+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of BCL10 R58Q filament | |||||||||
 Map data Map data | CryoEM structure of BCL10 R58Q | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | filament / CBM complex / IMMUNE SYSTEM | |||||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of lymphotoxin A production / polkadots / CBM complex / antifungal innate immune response / protein kinase B binding / T cell apoptotic process / positive regulation of mast cell cytokine production / CARD domain binding / programmed cell death / negative regulation of mature B cell apoptotic process ...positive regulation of lymphotoxin A production / polkadots / CBM complex / antifungal innate immune response / protein kinase B binding / T cell apoptotic process / positive regulation of mast cell cytokine production / CARD domain binding / programmed cell death / negative regulation of mature B cell apoptotic process / B cell apoptotic process / positive regulation of extrinsic apoptotic signaling pathway / response to food / non-canonical NF-kappaB signal transduction / toll-like receptor signaling pathway / positive regulation of T cell receptor signaling pathway / cytoplasmic microtubule / positive regulation of cysteine-type endopeptidase activity involved in apoptotic process / positive regulation of phosphorylation / general transcription initiation factor binding / immunological synapse / NF-kappaB binding / immunoglobulin mediated immune response / cellular defense response / lipopolysaccharide-mediated signaling pathway / positive regulation of protein ubiquitination / neural tube closure / positive regulation of interleukin-8 production / Activation of NF-kappaB in B cells / cellular response to mechanical stimulus / protein homooligomerization / CLEC7A (Dectin-1) signaling / FCERI mediated NF-kB activation / positive regulation of interleukin-6 production / positive regulation of T cell activation / Downstream TCR signaling / E3 ubiquitin ligases ubiquitinate target proteins / positive regulation of NF-kappaB transcription factor activity / protein-macromolecule adaptor activity / T cell receptor signaling pathway / positive regulation of canonical NF-kappaB signal transduction / protease binding / cellular response to lipopolysaccharide / transcription coactivator activity / adaptive immune response / lysosome / membrane raft / positive regulation of apoptotic process / innate immune response / ubiquitin protein ligase binding / positive regulation of DNA-templated transcription / protein-containing complex binding / perinuclear region of cytoplasm / protein-containing complex / identical protein binding / nucleus / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | helical reconstruction / cryo EM / Resolution: 4.6 Å | |||||||||
 Authors Authors | David L / Wu H | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Cancer Discov / Year: 2022 Journal: Cancer Discov / Year: 2022Title: BCL10 Mutations Define Distinct Dependencies Guiding Precision Therapy for DLBCL. Authors: Min Xia / Liron David / Matt Teater / Johana Gutierrez / Xiang Wang / Cem Meydan / Andrew Lytle / Graham W Slack / David W Scott / Ryan D Morin / Ozlem Onder / Kojo S J Elenitoba-Johnson / ...Authors: Min Xia / Liron David / Matt Teater / Johana Gutierrez / Xiang Wang / Cem Meydan / Andrew Lytle / Graham W Slack / David W Scott / Ryan D Morin / Ozlem Onder / Kojo S J Elenitoba-Johnson / Nahuel Zamponi / Leandro Cerchietti / Tianbao Lu / Ulrike Philippar / Lorena Fontan / Hao Wu / Ari M Melnick /    Abstract: Activated B cell-like diffuse large B-cell lymphomas (ABC-DLBCL) have unfavorable outcomes and chronic activation of CARD11-BCL10-MALT1 (CBM) signal amplification complexes that form due to ...Activated B cell-like diffuse large B-cell lymphomas (ABC-DLBCL) have unfavorable outcomes and chronic activation of CARD11-BCL10-MALT1 (CBM) signal amplification complexes that form due to polymerization of BCL10 subunits, which is affected by recurrent somatic mutations in ABC-DLBCLs. Herein, we show that BCL10 mutants fall into at least two functionally distinct classes: missense mutations of the BCL10 CARD domain and truncation of its C-terminal tail. Truncating mutations abrogated a motif through which MALT1 inhibits BCL10 polymerization, trapping MALT1 in its activated filament-bound state. CARD missense mutations enhanced BCL10 filament formation, forming glutamine network structures that stabilize BCL10 filaments. Mutant forms of BCL10 were less dependent on upstream