+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1j8k | ||||||
|---|---|---|---|---|---|---|---|
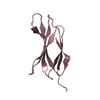
| Title | NMR STRUCTURE OF THE FIBRONECTIN EDA DOMAIN, NMR, 20 STRUCTURES | ||||||
 Components Components | FIBRONECTIN | ||||||
 Keywords Keywords |  PROTEIN BINDING / EDA / PROTEIN BINDING / EDA /  FIBRONECTIN / TYPEIII DOMAIN FIBRONECTIN / TYPEIII DOMAIN | ||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of monocyte activation / calcium-independent cell-matrix adhesion / negative regulation of transforming growth factor beta production / Fibronectin matrix formation / Extracellular matrix organization / positive regulation of substrate-dependent cell migration, cell attachment to substrate / neural crest cell migration involved in autonomic nervous system development / peptidase activator activity /  fibrinogen complex / peptide cross-linking ...negative regulation of monocyte activation / calcium-independent cell-matrix adhesion / negative regulation of transforming growth factor beta production / Fibronectin matrix formation / Extracellular matrix organization / positive regulation of substrate-dependent cell migration, cell attachment to substrate / neural crest cell migration involved in autonomic nervous system development / peptidase activator activity / fibrinogen complex / peptide cross-linking ...negative regulation of monocyte activation / calcium-independent cell-matrix adhesion / negative regulation of transforming growth factor beta production / Fibronectin matrix formation / Extracellular matrix organization / positive regulation of substrate-dependent cell migration, cell attachment to substrate / neural crest cell migration involved in autonomic nervous system development / peptidase activator activity /  fibrinogen complex / peptide cross-linking / integrin activation / ALK mutants bind TKIs / cell-substrate junction assembly / biological process involved in interaction with symbiont / Molecules associated with elastic fibres / fibrinogen complex / peptide cross-linking / integrin activation / ALK mutants bind TKIs / cell-substrate junction assembly / biological process involved in interaction with symbiont / Molecules associated with elastic fibres /  proteoglycan binding / extracellular matrix structural constituent / MET activates PTK2 signaling / Syndecan interactions / p130Cas linkage to MAPK signaling for integrins / endodermal cell differentiation / GRB2:SOS provides linkage to MAPK signaling for Integrins / Non-integrin membrane-ECM interactions / Signaling by ALK fusions and activated point mutants / proteoglycan binding / extracellular matrix structural constituent / MET activates PTK2 signaling / Syndecan interactions / p130Cas linkage to MAPK signaling for integrins / endodermal cell differentiation / GRB2:SOS provides linkage to MAPK signaling for Integrins / Non-integrin membrane-ECM interactions / Signaling by ALK fusions and activated point mutants /  basement membrane / basement membrane /  endoplasmic reticulum-Golgi intermediate compartment / ECM proteoglycans / positive regulation of axon extension / Integrin cell surface interactions / Nuclear events stimulated by ALK signaling in cancer / endoplasmic reticulum-Golgi intermediate compartment / ECM proteoglycans / positive regulation of axon extension / Integrin cell surface interactions / Nuclear events stimulated by ALK signaling in cancer /  collagen binding / collagen binding /  extracellular matrix / Degradation of the extracellular matrix / cell-matrix adhesion / Integrin signaling / substrate adhesion-dependent cell spreading / regulation of ERK1 and ERK2 cascade / platelet alpha granule lumen / integrin-mediated signaling pathway / extracellular matrix / Degradation of the extracellular matrix / cell-matrix adhesion / Integrin signaling / substrate adhesion-dependent cell spreading / regulation of ERK1 and ERK2 cascade / platelet alpha granule lumen / integrin-mediated signaling pathway /  Post-translational protein phosphorylation / acute-phase response / Cell surface interactions at the vascular wall / Post-translational protein phosphorylation / acute-phase response / Cell surface interactions at the vascular wall /  regulation of protein phosphorylation / Signaling by high-kinase activity BRAF mutants / regulation of protein phosphorylation / Signaling by high-kinase activity BRAF mutants /  wound healing / MAP2K and MAPK activation / response to wounding / Regulation of Insulin-like Growth Factor (IGF) transport and uptake by Insulin-like Growth Factor Binding Proteins (IGFBPs) / GPER1 signaling / Signaling by RAF1 mutants / Signaling by moderate kinase activity BRAF mutants / Paradoxical activation of RAF signaling by kinase inactive BRAF / Signaling downstream of RAS mutants / positive regulation of fibroblast proliferation / Signaling by BRAF and RAF1 fusions / wound healing / MAP2K and MAPK activation / response to wounding / Regulation of Insulin-like Growth Factor (IGF) transport and uptake by Insulin-like Growth Factor Binding Proteins (IGFBPs) / GPER1 