[English] 日本語
 Yorodumi
Yorodumi- PDB-4dfr: CRYSTAL STRUCTURES OF ESCHERICHIA COLI AND LACTOBACILLUS CASEI DI... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4dfr | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | CRYSTAL STRUCTURES OF ESCHERICHIA COLI AND LACTOBACILLUS CASEI DIHYDROFOLATE REDUCTASE REFINED AT 1.7 ANGSTROMS RESOLUTION. I. GENERAL FEATURES AND BINDING OF METHOTREXATE | |||||||||
 Components Components | DIHYDROFOLATE REDUCTASE | |||||||||
 Keywords Keywords |  OXIDOREDUCTASE / OXIDO-REDUCTASE OXIDOREDUCTASE / OXIDO-REDUCTASE | |||||||||
| Function / homology |  Function and homology information Function and homology information methotrexate binding / methotrexate binding /  dihydrofolic acid binding / response to methotrexate / dihydrofolate metabolic process / dihydrofolic acid binding / response to methotrexate / dihydrofolate metabolic process /  NADP+ binding / NADP+ binding /  folic acid binding / folic acid metabolic process / glycine biosynthetic process / folic acid binding / folic acid metabolic process / glycine biosynthetic process /  dihydrofolate reductase / dihydrofolate reductase /  dihydrofolate reductase activity ... dihydrofolate reductase activity ... methotrexate binding / methotrexate binding /  dihydrofolic acid binding / response to methotrexate / dihydrofolate metabolic process / dihydrofolic acid binding / response to methotrexate / dihydrofolate metabolic process /  NADP+ binding / NADP+ binding /  folic acid binding / folic acid metabolic process / glycine biosynthetic process / folic acid binding / folic acid metabolic process / glycine biosynthetic process /  dihydrofolate reductase / dihydrofolate reductase /  dihydrofolate reductase activity / dihydrofolate reductase activity /  NADPH binding / tetrahydrofolate biosynthetic process / one-carbon metabolic process / NADPH binding / tetrahydrofolate biosynthetic process / one-carbon metabolic process /  NADP binding / response to xenobiotic stimulus / response to antibiotic / NADP binding / response to xenobiotic stimulus / response to antibiotic /  cytosol cytosolSimilarity search - Function | |||||||||
| Biological species |   Escherichia coli (E. coli) Escherichia coli (E. coli) | |||||||||
| Method |  X-RAY DIFFRACTION / Resolution: 1.7 Å X-RAY DIFFRACTION / Resolution: 1.7 Å | |||||||||
 Authors Authors | Filman, D.J. / Matthews, D.A. / Bolin, J.T. / Kraut, J. | |||||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 1982 Journal: J.Biol.Chem. / Year: 1982Title: Crystal structures of Escherichia coli and Lactobacillus casei dihydrofolate reductase refined at 1.7 A resolution. I. General features and binding of methotrexate. Authors: Bolin, J.T. / Filman, D.J. / Matthews, D.A. / Hamlin, R.C. / Kraut, J. #1:  Journal: Biochemistry / Year: 1987 Journal: Biochemistry / Year: 1987Title: Effect of Single Amino Acid Replacements on the Folding and Stability of Dihydrofolate Reductase from Escherichia Coli Authors: Perry, K.M. / Onuffer, J.J. / Touchette, N.A. / Herndon, C.S. / Gittelman, M.S. / Matthews, C.R. / Chen, J.-T. / Mayer, R.J. / Taira, K. / Benkovic, S.J. / Howell, E.E. / Kraut, J. #2:  Journal: J.Biol.Chem. / Year: 1982 Journal: J.Biol.Chem. / Year: 1982Title: Crystal Structures of Escherichia Coli and Lactobacillus Casei Dihydrofolate Reductase Refined at 1.7 Angstroms Resolution. II. Environment of Bound Nadph and Implications for Catalysis Authors: Filman, D.J. / Bolin, J.T. / Matthews, D.A. / Kraut, J. #3:  Journal: J.Biol.Chem. / Year: 1982 Journal: J.Biol.Chem. / Year: 1982Title: Crystal Structure of Avian Dihydrofolate Reductase Containing Phenyltriazine and Nadph Authors: Volz, K.W. / Matthews, D.A. / Alden, R.A. / Freer, S.T. / Hansch, C. / Kaufman, B.T. / Kraut, J. #4:  Journal: Biochemistry / Year: 1979 Journal: Biochemistry / Year: 1979Title: Interpretation of Nuclear Magnetic Resonance Spectra for Lactobacillus Casei Dihydrofolate Reductase Based on the X-Ray Structure of the Enzyme-Methotrexate-Nadph Complex Authors: Matthews, D.A. #5:  Journal: J.Biol.Chem. / Year: 1979 Journal: J.Biol.Chem. / Year: 1979Title: Dihydrofolate Reductase from Lactobacillus Casei. Stereochemistry of Nadph Binding Authors: Matthews, D.A. / Alden, R.A. / Freer, S.T. / Xuong, N.