+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-3540 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
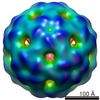
| Title | Asymmetric reconstruction of MS2 virion | |||||||||
 Map data Map data | Asymmetric reconstruction of bacteriophage MS | |||||||||
 Sample Sample | MS2 virion != Enterobacterio phage MS2 MS2 virion
| |||||||||
| Biological species |   Enterobacterio phage MS2 (virus) Enterobacterio phage MS2 (virus) | |||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 7.9 Å cryo EM / Resolution: 7.9 Å | |||||||||
 Authors Authors | Koning RI / Gomez Blanco J / Akopjana I / Vargas J / Kazaks A / Tars K / Carazo JM / Koster AJ | |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2016 Journal: Nat Commun / Year: 2016Title: Asymmetric cryo-EM reconstruction of phage MS2 reveals genome structure in situ. Authors: Roman I Koning / Josue Gomez-Blanco / Inara Akopjana / Javier Vargas / Andris Kazaks / Kaspars Tars / José María Carazo / Abraham J Koster /    Abstract: In single-stranded ribonucleic acid (RNA) viruses, virus capsid assembly and genome packaging are intertwined processes. Using cryo-electron microscopy and single particle analysis we determined the ...In single-stranded ribonucleic acid (RNA) viruses, virus capsid assembly and genome packaging are intertwined processes. Using cryo-electron microscopy and single particle analysis we determined the asymmetric virion structure of bacteriophage MS2, which includes 178 copies of the coat protein, a single copy of the A-protein and the RNA genome. This reveals that in situ, the viral RNA genome can adopt a defined conformation. The RNA forms a branched network of stem-loops that almost all allocate near the capsid inner surface, while predominantly binding to coat protein dimers that are located in one-half of the capsid. This suggests that genomic RNA is highly involved in genome packaging and virion assembly. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_3540.map.gz emd_3540.map.gz | 11.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-3540-v30.xml emd-3540-v30.xml emd-3540.xml emd-3540.xml | 17.6 KB 17.6 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_3540.png emd_3540.png | 149.1 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-3540 http://ftp.pdbj.org/pub/emdb/structures/EMD-3540 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-3540 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-3540 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_3540.map.gz / Format: CCP4 / Size: 12.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_3540.map.gz / Format: CCP4 / Size: 12.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Asymmetric reconstruction of bacteriophage MS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 2.356 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : MS2 virion