signaling / Signaling by RAF1 mutants / Signaling by moderate kinase activity BRAF mutants / Paradoxical activation of RAF signaling by kinase inactive BRAF / Signaling downstream of RAS mutants / positive regulation of fibroblast proliferation / Signaling by BRAF and RAF1 fusions /  integrin binding / Platelet degranulation / integrin binding / Platelet degranulation /  heparin binding / heparin binding /  nervous system development / nervous system development /  heart development / regulation of cell shape / collagen-containing extracellular matrix / heart development / regulation of cell shape / collagen-containing extracellular matrix /  angiogenesis / blood microparticle / Interleukin-4 and Interleukin-13 signaling / angiogenesis / blood microparticle / Interleukin-4 and Interleukin-13 signaling /  protease binding / positive regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction / protease binding / positive regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction /  cell adhesion / apical plasma membrane / cell adhesion / apical plasma membrane /  endoplasmic reticulum lumen / endoplasmic reticulum lumen /  signaling receptor binding / positive regulation of cell population proliferation / positive regulation of gene expression / signaling receptor binding / positive regulation of cell population proliferation / positive regulation of gene expression /  extracellular space / extracellular exosome / extracellular region / identical protein binding / extracellular space / extracellular exosome / extracellular region / identical protein binding /  plasma membrane plasma membraneSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  SOLUTION NMR / SOLUTION NMR /  simulated annealing simulated annealing | ||||||
 Authors Authors | Niimi, T. / Osawa, M. / Yamaji, N. / Yasunaga, K. / Sakashita, H. / Mase, T. / Tanaka, A. / Fujita, S. | ||||||
 Citation Citation |  Journal: J.Biomol.NMR / Year: 2001 Journal: J.Biomol.NMR / Year: 2001Title: NMR structure of human fibronectin EDA. Authors: Niimi, T. / Osawa, M. / Yamaji, N. / Yasunaga, K. / Sakashita, H. / Mase, T. / Tanaka, A. / Fujita, S. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1j8k.cif.gz 1j8k.cif.gz | 544.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1j8k.ent.gz pdb1j8k.ent.gz | 471.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1j8k.json.gz 1j8k.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/j8/1j8k https://data.pdbj.org/pub/pdb/validation_reports/j8/1j8k ftp://data.pdbj.org/pub/pdb/validation_reports/j8/1j8k ftp://data.pdbj.org/pub/pdb/validation_reports/j8/1j8k | HTTPS FTP |
|---|
-Related structure data
| Similar structure data | |
|---|---|
| Other databases |
|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein |  Mass: 10222.121 Da / Num. of mol.: 1 / Fragment: EXTRA DOMAIN 2 (RESIDUES 1631-1724) Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Plasmid: PET28 / Species (production host): Escherichia coli / Production host: Homo sapiens (human) / Plasmid: PET28 / Species (production host): Escherichia coli / Production host:   Escherichia coli BL21(DE3) (bacteria) / Strain (production host): BL21(DE3) / References: UniProt: P02751 Escherichia coli BL21(DE3) (bacteria) / Strain (production host): BL21(DE3) / References: UniProt: P02751 |
|---|
-Experimental details
-Experiment
| Experiment | Method:  SOLUTION NMR SOLUTION NMR | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
| ||||||||||||||||
| NMR details | Text: THREE-DIMENSIONAL STRUCTURE IN SOLUTION REPRESENTED BY 20 CONFORMERS DETERMINED BY NUCLEAR MAGNETIC RESONANCE, TORSION ANGLE DYNAMICS AND RESTRAINED ENERGY REFINEMENT. |
- Sample preparation
Sample preparation
| Details | Contents: 2mM EDA U-15N,13C; 50mM phosphate buffer, 400mM sodium sulfate; 90% H2O, 10% D2O Solvent system: 90% H2O/10% D2O |
|---|---|
| Sample conditions | Ionic strength: 400mM sodium sulfate / pH: 7.0 / Pressure: 1 atm / Temperature: 293 K |
Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M |
|---|---|
| Radiation wavelength | Relative weight: 1 |
| NMR spectrometer | Type: Bruker AMX / Manufacturer: Bruker / Model : AMX / Field strength: 600 MHz : AMX / Field strength: 600 MHz |
- Processing
Processing
| NMR software |
| ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method:  simulated annealing / Software ordinal: 1 simulated annealing / Software ordinal: 1 Details: THESE STRUCTURES CONSIST OF 1289 UPPER LIMITS ON DISTANCES OBTAINED FROM NOE MEASUREMENTS AND HTDROGEN EXCHANGE MEASUREMENTS AND 51 TORSION ANGLE CONSTRAINTS OBTAINED FROM COUPLING CONSTANT ...Details: THESE STRUCTURES CONSIST OF 1289 UPPER LIMITS ON DISTANCES OBTAINED FROM NOE MEASUREMENTS AND HTDROGEN EXCHANGE MEASUREMENTS AND 51 TORSION ANGLE CONSTRAINTS OBTAINED FROM COUPLING CONSTANT AND CHEMICAL SHIFT INDEX | ||||||||||||||||
| NMR ensemble | Conformer selection criteria: structures with the least restraint violations Conformers calculated total number: 50 / Conformers submitted total number: 20 |
 Movie
Movie Controller
Controller












 PDBj
PDBj
















