation_reports/yg/2ygd ftp://data.pdbj.org/pub/pdb/validation_reports/yg/2ygd ftp://data.pdbj.org/pub/pdb/validation_reports/yg/2ygd | HTTPS FTP |
-Related structure data
| Related structure data |  1894MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 20191.930 Da / Num. of mol.: 24 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  HOMO SAPIENS (human) / Plasmid: PET28B / Production host: HOMO SAPIENS (human) / Plasmid: PET28B / Production host:  |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: HUMAN ALPHAB CRYSTALLIN / Type: COMPLEX |
|---|---|
| Buffer solution | Name: PBS BUFFER / pH: 7.4 / Details: PBS BUFFER |
| Specimen | Conc.: 0.2 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Specimen support | Details: HOLEY CARBON |
| Vitrification | Cryogen name: ETHANE Details: VITRIFICATION 1 -- CRYOGEN- ETHANE, HUMIDITY- 50, METHOD- BLOT FOR 1 SECOND BEFORE PLUNGING, |
- Electron microscopy imaging
Electron microscopy imaging
| Microscopy | Model: JEOL 2010HT |
|---|---|
| Electron gun | Electron source: LAB6 / Accelerating voltage: 120 kV / Illumination mode: SPOT SCAN |
| Electron lens | Mode: BRIGHT FIELD / Nominal magnification: 50000 X / Calibrated magnification: 47000 X / Nominal defocus max: 1500 nm / Nominal defocus min: 600 nm |
| Specimen holder | Temperature: 100 K |
| Image recording | Electron dose: 10 e/Å2 |
| Image scans | Num. digital images: 33 |
- Processing
Processing
| EM software | Name: IMAGIC / Category: 3D reconstruction | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Details: EACH MICROGRAPH, PHASE FLIPPING | ||||||||||||
| Symmetry | Point symmetry: C1 (asymmetric) | ||||||||||||
| 3D reconstruction | Method: PROJECTION MATCHING / Resolution: 9.4 Å / Num. of particles: 17560 / Nominal pixel size: 1.69 Å / Actual pixel size: 1.8 Å Details: SUBMISSION BASED ON EXPERIMENTAL DATA EMDB EMD-1894.(DEPOSITION ID: 7925). Symmetry type: POINT | ||||||||||||
| Atomic model building | PDB-ID: 2KLR Accession code: 2KLR / Source name: PDB / Type: experimental model | ||||||||||||
| Refinement | Highest resolution: 9.4 Å | ||||||||||||
| Refinement step | Cycle: LAST / Highest resolution: 9.4 Å
|
 Movie
Movie Controller
Controller


 UCSF Chimera
UCSF Chimera








 PDBj
PDBj