CARD11 activation and thus manifested resistance to BTK inhibitors, whereas BCL10 truncating but not CARD mutants were hypersensitive to MALT1 inhibitors. Therefore, BCL10 mutations are potential biomarkers for BTK inhibitor resistance in ABC-DLBCL, and further precision can be achieved by selecting therapy based on specific biochemical effects of distinct mutation classes. SIGNIFICANCE: ABC-DLBCLs feature frequent mutations of signaling mediators that converge on the CBM complex. We use structure-function approaches to reveal that BCL10 mutations fall into two distinct ...SIGNIFICANCE: ABC-DLBCLs feature frequent mutations of signaling mediators that converge on the CBM complex. We use structure-function approaches to reveal that BCL10 mutations fall into two distinct biochemical classes. Both classes confer resistance to BTK inhibitors, whereas BCL10 truncations confer hyperresponsiveness to MALT1 inhibitors, providing a road map for precision therapies in ABC-DLBCLs. See related commentary by Phelan and Oellerich, p. 1844. This article is highlighted in the In This Issue feature, p. 1825. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_27095.map.gz emd_27095.map.gz | 71.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-27095-v30.xml emd-27095-v30.xml emd-27095.xml emd-27095.xml | 15.1 KB 15.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_27095.png emd_27095.png | 173 KB | ||
| Filedesc metadata |  emd-27095.cif.gz emd-27095.cif.gz | 5.2 KB | ||
| Others |  emd_27095_half_map_1.map.gz emd_27095_half_map_1.map.gz emd_27095_half_map_2.map.gz emd_27095_half_map_2.map.gz | 59.7 MB 59.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-27095 http://ftp.pdbj.org/pub/emdb/structures/EMD-27095 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27095 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27095 | HTTPS FTP |
-Related structure data
| Related structure data |  8czdMC  8czoC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_27095.map.gz / Format: CCP4 / Size: 76.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_27095.map.gz / Format: CCP4 / Size: 76.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | CryoEM structure of BCL10 R58Q | ||||||||||||||||||||
| Voxel size | X=Y=Z: 0.87 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: half map1
| File | emd_27095_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
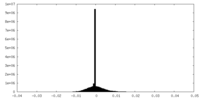
| Density Histograms |
-Half map: half map 2
| File | emd_27095_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map 2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
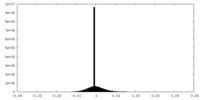
| Density Histograms |
- Sample components
Sample components
-Entire : filament
| Entire | Name: filament |
|---|---|
| Components |
|
-Supramolecule #1: filament
| Supramolecule | Name: filament / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: B-cell lymphoma/leukemia 10
| Macromolecule | Name: B-cell lymphoma/leukemia 10 / type: protein_or_peptide / ID: 1 / Number of copies: 20 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 12.585502 KDa |
| Recombinant expression | Organism:  Escherichia phage EcSzw-2 (virus) Escherichia phage EcSzw-2 (virus) |
| Sequence | String: EEDLTEVKKD ALENLRVYLC EKIIAERHFD HLRAKKILSR EDTEEISCQT SSRKRAGKLL DYLQENPKGL DTLVESIRRE KTQNFLIQK ITDEVLKLRN IKLEHLK UniProtKB: B-cell lymphoma/leukemia 10 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 Component:
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Grid | Model: C-flat-1.2/1.3 / Material: COPPER / Mesh: 300 | |||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Instrument: FEI VITROBOT MARK III |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Number grids imaged: 1 / Number real images: 3439 / Average electron dose: 38.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 2.7 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 36000 |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
- Image processing
Image processing
| Final reconstruction | Applied symmetry - Helical parameters - Δz: 5.0 Å Applied symmetry - Helical parameters - Δ&Phi: -100.8 ° Applied symmetry - Helical parameters - Axial symmetry: C1 (asymmetric) Resolution.type: BY AUTHOR / Resolution: 4.6 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: RELION (ver. 3.1) / Number images used: 30000 |
|---|---|
| Startup model | Type of model: EMDB MAP EMDB ID: |
| Final angle assignment | Type: NOT APPLICABLE |
 Movie
Movie Controller
Controller










 Z
Z Y
Y X
X