-H. / Kraut, J. #6:  Journal: J.Biol.Chem. / Year: 1979 Journal: J.Biol.Chem. / Year: 1979Title: Proton Magnetic Resonance Studies on Escherichia Coli Dihydrofolate Reductase. Assignment of Histidine C-2 Protons in Binary Complexes with Folates on the Basis of the Crystal Structure with ...Title: Proton Magnetic Resonance Studies on Escherichia Coli Dihydrofolate Reductase. Assignment of Histidine C-2 Protons in Binary Complexes with Folates on the Basis of the Crystal Structure with Methotrexate and on Chemical Modifications Authors: Poe, M. / Hoogsteen, K. / Matthews, D.A. #7:  Journal: J.Biol.Chem. / Year: 1978 Journal: J.Biol.Chem. / Year: 1978Title: Dihydrofolate Reductase from Lactobacillus Casei. X-Ray Structure of the Enzyme-Methotrexate-Nadph Complex Authors: Matthews, D.A. / Alden, R.A. / Bolin, J.T. / Filman, D.J. / Freer, S.T. / Hamlin, R. / Hol, W.G.J. / Kisliuk, R.L. / Pastore, E.J. / Plante, L.T. / Xuong, N.-H. / Kraut, J. #8:  Journal: Biochemistry / Year: 1978 Journal: Biochemistry / Year: 1978Title: Dihydrofolate Reductase. The Amino Acid Sequence of the Enzyme from a Methotrexate-Resistant Mutant of Escherichia Coli Authors: Bennett, C.D. / Rodkey, J.A. / Sondey, J.M. / Hirschmann, R. #9:  Journal: Science / Year: 1977 Journal: Science / Year: 1977Title: Dihydrofolate Reductase. X-Ray Structure of the Binary Complex with Methotrexate Authors: Matthews, D.A. / Alden, R.A. / Bolin, J.T. / Freer, S.T. / Hamlin, R. / Xuong, N. / Kraut, J. / Poe, M. / Williams, M. / Hoogsteen, K. #10:  Journal: Biochemistry / Year: 1972 Journal: Biochemistry / Year: 1972Title: Dihydrofolate Reductase. Purification and Characterization of the Enzyme from an Amethopterin-Resistant Mutant of Escherichia Coli Authors: Poe, M. / Greenfield, N.J. / Hirshfield, J.M. / Williams, M.N. / Hoogsteen, K. | |||||||||
| History |
|
- Structure visualization
Structure visualization
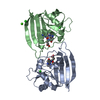
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4dfr.cif.gz 4dfr.cif.gz | 101.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4dfr.ent.gz pdb4dfr.ent.gz | 67.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4dfr.json.gz 4dfr.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/df/4dfr https://data.pdbj.org/pub/pdb/validation_reports/df/4dfr ftp://data.pdbj.org/pub/pdb/validation_reports/df/4dfr ftp://data.pdbj.org/pub/pdb/validation_reports/df/4dfr | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Atom site foot note | 1: SEE REMARK 8. / 2: SEE REMARK 9. / 3: SEE REMARK 10. / 4: SEE REMARK 11. | ||||||||
| Noncrystallographic symmetry (NCS) | NCS oper: (Code: given Matrix: (-0.92116, -0.37966, 0.636818), Vector  : : Details | THE MTRIX TRANSFORMATION PRESENTED BELOW WILL SUPERIMPOSE MOLECULE B ON MOLECULE A. | |
- Components
Components
| #1: Protein |  Mass: 18020.393 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Escherichia coli (E. coli) / Strain: B / References: UniProt: P0ABQ4, Escherichia coli (E. coli) / Strain: B / References: UniProt: P0ABQ4,  dihydrofolate reductase dihydrofolate reductase#2: Chemical |  Chloride Chloride#3: Chemical |  Methotrexate Methotrexate#4: Chemical | ChemComp-CA / | #5: Water | ChemComp-HOH / |  Water WaterSequence details | RESIDUE 142 IS LISTED AS ASN IN THE SEQUENCE PAPER (SEE REFERENCE 9 ABOVE). THE X-RAY STRUCTURE ...RESIDUE 142 IS LISTED AS ASN IN THE SEQUENCE PAPER (SEE REFERENCE 9 ABOVE). THE X-RAY STRUCTURE SUGGESTS IT IS ASP (EVIDENCE IS INTERMOLEC | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.56 Å3/Da / Density % sol: 51.93 % | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Crystal grow | *PLUS Method: unknown | ||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Radiation | Scattering type: x-ray |
|---|---|
| Radiation wavelength | Relative weight: 1 |
| Reflection | *PLUS Highest resolution: 1.7 Å / Num. obs: 32554 / % possible obs: 88 % |
- Processing
Processing
| Refinement | Rfactor Rwork : 0.155 / Highest resolution: 1.7 Å : 0.155 / Highest resolution: 1.7 ÅDetails: MOLECULE DESIGNATED AS CHAIN B BELOW IS PREFERRED FOR STRUCTURAL COMPARISONS BECAUSE IT IS MORE COMPLETE AND LESS PERTURBED BY INTERMOLECULAR CONTACTS. ALTERNATE LOCATIONS *A* AND *B* ARE ...Details: MOLECULE DESIGNATED AS CHAIN B BELOW IS PREFERRED FOR STRUCTURAL COMPARISONS BECAUSE IT IS MORE COMPLETE AND LESS PERTURBED BY INTERMOLECULAR CONTACTS. ALTERNATE LOCATIONS *A* AND *B* ARE PARTIALLY OCCUPIED CONFORMATIONS FOR RESIDUES SER A 64, SER A 150, HIS B 45, SER B 64 AND ASP B 122. IN ALL CASES, *A* IS BELIEVED TO BE THE MAJOR CONFORMER. NEITHER THE OCCUPANCIES NOR THE THERMAL PARAMETERS SHOULD BE CONSIDERED AS RELIABLE. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement step | Cycle: LAST / Highest resolution: 1.7 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Highest resolution: 1.7 Å / Num. reflection all: 32554 / Rfactor obs: 0.155 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS |
 Movie
Movie Controller
Controller















 PDBj
PDBj