+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7ins | ||||||
|---|---|---|---|---|---|---|---|
| Title | STRUCTURE OF PORCINE INSULIN COCRYSTALLIZED WITH CLUPEINE Z | ||||||
 Components Components |
| ||||||
 Keywords Keywords |  HORMONE HORMONE | ||||||
| Function / homology |  Function and homology information Function and homology informationInsulin processing / IRS activation / Signal attenuation / Insulin receptor signalling cascade / Signaling by Insulin receptor / Synthesis, secretion, and deacylation of Ghrelin / PI5P, PP2A and IER3 Regulate PI3K/AKT Signaling / Insulin receptor recycling / glycoprotein biosynthetic process / response to L-arginine ...Insulin processing / IRS activation / Signal attenuation / Insulin receptor signalling cascade / Signaling by Insulin receptor / Synthesis, secretion, and deacylation of Ghrelin / PI5P, PP2A and IER3 Regulate PI3K/AKT Signaling / Insulin receptor recycling / glycoprotein biosynthetic process / response to L-arginine / positive regulation of lipoprotein lipase activity / lactate biosynthetic process / lipoprotein biosynthetic process / positive regulation of fatty acid biosynthetic process / positive regulation of glucose metabolic process / COPI-mediated anterograde transport / lipid biosynthetic process / negative regulation of glycogen catabolic process / regulation of cellular amino acid metabolic process / nitric oxide-cGMP-mediated signaling / negative regulation of fatty acid metabolic process / negative regulation of feeding behavior / positive regulation of respiratory burst / positive regulation of dendritic spine maintenance / alpha-beta T cell activation / negative regulation of acute inflammatory response / negative regulation of respiratory burst involved in inflammatory response / negative regulation of protein secretion / fatty acid homeostasis / positive regulation of glycogen biosynthetic process / positive regulation of DNA replication / negative regulation of gluconeogenesis / positive regulation of nitric oxide mediated signal transduction / regulation of protein localization to plasma membrane / negative regulation of lipid catabolic process / positive regulation of insulin receptor signaling pathway / negative regulation of reactive oxygen species biosynthetic process / positive regulation of protein autophosphorylation /  insulin-like growth factor receptor binding / neuron projection maintenance / positive regulation of glycolytic process / positive regulation of mitotic nuclear division / positive regulation of cytokine production / acute-phase response / positive regulation of protein secretion / positive regulation of glucose import / negative regulation of proteolysis / insulin-like growth factor receptor binding / neuron projection maintenance / positive regulation of glycolytic process / positive regulation of mitotic nuclear division / positive regulation of cytokine production / acute-phase response / positive regulation of protein secretion / positive regulation of glucose import / negative regulation of proteolysis /  wound healing / wound healing /  insulin receptor binding / negative regulation of protein catabolic process / insulin receptor binding / negative regulation of protein catabolic process /  hormone activity / hormone activity /  vasodilation / positive regulation of protein localization to nucleus / glucose metabolic process / vasodilation / positive regulation of protein localization to nucleus / glucose metabolic process /  glucose homeostasis / insulin receptor signaling pathway / glucose homeostasis / insulin receptor signaling pathway /  protease binding / positive regulation of MAPK cascade / positive regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction / positive regulation of cell migration / G protein-coupled receptor signaling pathway / negative regulation of gene expression / positive regulation of cell population proliferation / protease binding / positive regulation of MAPK cascade / positive regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction / positive regulation of cell migration / G protein-coupled receptor signaling pathway / negative regulation of gene expression / positive regulation of cell population proliferation /  extracellular space / identical protein binding extracellular space / identical protein bindingSimilarity search - Function | ||||||
| Biological species |   Sus scrofa (pig) Sus scrofa (pig) | ||||||
| Method |  X-RAY DIFFRACTION / Resolution: 2 Å X-RAY DIFFRACTION / Resolution: 2 Å | ||||||
 Authors Authors | Balschmidt, P. / Hansen, F.B. / Dodson, E. / Dodson, G. / Korber, F. | ||||||
 Citation Citation |  Journal: Acta Crystallogr.,Sect.B / Year: 1991 Journal: Acta Crystallogr.,Sect.B / Year: 1991Title: Structure of porcine insulin cocrystallized with clupeine Z. Authors: Balschmidt, P. / Hansen, F.B. / Dodson, E.J. / Dodson, G.G. / Korber, F. #1:  Journal: Nature / Year: 1989 Journal: Nature / Year: 1989Title: Phenol Stabilizes More Helix in a New Symmetrical Zinc Insulin Hexamer Authors: Derewenda, U. / Derewenda, Z. / Dodson, E.J. / Dodson, G.G. / Reynolds, C.D. / Smith, G.D. / Sparks, C. / Swenson, D. #2:  Journal: Philos.Trans.R.Soc.London,Ser.B / Year: 1988 Journal: Philos.Trans.R.Soc.London,Ser.B / Year: 1988Title: The Structure of 2Zn Pig Insulin Crystals at 1.5 Angstroms Resolution Authors: Baker, E.N. / Blundell, T.L. / Cutfield, J.F. / Cutfield, S.M. / Dodson, E.J. / Dodson, G.G. / Hodgkin, D.M.C. / Hubbard, R.E. / Isaacs, N.W. / Reynolds, C.D. / Sakabe, K. / Sakabe, N. / Vijayan, N.M. | ||||||
| History |
| ||||||
| Remark 650 | HELIX THE B CHAIN HELIX IS EXTENDED TO THE FIRST SIX RESIDUES OF THE B CHAIN, THEREBY RUNNING FROM ...HELIX THE B CHAIN HELIX IS EXTENDED TO THE FIRST SIX RESIDUES OF THE B CHAIN, THEREBY RUNNING FROM B 1 TO B 20. |
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7ins.cif.gz 7ins.cif.gz | 49.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7ins.ent.gz pdb7ins.ent.gz | 36.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7ins.json.gz 7ins.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/in/7ins https://data.pdbj.org/pub/pdb/validation_reports/in/7ins ftp://data.pdbj.org/pub/pdb/validation_reports/in/7ins ftp://data.pdbj.org/pub/pdb/validation_reports/in/7ins | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| 3 | 
| ||||||||
| 4 | 
| ||||||||
| 5 |
| ||||||||
| 6 | 
| ||||||||
| 7 | 
| ||||||||
| 8 | 
| ||||||||
| 9 | 
| ||||||||
| 10 | 
| ||||||||
| 11 | 
| ||||||||
| 12 | 
| ||||||||
| Unit cell |
| ||||||||
| Atom site foot note | 1: GLY G 21 - GLY G 22 OMEGA ANGLE = 149.010 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION 2: LYS G 33 - GLY G 35 OMEGA ANGLE = 303.917 PEPTIDE BOND DEVIATES SIGNIFICANTLY FROM TRANS CONFORMATION |
- Components
Components
-INSULIN (CHAIN ... , 2 types, 6 molecules ACEBDF
| #1: Protein/peptide | Mass: 2383.698 Da / Num. of mol.: 3 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Sus scrofa (pig) / References: UniProt: P01315 Sus scrofa (pig) / References: UniProt: P01315#2: Protein/peptide | Mass: 3403.927 Da / Num. of mol.: 3 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Sus scrofa (pig) / References: UniProt: P01315 Sus scrofa (pig) / References: UniProt: P01315 |
|---|
-Protein/peptide , 1 types, 1 molecules G
| #3: Protein/peptide | Mass: 1379.692 Da / Num. of mol.: 1 / Source method: obtained synthetically |
|---|
-Non-polymers , 4 types, 115 molecules 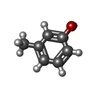

