[English] 日本語
 Yorodumi
Yorodumi- PDB-2vxw: Structural and Functional Studies of the Potent Anti-HIV Chemokin... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2vxw | ||||||
|---|---|---|---|---|---|---|---|
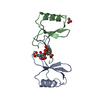
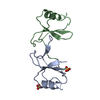
| Title | Structural and Functional Studies of the Potent Anti-HIV Chemokine Variant P2-RANTES | ||||||
 Components Components | C-C MOTIF CHEMOKINE 5 Chemokine Chemokine | ||||||
 Keywords Keywords |  IMMUNE SYSTEM / IMMUNE SYSTEM /  GLYCOPROTEIN / CELL-CELL FUSION / GLYCOPROTEIN / CELL-CELL FUSION /  INFLAMMATORY RESPONSE / HIV ENTRY / INFLAMMATORY RESPONSE / HIV ENTRY /  CHEMOTAXIS CHEMOTAXIS | ||||||
| Function / homology |  Function and homology information Function and homology informationregulation of chronic inflammatory response / CCR4 chemokine receptor binding / chemokine receptor antagonist activity / activation of phospholipase D activity / positive regulation of cellular biosynthetic process / CCR1 chemokine receptor binding / positive regulation of natural killer cell chemotaxis / negative regulation of macrophage apoptotic process /  chemokine receptor binding / positive regulation of cell-cell adhesion mediated by integrin ...regulation of chronic inflammatory response / CCR4 chemokine receptor binding / chemokine receptor antagonist activity / activation of phospholipase D activity / positive regulation of cellular biosynthetic process / CCR1 chemokine receptor binding / positive regulation of natural killer cell chemotaxis / negative regulation of macrophage apoptotic process / chemokine receptor binding / positive regulation of cell-cell adhesion mediated by integrin ...regulation of chronic inflammatory response / CCR4 chemokine receptor binding / chemokine receptor antagonist activity / activation of phospholipase D activity / positive regulation of cellular biosynthetic process / CCR1 chemokine receptor binding / positive regulation of natural killer cell chemotaxis / negative regulation of macrophage apoptotic process /  chemokine receptor binding / positive regulation of cell-cell adhesion mediated by integrin / positive regulation of activation of Janus kinase activity / receptor signaling protein tyrosine kinase activator activity / positive regulation of homotypic cell-cell adhesion / CCR5 chemokine receptor binding / positive regulation of T cell chemotaxis / negative regulation of G protein-coupled receptor signaling pathway / CCR chemokine receptor binding / lymphocyte chemotaxis / chemokine receptor binding / positive regulation of cell-cell adhesion mediated by integrin / positive regulation of activation of Janus kinase activity / receptor signaling protein tyrosine kinase activator activity / positive regulation of homotypic cell-cell adhesion / CCR5 chemokine receptor binding / positive regulation of T cell chemotaxis / negative regulation of G protein-coupled receptor signaling pathway / CCR chemokine receptor binding / lymphocyte chemotaxis /  neutrophil activation / phosphatidylinositol phospholipase C activity / negative regulation of T cell apoptotic process / positive regulation of T cell apoptotic process / positive regulation of calcium ion transport / eosinophil chemotaxis / positive regulation of innate immune response / cellular response to fibroblast growth factor stimulus / chemokine-mediated signaling pathway / positive regulation of monocyte chemotaxis / Chemokine receptors bind chemokines / neutrophil activation / phosphatidylinositol phospholipase C activity / negative regulation of T cell apoptotic process / positive regulation of T cell apoptotic process / positive regulation of calcium ion transport / eosinophil chemotaxis / positive regulation of innate immune response / cellular response to fibroblast growth factor stimulus / chemokine-mediated signaling pathway / positive regulation of monocyte chemotaxis / Chemokine receptors bind chemokines /  chemokine activity / dendritic cell chemotaxis / chemokine activity / dendritic cell chemotaxis /  regulation of T cell activation / leukocyte cell-cell adhesion / positive regulation of macrophage chemotaxis / negative regulation of viral genome replication / phospholipase activator activity / macrophage chemotaxis / positive regulation of smooth muscle cell migration / regulation of T cell activation / leukocyte cell-cell adhesion / positive regulation of macrophage chemotaxis / negative regulation of viral genome replication / phospholipase activator activity / macrophage chemotaxis / positive regulation of smooth muscle cell migration /  exocytosis / exocytosis /  chemoattractant activity / Interleukin-10 signaling / monocyte chemotaxis / positive regulation of translational initiation / regulation of insulin secretion / positive regulation of cell adhesion / negative regulation by host of viral transcription / positive regulation of T cell migration / positive regulation of viral genome replication / cellular response to interleukin-1 / positive regulation of phosphorylation / positive regulation of tyrosine phosphorylation of STAT protein / positive regulation of T cell proliferation / chemoattractant activity / Interleukin-10 signaling / monocyte chemotaxis / positive regulation of translational initiation / regulation of insulin secretion / positive regulation of cell adhesion / negative regulation by host of viral transcription / positive regulation of T cell migration / positive regulation of viral genome replication / cellular