| Entire | Name: MS2 virion |
|---|---|
| Components |
|
-Supramolecule #1: Enterobacterio phage MS2
| Supramolecule | Name: Enterobacterio phage MS2 / type: virus / ID: 1 / Parent: 0 / Macromolecule list: #1 / NCBI-ID: 12022 / Sci species name: Enterobacterio phage MS2 / Virus type: VIRION / Virus isolate: SUBSPECIES / Virus enveloped: No / Virus empty: No |
|---|---|
| Host (natural) | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Molecular weight | Theoretical: 3.6 MDa |
| Virus shell | Shell ID: 1 / Name: MS2 / Diameter: 275.0 Å / T number (triangulation number): 3 |
-Macromolecule #1: Enterobacterio phage MS2 RNA
| Macromolecule | Name: Enterobacterio phage MS2 RNA / type: protein_or_peptide / ID: 1 / Enantiomer: LEVO |
|---|---|
| Sequence | String: MRAFSTLDRE NETFVPSVRV YADGETEDNS FSLKYRSNWT PGRFNSTGAK TKQWHYPSPY SRGALSVTSI DQGAYKRSGS SWGRPYEEKA GFGFSLDARS CYSLFPVSQN LTYIEVPQNV ANRASTEVLQ KVTQGNFNLG VALAEARSTA SQLATQTIAL VKAYTAARRG ...String: MRAFSTLDRE NETFVPSVRV YADGETEDNS FSLKYRSNWT PGRFNSTGAK TKQWHYPSPY SRGALSVTSI DQGAYKRSGS SWGRPYEEKA GFGFSLDARS CYSLFPVSQN LTYIEVPQNV ANRASTEVLQ KVTQGNFNLG VALAEARSTA SQLATQTIAL VKAYTAARRG NWRQALRYLA LNEDRKFRSK HVAGRWLELQ FGWLPLMSDI QGAYEMLTKV HLQEFLPMRA VRQVGTNIKL DGRLSYPAAN FQTTCNISRR IVIWFYINDA RLAWLSSLGI LNPLGIVWEK VPFSFVVDWL LPVGNMLEGL TAPVGCSYMS GTVTDVITGE SIISVDAPYG WTVERQGTAK AQISAMHRGV QSVWPTTGAY VKSPFSMVHT LDALALIRQR LSR |
-Macromolecule #2: Enterobacterio phage MS2 RNA
| Macromolecule | Name: Enterobacterio phage MS2 RNA / type: protein_or_peptide / ID: 2 / Enantiomer: LEVO |
|---|---|
| Sequence | String: MASNFTQFVL VDNGGTGDVT VAPSNFANGV AEWISSNSRS QAYKVTCSVR QSSAQNRKYT IKVEVPKVAT QTVGGVQLPV AAWRSYLNME LTIPIFATND DCALIVKAMQ GLLKDGNPIP SAIAANSGIY |
-Macromolecule #3: Enterobacterio phage MS2 RNA
| Macromolecule | Name: Enterobacterio phage MS2 RNA / type: dna / ID: 3 / Classification: DNA |
|---|---|
| Sequence | String: GGGTGGGACC CCTTTCGGGG TCCTGCTCAA CTTCCTGTCG AGCTAATGCC ATTTTTAATG TCTTTAGCGA GACGCTACCA TGGCTATCGC TGTAGGTAGC CGGAATTCCA TTCCTAGGAG GTTTGACCTG TGCGAGCTTT TAGTACCCTT GATAGGGAGA ACGAGACCTT ...String: GGGTGGGACC CCTTTCGGGG TCCTGCTCAA CTTCCTGTCG AGCTAATGCC ATTTTTAATG TCTTTAGCGA GACGCTACCA TGGCTATCGC TGTAGGTAGC CGGAATTCCA TTCCTAGGAG GTTTGACCTG TGCGAGCTTT TAGTACCCTT GATAGGGAGA ACGAGACCTT CGTCCCCTCC GTTCGCGTTT ACGCGGACGG TGAGACTGAA GATAACTCAT TCTCTTTAAA ATATCGTTCG AACTGGACTC CCGGTCGTTT TAACTCGACT GGGGCCAAAA CGAAACAGTG GCACTACCCC TCTCCGTATT CACGGGGGGC GTTAAGTGTC ACATCGATAG ATCAAGGTGC CTACAAGCGA AGTGGGTCAT CGTGGGGTCG CCCGTACGAG GAGAAAGCCG GTTTCGGCTT