[English] 日本語
 Yorodumi
Yorodumi- PDB-1p12: CRYSTAL STRUCTURES OF ALPHA-LYTIC PROTEASE COMPLEXES WITH IRREVER... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1p12 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | CRYSTAL STRUCTURES OF ALPHA-LYTIC PROTEASE COMPLEXES WITH IRREVERSIBLY BOUND PHOSPHONATE ESTERS | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | HYDROLASE/HYDROLASE INHIBITOR /  SERINE PROTEINASE / HYDROLASE-HYDROLASE INHIBITOR complex SERINE PROTEINASE / HYDROLASE-HYDROLASE INHIBITOR complex | |||||||||
| Function / homology |  Function and homology information Function and homology information alpha-lytic endopeptidase / serine-type endopeptidase activity / alpha-lytic endopeptidase / serine-type endopeptidase activity /  proteolysis / extracellular region proteolysis / extracellular regionSimilarity search - Function | |||||||||
| Biological species |   Lysobacter enzymogenes (bacteria) Lysobacter enzymogenes (bacteria) | |||||||||
| Method |  X-RAY DIFFRACTION / Resolution: 1.9 Å X-RAY DIFFRACTION / Resolution: 1.9 Å | |||||||||
 Authors Authors | Bone, R. / Agard, D.A. | |||||||||
 Citation Citation |  Journal: Biochemistry / Year: 1991 Journal: Biochemistry / Year: 1991Title: Crystal structures of alpha-lytic protease complexes with irreversibly bound phosphonate esters. Authors: Bone, R. / Sampson, N.S. / Bartlett, P.A. / Agard, D.A. #1:  Journal: Biochemistry / Year: 1991 Journal: Biochemistry / Year: 1991Title: Peptidic Phosphonylating Agents as Irreversible Inhibitors of Serine Proteases and Models of the Tetrahedral Intermediates Authors: Sampson, N.S. / Bartlett, P.A. #2:  Journal: J.Mol.Biol. / Year: 1985 Journal: J.Mol.Biol. / Year: 1985Title: Refined Structure of Alpha-Lytic Protease at 1.7 Angstroms Resolution. Analysis of Hydrogen Bonding and Solvent Structure Authors: Fujinaga, M. / Delbaere, L.T.J. / Brayer, G.D. / James, M.N.G. #3:  Journal: J.Mol.Biol. / Year: 1979 Journal: J.Mol.Biol. / Year: 1979Title: Molecular Structure of the Alpha-Lytic Protease from Myxobacter 495 at 2.8 Angstroms Resolution Authors: Brayer, G.D. / Delbaere, L.T.J. / James, M.N.G. | |||||||||
| History |
|
- Structure visualization
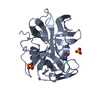
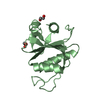
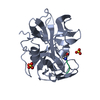
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1p12.cif.gz 1p12.cif.gz | 56.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1p12.ent.gz pdb1p12.ent.gz | 38.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1p12.json.gz 1p12.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/p1/1p12 https://data.pdbj.org/pub/pdb/validation_reports/p1/1p12 ftp://data.pdbj.org/pub/pdb/validation_reports/p1/1p12 ftp://data.pdbj.org/pub/pdb/validation_reports/p1/1p12 | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Atom site foot note | 1: PRO E 99A IS A CIS PROLINE. / 2: SEE REMARK 5. / 3: SEE REMARK 6. |
- Components
Components
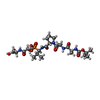
| #1: Protein |  Alpha-lytic endopeptidase Alpha-lytic endopeptidaseMass: 19875.131 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Lysobacter enzymogenes (bacteria) / Production host: Lysobacter enzymogenes (bacteria) / Production host:   Escherichia coli (E. coli) / References: UniProt: P00778, Escherichia coli (E. coli) / References: UniProt: P00778,  alpha-lytic endopeptidase alpha-lytic endopeptidase |
|---|---|
| #2: Protein/peptide | |
| #3: Chemical | ChemComp-SO4 /  Sulfate Sulfate |
| #4: Water | ChemComp-HOH /  Water Water |
| Compound details | THE PVA RESIDUE IN THE INHIBITOR IS THE ALPHA-AMINO PHOSPHONIC ACID ANALOG OF VAL IN WHICH THE C- ...THE PVA RESIDUE IN THE INHIBITOR IS THE ALPHA-AMINO PHOSPHONIC |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.51 Å3/Da / Density % sol: 50.95 % | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Crystal grow | *PLUS pH: 7.2 / Method: vapor diffusion | ||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Radiation | Scattering type: x-ray |
|---|---|
| Radiation wavelength | Relative weight: 1 |
- Processing
Processing
| Software | Name: PROLSQ / Classification: refinement | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Rfactor obs: 0.124 / Highest resolution: 1.9 Å | ||||||||||||
| Refinement step | Cycle: LAST / Highest resolution: 1.9 Å
| ||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller










 PDBj
PDBj