response to interleukin-1 / positive regulation of phosphorylation / positive regulation of tyrosine phosphorylation of STAT protein / positive regulation of T cell proliferation /  neutrophil chemotaxis / epithelial cell proliferation / positive regulation of epithelial cell proliferation / positive regulation of smooth muscle cell proliferation / positive regulation of receptor signaling pathway via JAK-STAT / response to virus / intracellular calcium ion homeostasis / response to toxic substance / cellular response to virus / cellular response to type II interferon / : / calcium ion transport / neutrophil chemotaxis / epithelial cell proliferation / positive regulation of epithelial cell proliferation / positive regulation of smooth muscle cell proliferation / positive regulation of receptor signaling pathway via JAK-STAT / response to virus / intracellular calcium ion homeostasis / response to toxic substance / cellular response to virus / cellular response to type II interferon / : / calcium ion transport /  chemotaxis / cell-cell signaling / cellular response to tumor necrosis factor / G alpha (i) signalling events / positive regulation of ERK1 and ERK2 cascade / positive regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction / chemotaxis / cell-cell signaling / cellular response to tumor necrosis factor / G alpha (i) signalling events / positive regulation of ERK1 and ERK2 cascade / positive regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction /  protein kinase activity / positive regulation of cell migration / protein kinase activity / positive regulation of cell migration /  inflammatory response / G protein-coupled receptor signaling pathway / protein homodimerization activity / inflammatory response / G protein-coupled receptor signaling pathway / protein homodimerization activity /  extracellular space / extracellular region / identical protein binding / extracellular space / extracellular region / identical protein binding /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |   HOMO SAPIENS (human) HOMO SAPIENS (human) | ||||||
| Method |  X-RAY DIFFRACTION / OTHER / Resolution: 1.7 Å X-RAY DIFFRACTION / OTHER / Resolution: 1.7 Å | ||||||
 Authors Authors | Jin, H. / Li, P. / LiWang, P.J. | ||||||
 Citation Citation |  Journal: Proteins / Year: 2010 Journal: Proteins / Year: 2010Title: Structural and Functional Studies of the Potent Anti-HIV Chemokine Variant P2-Rantes. Authors: Jin, H. / Kagiampakis, I. / Li, P. / Liwang, P.J. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2vxw.cif.gz 2vxw.cif.gz | 67.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2vxw.ent.gz pdb2vxw.ent.gz | 54 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2vxw.json.gz 2vxw.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/vx/2vxw https://data.pdbj.org/pub/pdb/validation_reports/vx/2vxw ftp://data.pdbj.org/pub/pdb/validation_reports/vx/2vxw ftp://data.pdbj.org/pub/pdb/validation_reports/vx/2vxw | HTTPS FTP |
|---|
-Related structure data
| Related structure data | |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein |  Chemokine / P2-RANTES / SMALL-INDUCIBLE CYTOKINE A5 / SIS-DELTA / T CELL-SPECIFIC PROTEIN P228 / TCP228 / T- ...P2-RANTES / SMALL-INDUCIBLE CYTOKINE A5 / SIS-DELTA / T CELL-SPECIFIC PROTEIN P228 / TCP228 / T-CELL-SPECIFIC PROTEIN RANTES / EOSINOPHIL-CHEMOTACTIC CYTOKINE / EOCP Chemokine / P2-RANTES / SMALL-INDUCIBLE CYTOKINE A5 / SIS-DELTA / T CELL-SPECIFIC PROTEIN P228 / TCP228 / T- ...P2-RANTES / SMALL-INDUCIBLE CYTOKINE A5 / SIS-DELTA / T CELL-SPECIFIC PROTEIN P228 / TCP228 / T-CELL-SPECIFIC PROTEIN RANTES / EOSINOPHIL-CHEMOTACTIC CYTOKINE / EOCPMass: 7918.121 Da / Num. of mol.: 4 / Fragment: CHEMOKINE, RESIDUES 33-91 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   HOMO SAPIENS (human) / Plasmid: PET32A / Production host: HOMO SAPIENS (human) / Plasmid: PET32A / Production host:   ESCHERICHIA COLI (E. coli) / Strain (production host): BL21(DE3) / References: UniProt: P13501 ESCHERICHIA COLI (E. coli) / Strain (production host): BL21(DE3) / References: UniProt: P13501#2: Water | ChemComp-HOH / |  Water Water |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.43 Å3/Da / Density % sol: 48.9 % / Description: NONE |
|---|---|
Crystal grow | pH: 5.5 / Details: pH 5.5 |
-Data collection
| Diffraction | Mean temperature: 287 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU MICROMAX-007 HF / Wavelength: 1.5616 ROTATING ANODE / Type: RIGAKU MICROMAX-007 HF / Wavelength: 1.5616 |
| Detector | Type: RIGAKU MICROMAX |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.5616 Å / Relative weight: 1 : 1.5616 Å / Relative weight: 1 |
| Reflection | Resolution: 1.7→50 Å / Num. obs: 34463 / % possible obs: 97.4 % / Observed criterion σ(I): 2 / Redundancy: 3.4 % / Biso Wilson estimate: 17.3 Å2 / Rmerge(I) obs: 0.05 / Net I/σ(I): 12 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : OTHER : OTHERStarting model: NONE Resolution: 1.7→50 Å / Rfactor Rfree error: 0.017 / Data cutoff high absF: 10000 / Cross valid method: THROUGHOUT / σ(F): 0
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Bsol: 65.9606 Å2 / ksol: 0.341826 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 16.8 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.7→50 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2→2.12 Å / Rfactor Rfree error: 0.042 / Total num. of bins used: 6
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xplor file |
|
 Movie
Movie Controller
Controller

















 PDBj
PDBj