CTCCCTCGAC GCACGCTCCT GCTACAGCCT CTTCCCTGTA AGCCAAAACT TGACTTACAT CGAAGTGCCG CAGAACGTTG CGAACCGGGC GTCGACCGAA GTCCTGCAAA AGGTCACCCA GGGTAATTTT AACCTTGGTG TTGCTTTAGC AGAGGCCAGG TCGACAGCCT CACAACTCGC GACGCAAACC ATTGCGCTCG TGAAGGCGTA CACTGCCGCT CGTCGCGGTA ATTGGCGCCA GGCGCTCCGC TACCTTGCCC TAAACGAAGA TCGAAAGTTT CGATCAAAAC ACGTGGCCGG CAGGTGGTTG GAGTTGCAGT TCGGTTGGTT ACCACTAATG AGTGATATCC AGGGTGCATA TGAGATGCTT ACGAAGGTTC ACCTTCAAGA GTTTCTTCCT ATGAGAGCCG TACGTCAGGT CGGTACTAAC ATCAAGTTAG ATGGCCGTCT GTCGTATCCA GCTGCAAACT TCCAGACAAC GTGCAACATA TCGCGACGTA TCGTGATATG GTTTTACATA AACGATGCAC GTTTGGCATG GTTGTCGTCT CTAGGTATCT TGAACCCACT AGGTATAGTG TGGGAAAAGG TGCCTTTCTC ATTCGTTGTC GACTGGCTCC TACCTGTAGG TAACATGCTC GAGGGCCTTA CGGCCCCCGT GGGATGCTCC TACATGTCAG GAACAGTTAC TGACGTAATA ACGGGTGAGT CCATCATAAG CGTTGACGCT CCCTACGGGT GGACTGTGGA GAGACAGGGC ACTGCTAAGG CCCAAATCTC AGCCATGCAT CGAGGGGTAC AATCCGTATG GCCAACAACT GGCGCGTACG TAAAGTCTCC TTTCTCGATG GTCCATACCT TAGATGCGTT AGCATTAATC AGGCAACGGC TCTCTAGATA GAGCCCTCAA CCGGAGTTTG AAGCATGGCT TCTAACTTTA CTCAGTTCGT TCTCGTCGAC AATGGCGGAA CTGGCGACGT GACTGTCGCC CCAAGCAACT TCGCTAACGG GGTCGCTGAA TGGATCAGCT CTAACTCGCG TTCACAGGCT TACAAAGTAA CCTGTAGCGT TCGTCAGAGC TCTGCGCAGA ATCGCAAATA CACCATCAAA GTCGAGGTGC CTAAAGTGGC AACCCAGACT GTTGGTGGTG TAGAGCTTCC TGTAGCCGCA TGGCGTTCGT ACTTAAATAT GGAACTAACC ATTCCAATTT TCGCTACGAA TTCCGACTGC GAGCTTATTG TTAAGGCAAT GCAAGGTCTC CTAAAAGATG GAAACCCGAT TCCCTCAGCA ATCGCAGCAA ACTCCGGCAT CTACTAATAG ACGCCGGCCA TTCAAACATG AGGATTACCC ATGTCGAAGA CAACAAAGAA GTTCAACTCT TTATGTATTG ATCTTCCTCG CGATCTTTCT CTCGAAATT TACCAATCAA TTGCTTCTGT CGCTACTGGA AGCGGTGATC CGCACAGTGA CGACTTTACA GCAATTGCTT ACTTAAGGGA CGAATTGCTC ACAAAGCATC CGACCTTAGG TTCTGGTAAT GACGAGGCGA CCCGTCGTAC CTTAGCTATC GCTAAGCTAC GGGAGGCGAA TGGTGATCGC GGTCAGATAA ATAGAGAAGG TTTCTTACAT GACAAATCCT TGTCATGGGA TCCGGATGTT TTACAAACCA GCATCCGTAG CCTTATTGGC AACCTCCTCT CTGGCTACCG ATCGTCGTTG TTTGGGCAAT GCACGTTCTC CAACGGTGCT CCTATGGGGC ACAAGTTGCA GGATGCAGCG CCTTACAAGA AGTTCGCTGA ACAAGCAACC GTTACCCCCC GCGCTCTGAG AGCGGCTCTA TTGGTCCGAG ACCAATGTGC GCCGTGGATC AGACACGCGG TCCGCTATAA CGAGTCATAT GAATTTAGGC TCGTTGTAGG GAACGGAGTG TTTACAGTTC CGAAGAATAA TAAAATAGAT CGGGCTGCCT GTAAGGAGCC TGATATGAAT ATGTACCTCC AGAAAGGGGT CGGTGCTTTC ATCAGACGCC GGCTCAAATC CGTTGGTATA GACCTGAATG ATCAATCGAT CAACCAGCGT CTGGCTCAGC AGGGCAGCGT AGATGGTTCG CTTGCGACGA TAGACTTATC GTCTGCATCC GATTCCATCT CCGATCGCCT GGTGTGGAGT TTTCTCCCAC CAGAGCTATA TTCATATCTC GATCGTATCC GCTCACACTA CGGAATCGTA GATGGCGAGA CGATACGATG GGAACTATTT TCCACAATGG GAAATGGGTT CACATTTGAG CTAGAGTCCA TGATATTCTG GGCAATAGTC AAAGCGACCC AAATCCATTT TGGTAACGCC GGAACCATAG GCATCTACGG GGACGATATT ATATGTCCCA GTGAGATTGC ACCCCGTGTG CTAGAGGCAC TTGCCTACTA CGGTTTTAAA CCGAATCTTC GTAAAACGTT CGTGTCCGGG CTCTTTCGCG AGAGCTGCGG CGCGCACTTT TACCGTGGTG TCGATGTCAA ACCGTTTTAC ATCAAGAAAC CTGTTGACAA TCTCTTCGCC CTGATGCTGA TATTAAATCG GCTACGGGGT TGGGGAGTTG TCGGAGGTAT GTCAGATCCA CGCCTCTATA AGGTGTGGGT ACGGCTCTCC TCCCAGGTGC CTTCGATGTT CTTCGGTGGG ACGGACCTCG CTGCCGACTA CTACGTAGTC AGCCCGCCTA CGGCAGTCTC GGTATACACC A AGACTCCG TACGGGCGGC TGCTCGCGGA TACCCGTACC TCGGGTTTCC GTCTTGCTCG TATCGCTCGA GAACGCAAGT TCTTCAGCGA AAAGCACGAC AGTGGTCGCT ACATAGCGTG GTTCCATACT GGAGGTGAAA TCACCGACAG CATGAAGTCC GCCGGCGTGC GCGTTATACG CACTTCGGAG TGGCTAACGC CGGTTCCCAC ATTCCCTCAG GAGTGTGGGC CAGCGAGCTC TCCTCGGTAG CTGACCGAGG GACCCCCGTA AACGGGGTGG GTGTGCTCGA AAGAGCACGG GTGCGAAAGC GGTCCGGCTC CACCGAAAGG TGGGCGGGCT TCGGCCCAGG GACCTCCCCC TAAAGAGAGG ACCCGGGATT CTCCCGATTT GGTAACTAGC TGCTTGGCTA GTTACCACCC A |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.4 Component:
| |||||||||
| Grid | Model: Quantifoil R2/2 / Material: COPPER / Mesh: 300 / Support film - Material: FORMVAR / Support film - topology: HOLEY ARRAY / Support film - Film thickness: 2.0 nm / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Atmosphere: AIR / Pretreatment - Pressure: 0.02 kPa / Details: 30 mA | |||||||||
| Vitrification | Cryogen name: ETHANE-PROPANE / Chamber humidity: 70 % / Chamber temperature: 298 K / Instrument: LEICA EM GP Details: Blotted using filter paper for 1 to 2 seconds before blotting. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Calibrated magnification: 61403 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Cs: 0.02 mm / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 59000 Bright-field microscopy / Cs: 0.02 mm / Nominal defocus max: 2.5 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 59000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Temperature | Min: 77.0 K / Max: 97.0 K |
| Image recording | Film or detector model: FEI FALCON II (4k x 4k) / Detector mode: OTHER / Digitization - Dimensions - Width: 4096 pixel / Digitization - Dimensions - Height: 4096 pixel / Number grids imaged: 1 / Number real images: 751 / Average exposure time: 1.0 sec. / Average electron dose: 5.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Particle selection | Number selected: 22441 |
|---|---|
| CTF correction | Software - Name: CTFFIND (ver. 3) |
| Startup model | Type of model: OTHER Details: Icosahedral symmetrized map was used as start for asymmetric reconstruction |
| Initial angle assignment | Type: OTHER / Software - Name: RELION (ver. 1.4) |
| Final angle assignment | Type: OTHER |
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Resolution.type: BY AUTHOR / Resolution: 7.9 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: RELION (ver. 1.4) / Number images used: 18977 |
 Movie
Movie Controller
Controller