[English] 日本語
 Yorodumi
Yorodumi- PDB-4pad: Binding of chloromethyl ketone substrate analogues to crystalline... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4pad | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Binding of chloromethyl ketone substrate analogues to crystalline papain | ||||||||||||
 Components Components | PAPAIN | ||||||||||||
 Keywords Keywords | HYDROLASE/HYDROLASE INHIBITOR /  SULFHYDRYL PROTEINASE / HYDROLASE-HYDROLASE INHIBITOR complex SULFHYDRYL PROTEINASE / HYDROLASE-HYDROLASE INHIBITOR complex | ||||||||||||
| Function / homology |  Function and homology information Function and homology information papain / papain /  serpin family protein binding / cysteine-type peptidase activity / serpin family protein binding / cysteine-type peptidase activity /  proteolysis proteolysisSimilarity search - Function | ||||||||||||
| Biological species |   Carica papaya (papaya) Carica papaya (papaya) | ||||||||||||
| Method |  X-RAY DIFFRACTION / Resolution: 2.8 Å X-RAY DIFFRACTION / Resolution: 2.8 Å | ||||||||||||
 Authors Authors | Drenth, J. | ||||||||||||
 Citation Citation |  Journal: Biochemistry / Year: 1976 Journal: Biochemistry / Year: 1976Title: Binding of chloromethyl ketone substrate analogues to crystalline papain. Authors: Drenth, J. / Kalk, K.H. / Swen, H.M. #1:  Journal: Adv.Protein Chem. / Year: 1971 Journal: Adv.Protein Chem. / Year: 1971Title: The Structure of Papain Authors: Drenth, J. / Jansonius, J.N. / Koekoek, R. / Wolthers, B.G. #2:  Journal: Philos.Trans.R.Soc.London,Ser.B / Year: 1970 Journal: Philos.Trans.R.Soc.London,Ser.B / Year: 1970Title: The Structure of the Papain Molecule Authors: Drenth, J. / Jansonius, J.N. / Koekoek, R. / Sluyterman, L.A.A. / Wolthers, B.G. #3:  Journal: Nature / Year: 1968 Journal: Nature / Year: 1968Title: Structure of Papain Authors: Drenth, J. / Jansonius, J.N. / Koekoek, R. / Swen, H.M. / Wolthers, B.G. | ||||||||||||
| History |
| ||||||||||||
| Remark 700 | SHEET THE SHEET SUBSTRUCTURE OF THIS MOLECULE IS DOUBLY BIFURCATED. IN ORDER TO REPRESENT THIS ...SHEET THE SHEET SUBSTRUCTURE OF THIS MOLECULE IS DOUBLY BIFURCATED. IN ORDER TO REPRESENT THIS FEATURE IN THE SHEET RECORDS BELOW, TWO SHEETS ARE DEFINED. STRANDS 4,5,6 OF S1A ARE IDENTICAL TO STRANDS 2,3,4 OF S1B. |
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4pad.cif.gz 4pad.cif.gz | 52.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4pad.ent.gz pdb4pad.ent.gz | 36.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4pad.json.gz 4pad.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/pa/4pad https://data.pdbj.org/pub/pdb/validation_reports/pa/4pad ftp://data.pdbj.org/pub/pdb/validation_reports/pa/4pad ftp://data.pdbj.org/pub/pdb/validation_reports/pa/4pad | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Atom site foot note | 1: RESIDUE 152 IS A CIS PROLINE. 2: SOME COORDINATES WERE AFFECTED BY THE BINDING OF THE INHIBITOR. |
- Components
Components
| #1: Protein |  Mass: 23449.346 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Carica papaya (papaya) / References: UniProt: P00784, Carica papaya (papaya) / References: UniProt: P00784,  papain papain |
|---|---|
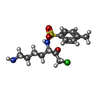
| #2: Chemical | ChemComp-TCK / |
| #3: Water | ChemComp-HOH /  Water Water |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.51 Å3/Da / Density % sol: 51.01 % | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Crystal grow | *PLUS Temperature: 4 ℃ / Method: vapor diffusion | |||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Radiation | Scattering type: x-ray |
|---|---|
| Radiation wavelength | Relative weight: 1 |
- Processing
Processing
| Refinement | Highest resolution: 2.8 Å Details: THE COORDINATES OF THE INHIBITOR WERE NOT REFINED. THIS DATA SET WAS OBTAINED BY MERGING THE INHIBITOR COORDINATES WITH THE REFINED NATIVE PAPAIN COORDINATES OF SET 8PAP. THE CONFORMATION OF ...Details: THE COORDINATES OF THE INHIBITOR WERE NOT REFINED. THIS DATA SET WAS OBTAINED BY MERGING THE INHIBITOR COORDINATES WITH THE REFINED NATIVE PAPAIN COORDINATES OF SET 8PAP. THE CONFORMATION OF RESIDUE CYS 25 IN THIS DERIVATIVE STRUCTURE IS NOT THE SAME AS THAT IN THE NATIVE ENZYME AND THIS FACT IS REFLECTED IN THE COORDINATES | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement step | Cycle: LAST / Highest resolution: 2.8 Å
|
 Movie
Movie Controller
Controller
















 PDBj
PDBj