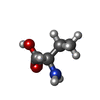




| #4: Chemical |  M-Cresol M-Cresol#5: Chemical | ChemComp-ZN / | #6: Chemical | #7: Water | ChemComp-HOH / |  Water Water |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.27 Å3/Da / Density % sol: 45.72 % | ||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Crystal grow | *PLUS Temperature: 277 K / pH: 7.3 / Method: batch method | ||||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Reflection | *PLUS % possible obs: 75 % / Rmerge(I) obs: 0.071 |
|---|
- Processing
Processing
| Software | Name: PROLSQ / Classification: refinement | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Rfactor obs: 0.194 / Highest resolution: 2 Å Details: CHAIN *G* IS THE GENERAL PROTAMINE CHAIN. THE PROTAMINE RESIDUES HAVE BEEN SELECTED TO FIT THE ELECTRON DENSITY WITHOUT ANY REFERENCE TO THE SEQUENCE OR CONTINUITY OF THE CLUPEINE Z. FOR ...Details: CHAIN *G* IS THE GENERAL PROTAMINE CHAIN. THE PROTAMINE RESIDUES HAVE BEEN SELECTED TO FIT THE ELECTRON DENSITY WITHOUT ANY REFERENCE TO THE SEQUENCE OR CONTINUITY OF THE CLUPEINE Z. FOR THIS REASON SOME ATOMS HAVE ZERO OCCUPANCY. THE SEQRES RECORDS FOR CHAIN *G* PRESENT THE ACTUAL SEQUENCE OF CLUPEINE Z. THE ATOM RECORDS FOR CHAIN *G* HAVE BEEN PRESENTED WITH RESIDUE NAME *UNK*. THE FOLLOWING PROTAMINE RESIDUES BELONG (LOOSELY) TOGETHER: ASSOCIATED WITH THE DIMER-DIMER INTERFACE G41, G 25, G 5, G 6, G 43; ASSOCIATED WITH THE TOP OF THE CENTRAL CAVITY G 8, G 21 - G 23, G 33. G 35 CA, C, O HAVE BEEN FITTED INTO DENSITY FOR THE UNIDENTIFIED ZINC LIGAND, NOTE THE OCCUPANCY FOR THE OTHER ATOMS OF G 35 IS ZERO. THERE IS AN APPROXIMATE THREEFOLD AXIS THROUGH (26.72, 26.72, 0) WITH DIRECTION COSINES (0.543, -0.543, 0.640). | ||||||||||||
| Refinement step | Cycle: LAST / Highest resolution: 2 Å
| ||||||||||||
| Software | *PLUS Name: PROLSQ / Classification: refinement | ||||||||||||
| Refinement | *PLUS Highest resolution: 2 Å / Lowest resolution: 10 Å / Num. reflection obs: 11522 / Rfactor obs: 0.194 | ||||||||||||
| Solvent computation | *PLUS | ||||||||||||
| Displacement parameters | *PLUS Biso mean: 33.7 Å2 |
 Movie
Movie Controller
Controller










 PDBj
PDBj







