[English] 日本語
 Yorodumi
Yorodumi- EMDB-1170: Structural roles for human translation factor eIF3 in initiation ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-1170 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structural roles for human translation factor eIF3 in initiation of protein synthesis. | |||||||||
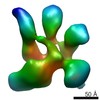
 Map data Map data | Cryo-EM density of human translation initiation factor eIF3. | |||||||||
 Sample Sample |
| |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / negative staining / Resolution: 33.0 Å | |||||||||
 Authors Authors | Siridechadilok B / Fraser CS / Hall RJ / Doudna JA / Nogales E | |||||||||
 Citation Citation |  Journal: Science / Year: 2005 Journal: Science / Year: 2005Title: Structural roles for human translation factor eIF3 in initiation of protein synthesis. Authors: Bunpote Siridechadilok / Christopher S Fraser / Richard J Hall / Jennifer A Doudna / Eva Nogales /  Abstract: Protein synthesis in mammalian cells requires initiation factor eIF3, a approximately 750-kilodalton complex that controls assembly of 40S ribosomal subunits on messenger RNAs (mRNAs) bearing either ...Protein synthesis in mammalian cells requires initiation factor eIF3, a approximately 750-kilodalton complex that controls assembly of 40S ribosomal subunits on messenger RNAs (mRNAs) bearing either a 5'-cap or an internal ribosome entry site (IRES). Cryo-electron microscopy reconstructions show that eIF3, a five-lobed particle, interacts with the hepatitis C virus (HCV) IRES RNA and the 5'-cap binding complex eIF4F via the same domain. Detailed modeling of eIF3 and eIF4F onto the 40S ribosomal subunit reveals that eIF3 uses eIF4F or the HCV IRES in structurally similar ways to position the mRNA strand near the exit site of 40S, promoting initiation complex assembly. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_1170.map.gz emd_1170.map.gz | 1.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-1170-v30.xml emd-1170-v30.xml emd-1170.xml emd-1170.xml | 8.4 KB 8.4 KB | Display Display |  EMDB header EMDB header |
| Images |  1170.gif 1170.gif | 13.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-1170 http://ftp.pdbj.org/pub/emdb/structures/EMD-1170 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1170 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1170 | HTTPS FTP |
-Validation report
| Summary document |  emd_1170_validation.pdf.gz emd_1170_validation.pdf.gz | 184.5 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_1170_full_validation.pdf.gz emd_1170_full_validation.pdf.gz | 183.6 KB | Display | |
| Data in XML |  emd_1170_validation.xml.gz emd_1170_validation.xml.gz | 5.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-1170 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-1170 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-1170 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-1170 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_1170.map.gz / Format: CCP4 / Size: 10.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_1170.map.gz / Format: CCP4 / Size: 10.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM density of human translation initiation factor eIF3. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 5.08 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Human eIF3
| Entire | Name: Human eIF3 |
|---|---|
| Components |
|
-Supramolecule #1000: Human eIF3
| Supramolecule | Name: Human eIF3 / type: sample / ID: 1000 / Details: human eIF3 purified from HeLa cytosol lysate. / Number unique components: 1 |
|---|---|
| Molecular weight | Theoretical: 750 KDa |
-Macromolecule #1: eIF3
| Macromolecule | Name: eIF3 / type: protein_or_peptide / ID: 1 / Number of copies: 12 / Oligomeric state: monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human / Cell: HeLa / Organelle: Cytosol / Location in cell: Cytoplasm Homo sapiens (human) / synonym: Human / Cell: HeLa / Organelle: Cytosol / Location in cell: Cytoplasm |
| Molecular weight | Experimental: 750 KDa |
| Recombinant expression | Organism: HeLa |
-Experimental details
-Structure determination
| Method | negative staining, cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.08 mg/mL |
|---|---|
| Buffer | pH: 7.5 Details: 20mM Tris pH7.5, 100mM KCl, 3mM MgCl2, 1mM DTT,0.1mM CHAPS, 2% Trehalose |
| Staining | Type: NEGATIVE / Details: cryo |
| Grid | Details: holey-carbon with thin layer of carbon |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 22 K / Instrument: OTHER / Details: Vitrification instrument: Vitrobot |
- Electron microscopy
Electron microscopy
| Microscope | FEI/PHILIPS CM200FEG |
|---|---|
| Image recording | Category: FILM / Film or detector model: KODAK SO-163 FILM / Digitization - Scanner: OTHER / Average electron dose: 20 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 50000 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.2 mm / Nominal magnification: 50000 |
| Sample stage | Specimen holder: eucentric / Specimen holder model: GATAN LIQUID NITROGEN |
- Image processing
Image processing
| CTF correction | Details: Phase flip |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 33.0 Å / Resolution method: FSC 0.5 CUT-OFF / Software - Name: SPIDER, IMAGIC / Number images used: 12000 |
 Movie
Movie Controller
Controller