[English] 日本語
 Yorodumi
Yorodumi- PDB-2y65: Crystal structure of Drosophila melanogaster kinesin-1 motor doma... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2y65 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of Drosophila melanogaster kinesin-1 motor domain dimer-tail complex | ||||||
 Components Components | (KINESIN HEAVY CHAIN ) x 2 ) x 2 | ||||||
 Keywords Keywords |  MOTOR PROTEIN MOTOR PROTEIN | ||||||
| Function / homology |  Function and homology information Function and homology information : / actin filament bundle organization / anterograde axonal transport of mitochondrion / anterograde dendritic transport / mitochondrion distribution / larval locomotory behavior / oocyte microtubule cytoskeleton polarization / eye photoreceptor cell differentiation / regulation of pole plasm oskar mRNA localization / pole plasm oskar mRNA localization ... : / actin filament bundle organization / anterograde axonal transport of mitochondrion / anterograde dendritic transport / mitochondrion distribution / larval locomotory behavior / oocyte microtubule cytoskeleton polarization / eye photoreceptor cell differentiation / regulation of pole plasm oskar mRNA localization / pole plasm oskar mRNA localization ... : / actin filament bundle organization / anterograde axonal transport of mitochondrion / anterograde dendritic transport / mitochondrion distribution / larval locomotory behavior / oocyte microtubule cytoskeleton polarization / eye photoreceptor cell differentiation / regulation of pole plasm oskar mRNA localization / pole plasm oskar mRNA localization / oocyte dorsal/ventral axis specification / pole plasm assembly / microtubule sliding / dorsal appendage formation / larval somatic muscle development / COPI-dependent Golgi-to-ER retrograde traffic / Kinesins / transport along microtubule / centrosome separation / anterograde dendritic transport of neurotransmitter receptor complex / actin cap / microtubule plus-end / plus-end-directed microtubule motor activity / axo-dendritic transport / stress granule disassembly / nuclear migration / dendrite morphogenesis / : / actin filament bundle organization / anterograde axonal transport of mitochondrion / anterograde dendritic transport / mitochondrion distribution / larval locomotory behavior / oocyte microtubule cytoskeleton polarization / eye photoreceptor cell differentiation / regulation of pole plasm oskar mRNA localization / pole plasm oskar mRNA localization / oocyte dorsal/ventral axis specification / pole plasm assembly / microtubule sliding / dorsal appendage formation / larval somatic muscle development / COPI-dependent Golgi-to-ER retrograde traffic / Kinesins / transport along microtubule / centrosome separation / anterograde dendritic transport of neurotransmitter receptor complex / actin cap / microtubule plus-end / plus-end-directed microtubule motor activity / axo-dendritic transport / stress granule disassembly / nuclear migration / dendrite morphogenesis /  kinesin complex / synaptic vesicle transport / kinesin complex / synaptic vesicle transport /  microtubule motor activity / microtubule motor activity /  tropomyosin binding / intracellular distribution of mitochondria / microtubule-based movement / tropomyosin binding / intracellular distribution of mitochondria / microtubule-based movement /  microtubule polymerization / cytoskeletal motor activity / axon cytoplasm / dendrite cytoplasm / microtubule polymerization / cytoskeletal motor activity / axon cytoplasm / dendrite cytoplasm /  axonogenesis / axonogenesis /  axon guidance / axon guidance /  microtubule binding / microtubule binding /  microtubule / microtubule /  ATP hydrolysis activity / ATP hydrolysis activity /  ATP binding / ATP binding /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |   DROSOPHILA MELANOGASTER (fruit fly) DROSOPHILA MELANOGASTER (fruit fly) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.2 Å MOLECULAR REPLACEMENT / Resolution: 2.2 Å | ||||||
 Authors Authors | Kaan, H.Y.K. / Hackney, D.D. / Kozielski, F. | ||||||
 Citation Citation |  Journal: Science / Year: 2011 Journal: Science / Year: 2011Title: The Structure of the Kinesin-1 Motor-Tail Complex Reveals the Mechanism of Autoinhibition. Authors: Kaan, H.Y.K. / Hackney, D.D. / Kozielski, F. | ||||||
