+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1i2u | ||||||
|---|---|---|---|---|---|---|---|
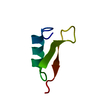
| Title | NMR SOLUTION STRUCTURES OF ANTIFUNGAL HELIOMICIN | ||||||
 Components Components | DEFENSIN HELIOMICIN | ||||||
 Keywords Keywords |  ANTIFUNGAL PROTEIN / ANTIFUNGAL PROTEIN /  alpha-beta protein / CSab motif (cysteine stabilized alpha-helix beta-sheet motif) alpha-beta protein / CSab motif (cysteine stabilized alpha-helix beta-sheet motif) | ||||||
| Function / homology |  Function and homology information Function and homology informationdefense response to fungus / killing of cells of another organism /  innate immune response / extracellular region innate immune response / extracellular regionSimilarity search - Function | ||||||
| Biological species |   Heliothis virescens (tobacco budworm) Heliothis virescens (tobacco budworm) | ||||||
| Method |  SOLUTION NMR / torsion angle dynamics (DYANA), energy minimisation (XPLOR) SOLUTION NMR / torsion angle dynamics (DYANA), energy minimisation (XPLOR) | ||||||
 Authors Authors | Lamberty, M. / Caille, A. / Landon, C. / Tassin-Moindrot, S. / Hetru, C. / Bulet, P. / Vovelle, F. | ||||||
 Citation Citation |  Journal: Biochemistry / Year: 2001 Journal: Biochemistry / Year: 2001Title: Solution structures of the antifungal heliomicin and a selected variant with both antibacterial and antifungal activities. Authors: Lamberty, M. / Caille, A. / Landon, C. / Tassin-Moindrot, S. / Hetru, C. / Bulet, P. / Vovelle, F. #1:  Journal: J.Biol.Chem. / Year: 1999 Journal: J.Biol.Chem. / Year: 1999Title: Insect Immunity. Isolation from the Lepidopteran Heliothis virescens of a Novel Insect Defensin with Potent Antifungal Activity Authors: LAMBERTY, M. / ADES, S. / UTTENWEILER-JOSEPH, S. / BROOKHART, G. / BUSHEY, D. / HOFFMANN, J.A. / BULET, P. | ||||||
| History |
|
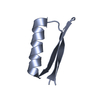
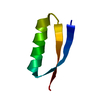
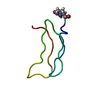
- Structure visualization
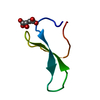
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1i2u.cif.gz 1i2u.cif.gz | 226.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1i2u.ent.gz pdb1i2u.ent.gz | 194.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1i2u.json.gz 1i2u.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/i2/1i2u https://data.pdbj.org/pub/pdb/validation_reports/i2/1i2u ftp://data.pdbj.org/pub/pdb/validation_reports/i2/1i2u ftp://data.pdbj.org/pub/pdb/validation_reports/i2/1i2u | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein/peptide | Mass: 4796.368 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Heliothis virescens (tobacco budworm) / Plasmid: PSEA2 / Production host: Heliothis virescens (tobacco budworm) / Plasmid: PSEA2 / Production host:   Saccharomyces cerevisiae (brewer's yeast) / Strain (production host): TGY 48-1 / References: UniProt: P81544 Saccharomyces cerevisiae (brewer's yeast) / Strain (production host): TGY 48-1 / References: UniProt: P81544 |
|---|
-Experimental details
-Experiment
| Experiment | Method:  SOLUTION NMR SOLUTION NMR |
|---|---|
| NMR experiment | Type : 2D 1H DQF- : 2D 1H DQF- COSY; TQF- COSY; TQF- COSY; TOCSY; COSY; TOCSY;  NOESY NOESY |
| NMR details | Text: This structure was determined using standard 2D homonuclear techniques. |
- Sample preparation
Sample preparation
| Details | Contents: 4 mM heliomicin ; 40mM sodium acetate buffer / Solvent system: 90% H2O/10% D2O |
|---|---|
| Sample conditions | Ionic strength: 40mM sodium acetate buffer / pH: 4.3 / Pressure: ambient / Temperature: 293 K |
Crystal grow | *PLUS Method: other / Details: NMR |
-NMR measurement
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M |
|---|---|
| Radiation wavelength | Relative weight: 1 |
| NMR spectrometer | Type: Bruker AMX / Manufacturer: Bruker / Model : AMX / Field strength: 500 MHz : AMX / Field strength: 500 MHz |
- Processing
Processing
| NMR software |
| |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: torsion angle dynamics (DYANA), energy minimisation (XPLOR) Software ordinal: 1 Details: the structures are based on a total of 130 intra-residue, 163 sequential, 105 medium range, 198 long range NOE-derived distance restraints, 9 distances restraints corresponding to the 3 ...Details: the structures are based on a total of 130 intra-residue, 163 sequential, 105 medium range, 198 long range NOE-derived distance restraints, 9 distances restraints corresponding to the 3 disulfide bridge, 85 dihedral angle restraints, and 14 distance restraints for hydrogen bonds | |||||||||||||||
| NMR representative | Selection criteria: lowest target function | |||||||||||||||
| NMR ensemble | Conformer selection criteria: structures with acceptable covalent geometry, structures with favorable non-bond energy, structures with the least restraint violations, structures with the lowest energy, target function Conformers calculated total number: 40 / Conformers submitted total number: 18 |
 Movie
Movie Controller
Controller







 PDBj
PDBj

