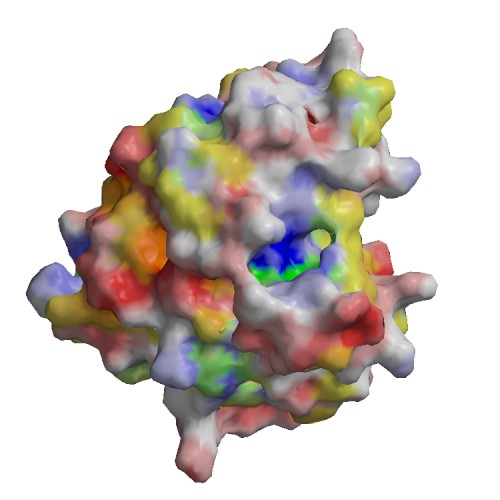
Functional site
| 1) | chain | A |
| residue | 1 | |
| type | ||
| sequence | M | |
| description | binding site for residue SO4 A 301 | |
| source | : AC1 | |
| 2) | chain | A |
| residue | 72 | |
| type | ||
| sequence | A | |
| description | binding site for residue SO4 A 301 | |
| source | : AC1 | |
| 3) | chain | A |
| residue | 99-127 | |
| type | prosite | |
| sequence | DLEAAIRYARHLPYSNGKVGLVGYSLGGA | |
| description | ADH_SHORT Short-chain dehydrogenases/reductases family signature. DleaairyarHlpYSNGKVGLvGYSlGGA | |
| source | prosite : PS00061 | |
| 4) | chain | A |
| residue | 36 | |
| type | catalytic | |
| sequence | E | |
| description | 492 | |
| source | MCSA : MCSA1 | |
| 5) | chain | A |
| residue | 37 | |
| type | catalytic | |
| sequence | I | |
| description | 492 | |
| source | MCSA : MCSA1 | |
| 6) | chain | A |
| residue | 85 | |
| type | catalytic | |
| sequence | Y | |
| description | 492 | |
| source | MCSA : MCSA1 | |
| 7) | chain | A |
| residue | 123 | |
| type | catalytic | |
| sequence | S | |
| description | 492 | |
| source | MCSA : MCSA1 | |
| 8) | chain | A |
| residue | 124 | |
| type | catalytic | |
| sequence | L | |
| description | 492 | |
| source | MCSA : MCSA1 | |
| 9) | chain | A |
| residue | 171 | |
| type | catalytic | |
| sequence | D | |
| description | 492 | |
| source | MCSA : MCSA1 | |
| 10) | chain | A |
| residue | 202 | |
| type | catalytic | |
| sequence | H | |
| description | 492 | |
| source | MCSA : MCSA1 | |
| 11) | chain | A |
| residue | 123 | |
| type | ACT_SITE | |
| sequence | S | |
| description | ACT_SITE => ECO:0000269|PubMed:3221394 | |
| source | Swiss-Prot : SWS_FT_FI1 | |
| 12) | chain | A |
| residue | 171 | |
| type | ACT_SITE | |
| sequence | D | |
| description | ACT_SITE => ECO:0000269|PubMed:3221394 | |
| source | Swiss-Prot : SWS_FT_FI1 | |
| 13) | chain | A |
| residue | 202 | |
| type | ACT_SITE | |
| sequence | H | |
| description | ACT_SITE => ECO:0000269|PubMed:3221394 | |
| source | Swiss-Prot : SWS_FT_FI1 | |