| History |
| ||||||
| Remark 700 | SHEET THE SHEET STRUCTURE OF THIS MOLECULE IS BIFURCATED. IN ORDER TO REPRESENT THIS FEATURE IN ... SHEET THE SHEET STRUCTURE OF THIS MOLECULE IS BIFURCATED. IN ORDER TO REPRESENT THIS FEATURE IN THE SHEET RECORDS BELOW, TWO SHEETS ARE DEFINED. |
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2y65.cif.gz 2y65.cif.gz | 301.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2y65.ent.gz pdb2y65.ent.gz | 243.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2y65.json.gz 2y65.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/y6/2y65 https://data.pdbj.org/pub/pdb/validation_reports/y6/2y65 ftp://data.pdbj.org/pub/pdb/validation_reports/y6/2y65 ftp://data.pdbj.org/pub/pdb/validation_reports/y6/2y65 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  2y5wSC S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 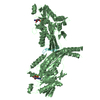
| ||||||||
| 3 | 
| ||||||||
| Unit cell |
| ||||||||
| Noncrystallographic symmetry (NCS) | NCS oper: (Code: given Matrix: (-0.9805, 0.0458, -0.1911), Vector  : : Details | OWING TO ITS AMINO ACID SEQUENCE, CHAINS X AND Y ARE ALMOST SYMMETRICAL, IN TERMS OF SIDE CHAIN PROPERTIES, ABOUT THE LYS 944 RESIDUE. THIS DISTINCTIVE FEATURE OF THE CHAINS X AND Y ALLOWS THEM TO BIND IN TWO DIRECTIONS BETWEEN THE DIMERS A-A' AND B-B', WHICH ALSO EXHIBIT TWO-FOLD SYMMETRY. | |
- Components
Components
| #1: Protein |  / KINESIN / KINESINMass: 40885.301 Da / Num. of mol.: 4 / Fragment: MOTOR DOMAIN, RESIDUES 1-365 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   DROSOPHILA MELANOGASTER (fruit fly) / Production host: DROSOPHILA MELANOGASTER (fruit fly) / Production host:   ESCHERICHIA COLI (E. coli) / Strain (production host): BL21 / Variant (production host): CODONPLUS / References: UniProt: P17210 ESCHERICHIA COLI (E. coli) / Strain (production host): BL21 / Variant (production host): CODONPLUS / References: UniProt: P17210#2: Protein/peptide |  / KINESIN / KINESINMass: 1913.098 Da / Num. of mol.: 3 / Fragment: TAIL DOMAIN RESIDUES 937-952 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   DROSOPHILA MELANOGASTER (fruit fly) / Production host: DROSOPHILA MELANOGASTER (fruit fly) / Production host:   ESCHERICHIA COLI (E. coli) / References: UniProt: P17210 ESCHERICHIA COLI (E. coli) / References: UniProt: P17210#3: Chemical | ChemComp-ADP /  Adenosine diphosphate Adenosine diphosphate#4: Chemical | ChemComp-MG / #5: Water | ChemComp-HOH / |  Water Water |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.15 Å3/Da / Density % sol: 42.87 % / Description: NONE |
|---|---|
Crystal grow | pH: 7.4 Details: 18% POLYETHYLENE GLYCOL-3350, 0.2M POTASSIUM CHLORIDE, 0.1M HEPES SODIUM PH 7.4 |
-Data collection
| Diffraction | Mean temperature: 93 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SLS SLS  / Beamline: X06SA / Wavelength: 1 / Beamline: X06SA / Wavelength: 1 |
| Detector | Type: DECTRIS PILATUS 6M / Detector: PIXEL / Date: Oct 16, 2010 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1 Å / Relative weight: 1 : 1 Å / Relative weight: 1 |
| Reflection | Resolution: 2.2→30 Å / Num. obs: 80517 / % possible obs: 99.9 % / Observed criterion σ(I): 2 / Redundancy: 8.8 % / Biso Wilson estimate: 41.383 Å2 / Rmerge(I) obs: 0.06 / Net I/σ(I): 21.7 |
| Reflection shell | Resolution: 2.2→2.32 Å / Redundancy: 8.3 % / Rmerge(I) obs: 0.37 / Mean I/σ(I) obs: 5.5 / % possible all: 99.6 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 2Y5W Resolution: 2.2→50 Å / Cor.coef. Fo:Fc: 0.955 / Cor.coef. Fo:Fc free: 0.924 / SU B: 5.611 / SU ML: 0.147 / Cross valid method: THROUGHOUT / ESU R: 0.276 / ESU R Free: 0.221 / Stereochemistry target values: MAXIMUM LIKELIHOOD Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS. U VALUES REFINED INDIVIDUALLY. PSEUDOSYMMETRY IN THE CRYSTAL STRUCTURE, WHERE CHAINS X AND Y LIE ON THE TWO-FOLD CRYSTALLOGRAPHIC SYMMETRY AXIS.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.4 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 42.405 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.2→50 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj








