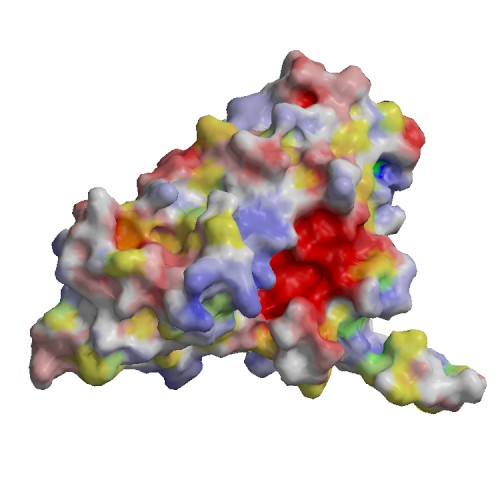
Functional site
| 1) | chain | C |
| residue | 16 | |
| type | ||
| sequence | F | |
| description | binding site for residue 95V C 401 | |
| source | : AC3 | |
| 2) | chain | C |
| residue | 18 | |
| type | ||
| sequence | M | |
| description | binding site for residue 95V C 401 | |
| source | : AC3 | |
| 3) | chain | C |
| residue | 20 | |
| type | ||
| sequence | E | |
| description | binding site for residue 95V C 401 | |
| source | : AC3 | |
| 4) | chain | C |
| residue | 21 | |
| type | ||
| sequence | G | |
| description | binding site for residue 95V C 401 | |
| source | : AC3 | |
| 5) | chain | C |
| residue | 22 | |
| type | ||
| sequence | R | |
| description | binding site for residue 95V C 401 | |
| source | : AC3 | |
| 6) | chain | C |
| residue | 24 | |
| type | ||
| sequence | A | |
| description | binding site for residue 95V C 401 | |
| source | : AC3 | |
| 7) | chain | C |
| residue | 26 | |
| type | ||
| sequence | G | |
| description | binding site for residue 95V C 401 | |
| source | : AC3 | |
| 8) | chain | C |
| residue | 27 | |
| type | ||
| sequence | T | |
| description | binding site for residue 95V C 401 | |
| source | : AC3 | |
| 9) | chain | C |
| residue | 28 | |
| type | ||
| sequence | G | |
| description | binding site for residue 95V C 401 | |
| source | : AC3 | |
| 10) | chain | C |
| residue | 29 | |
| type | ||
| sequence | E | |
| description | binding site for residue 95V C 401 | |
| source | : AC3 | |
| 11) | chain | C |
| residue | 30 | |
| type | ||
| sequence | L | |
| description | binding site for residue 95V C 401 | |
| source | : AC3 | |
| 12) | chain | C |
| residue | 31 | |
| type | ||
| sequence | T | |
| description | binding site for residue 95V C 401 | |
| source | : AC3 | |
| 13) | chain | C |
| residue | 177 | |
| type | ||
| sequence | M | |
| description | binding site for residue 95V C 401 | |
| source | : AC3 | |
| 14) | chain | C |
| residue | 179 | |
| type | ||
| sequence | C | |
| description | binding site for residue 95V C 401 | |
| source | : AC3 | |
| 15) | chain | C |
| residue | 17 | |
| type | BINDING | |
| sequence | V | |
| description | BINDING => ECO:0000269|PubMed:7809062, ECO:0007744|PDB:1FTA | |
| source | Swiss-Prot : SWS_FT_FI1 | |
| 16) | chain | C |
| residue | 27 | |
| type | BINDING | |
| sequence | T | |
| description | BINDING => ECO:0000269|PubMed:7809062, ECO:0007744|PDB:1FTA | |
| source | Swiss-Prot : SWS_FT_FI1 | |
| 17) | chain | C |
| residue | 112 | |
| type | BINDING | |
| sequence | K | |
| description | BINDING => ECO:0000269|PubMed:7809062, ECO:0007744|PDB:1FTA | |
| source | Swiss-Prot : SWS_FT_FI1 | |
| 18) | chain | C |
| residue | 140 | |
| type | BINDING | |
| sequence | R | |
| description | BINDING => ECO:0000269|PubMed:7809062, ECO:0007744|PDB:1FTA | |
| source | Swiss-Prot : SWS_FT_FI1 | |
| 19) | chain | C |
| residue | 97 | |
| type | BINDING | |
| sequence | E | |
| description | BINDING => ECO:0000250|UniProtKB:P00636 | |
| source | Swiss-Prot : SWS_FT_FI2 | |
| 20) | chain | C |
| residue | 120 | |
| type | BINDING | |
| sequence | L | |
| description | BINDING => ECO:0000250|UniProtKB:P00636 | |
| source | Swiss-Prot : SWS_FT_FI2 | |
| 21) | chain | C |
| residue | 212 | |
| type | BINDING | |
| sequence | N | |
| description | BINDING => ECO:0000250|UniProtKB:P00636 | |
| source | Swiss-Prot : SWS_FT_FI2 | |
| 22) | chain | C |
| residue | 243 | |
| type | BINDING | |
| sequence | R | |
| description | BINDING => ECO:0000250|UniProtKB:P00636 | |
| source | Swiss-Prot : SWS_FT_FI2 | |
| 23) | chain | C |
| residue | 264 | |
| type | BINDING | |
| sequence | Y | |
| description | BINDING => ECO:0000250|UniProtKB:P00636 | |
| source | Swiss-Prot : SWS_FT_FI2 | |
| 24) | chain | C |
| residue | 274 | |
| type | BINDING | |
| sequence | K | |
| description | BINDING => ECO:0000250|UniProtKB:P00636 | |
| source | Swiss-Prot : SWS_FT_FI2 | |
| 25) | chain | C |
| residue | 118 | |
| type | BINDING | |
| sequence | D | |
| description | BINDING => ECO:0007744|PDB:2FHY | |
| source | Swiss-Prot : SWS_FT_FI3 | |
| 26) | chain | C |
| residue | 121 | |
| type | BINDING | |
| sequence | D | |
| description | BINDING => ECO:0007744|PDB:2FHY | |
| source | Swiss-Prot : SWS_FT_FI3 | |
| 27) | chain | C |
| residue | 280 | |
| type | BINDING | |
| sequence | E | |
| description | BINDING => ECO:0007744|PDB:2FHY | |
| source | Swiss-Prot : SWS_FT_FI3 | |
| 28) | chain | C |
| residue | 150 | |
| type | MOD_RES | |
| sequence | K | |
| description | N6-succinyllysine => ECO:0000250|UniProtKB:Q9QXD6 | |
| source | Swiss-Prot : SWS_FT_FI5 | |
| 29) | chain | C |
| residue | 215 | |
| type | MOD_RES | |
| sequence | Y | |
| description | Phosphotyrosine => ECO:0000250|UniProtKB:Q9QXD6 | |
| source | Swiss-Prot : SWS_FT_FI6 | |
| 30) | chain | C |
| residue | 244 | |
| type | MOD_RES | |
| sequence | Y | |
| description | Phosphotyrosine => ECO:0000250|UniProtKB:Q9QXD6 | |
| source | Swiss-Prot : SWS_FT_FI6 | |
| 31) | chain | C |
| residue | 264 | |
| type | MOD_RES | |
| sequence | Y | |
| description | Phosphotyrosine => ECO:0007744|PubMed:24275569 | |
| source | Swiss-Prot : SWS_FT_FI7 | |