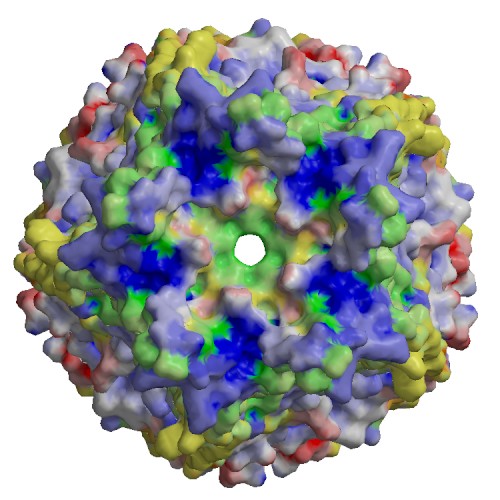
Functional site
| 1) | chain | A |
| residue | 154-168 | |
| type | prosite | |
| sequence | CPMDLKNFPMDVQTC | |
| description | NEUROTR_ION_CHANNEL Neurotransmitter-gated ion-channels signature. CpMdLknFPmDvqtC | |
| source | prosite : PS00236 | |
| 2) | chain | A |
| residue | 239-260 | |
| type | TRANSMEM | |
| sequence | YLIQMYIPSLLIVILSWVSFWI | |
| description | Helical; Name=1 => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI1 | |
| 3) | chain | B |
| residue | 239-260 | |
| type | TRANSMEM | |
| sequence | YLIQMYIPSLLIVILSWVSFWI | |
| description | Helical; Name=1 => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI1 | |
| 4) | chain | C |
| residue | 239-260 | |
| type | TRANSMEM | |
| sequence | YLIQMYIPSLLIVILSWVSFWI | |
| description | Helical; Name=1 => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI1 | |
| 5) | chain | D |
| residue | 239-260 | |
| type | TRANSMEM | |
| sequence | YLIQMYIPSLLIVILSWVSFWI | |
| description | Helical; Name=1 => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI1 | |
| 6) | chain | E |
| residue | 239-260 | |
| type | TRANSMEM | |
| sequence | YLIQMYIPSLLIVILSWVSFWI | |
| description | Helical; Name=1 => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI1 | |
| 7) | chain | A |
| residue | 54 | |
| type | CARBOHYD | |
| sequence | N | |
| description | N-linked (GlcNAc...) asparagine => ECO:0000255, ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI10 | |
| 8) | chain | B |
| residue | 54 | |
| type | CARBOHYD | |
| sequence | N | |
| description | N-linked (GlcNAc...) asparagine => ECO:0000255, ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI10 | |
| 9) | chain | C |
| residue | 54 | |
| type | CARBOHYD | |
| sequence | N | |
| description | N-linked (GlcNAc...) asparagine => ECO:0000255, ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI10 | |
| 10) | chain | D |
| residue | 54 | |
| type | CARBOHYD | |
| sequence | N | |
| description | N-linked (GlcNAc...) asparagine => ECO:0000255, ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI10 | |
| 11) | chain | E |
| residue | 54 | |
| type | CARBOHYD | |
| sequence | N | |
| description | N-linked (GlcNAc...) asparagine => ECO:0000255, ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI10 | |
| 12) | chain | A |
| residue | 261-265 | |
| type | TOPO_DOM | |
| sequence | NMDAA | |
| description | Cytoplasmic => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI2 | |
| 13) | chain | B |
| residue | 261-265 | |
| type | TOPO_DOM | |
| sequence | NMDAA | |
| description | Cytoplasmic => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI2 | |
| 14) | chain | C |
| residue | 261-265 | |
| type | TOPO_DOM | |
| sequence | NMDAA | |
| description | Cytoplasmic => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI2 | |
| 15) | chain | D |
| residue | 261-265 | |
| type | TOPO_DOM | |
| sequence | NMDAA | |
| description | Cytoplasmic => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI2 | |
| 16) | chain | E |
| residue | 261-265 | |
| type | TOPO_DOM | |
| sequence | NMDAA | |
| description | Cytoplasmic => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI2 | |
| 17) | chain | A |
| residue | 266-286 | |
| type | TRANSMEM | |
| sequence | PARVGLGITTVLTMTTQSSGS | |
| description | Helical; Name=2 => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI3 | |
| 18) | chain | B |
| residue | 266-286 | |
| type | TRANSMEM | |
| sequence | PARVGLGITTVLTMTTQSSGS | |
| description | Helical; Name=2 => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI3 | |
| 19) | chain | C |
| residue | 266-286 | |
| type | TRANSMEM | |
| sequence | PARVGLGITTVLTMTTQSSGS | |
| description | Helical; Name=2 => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI3 | |
| 20) | chain | D |
| residue | 266-286 | |
| type | TRANSMEM | |
| sequence | PARVGLGITTVLTMTTQSSGS | |
| description | Helical; Name=2 => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI3 | |
| 21) | chain | E |
| residue | 266-286 | |
| type | TRANSMEM | |
| sequence | PARVGLGITTVLTMTTQSSGS | |
| description | Helical; Name=2 => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI3 | |
| 22) | chain | A |
| residue | 287-297 | |
| type | TOPO_DOM | |
| sequence | RASLPKVSYVK | |
| description | Extracellular => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI4 | |
| 23) | chain | B |
| residue | 287-297 | |
| type | TOPO_DOM | |
| sequence | RASLPKVSYVK | |
| description | Extracellular => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI4 | |
| 24) | chain | C |
| residue | 287-297 | |
| type | TOPO_DOM | |
| sequence | RASLPKVSYVK | |
| description | Extracellular => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI4 | |
| 25) | chain | D |
| residue | 287-297 | |
| type | TOPO_DOM | |
| sequence | RASLPKVSYVK | |
| description | Extracellular => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI4 | |
| 26) | chain | E |
| residue | 287-297 | |
| type | TOPO_DOM | |
| sequence | RASLPKVSYVK | |
| description | Extracellular => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI4 | |
| 27) | chain | A |
| residue | 298-318 | |
| type | TRANSMEM | |
| sequence | AIDIWMAVCLLFVFSALLEYA | |
| description | Helical; Name=3 => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI5 | |
| 28) | chain | B |
| residue | 298-318 | |
| type | TRANSMEM | |
| sequence | AIDIWMAVCLLFVFSALLEYA | |
| description | Helical; Name=3 => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI5 | |
| 29) | chain | C |
| residue | 298-318 | |
| type | TRANSMEM | |
| sequence | AIDIWMAVCLLFVFSALLEYA | |
| description | Helical; Name=3 => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI5 | |
| 30) | chain | D |
| residue | 298-318 | |
| type | TRANSMEM | |
| sequence | AIDIWMAVCLLFVFSALLEYA | |
| description | Helical; Name=3 => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI5 | |
| 31) | chain | E |
| residue | 298-318 | |
| type | TRANSMEM | |
| sequence | AIDIWMAVCLLFVFSALLEYA | |
| description | Helical; Name=3 => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI5 | |
| 32) | chain | A |
| residue | 208 | |
| type | BINDING | |
| sequence | E | |
| description | BINDING => ECO:0000250|UniProtKB:P23415 | |
| source | Swiss-Prot : SWS_FT_FI7 | |
| 33) | chain | D |
| residue | 208 | |
| type | BINDING | |
| sequence | E | |
| description | BINDING => ECO:0000250|UniProtKB:P23415 | |
| source | Swiss-Prot : SWS_FT_FI7 | |
| 34) | chain | D |
| residue | 210 | |
| type | BINDING | |
| sequence | D | |
| description | BINDING => ECO:0000250|UniProtKB:P23415 | |
| source | Swiss-Prot : SWS_FT_FI7 | |
| 35) | chain | D |
| residue | 231 | |
| type | BINDING | |
| sequence | H | |
| description | BINDING => ECO:0000250|UniProtKB:P23415 | |
| source | Swiss-Prot : SWS_FT_FI7 | |
| 36) | chain | E |
| residue | 208 | |
| type | BINDING | |
| sequence | E | |
| description | BINDING => ECO:0000250|UniProtKB:P23415 | |
| source | Swiss-Prot : SWS_FT_FI7 | |
| 37) | chain | E |
| residue | 210 | |
| type | BINDING | |
| sequence | D | |
| description | BINDING => ECO:0000250|UniProtKB:P23415 | |
| source | Swiss-Prot : SWS_FT_FI7 | |
| 38) | chain | E |
| residue | 231 | |
| type | BINDING | |
| sequence | H | |
| description | BINDING => ECO:0000250|UniProtKB:P23415 | |
| source | Swiss-Prot : SWS_FT_FI7 | |
| 39) | chain | A |
| residue | 210 | |
| type | BINDING | |
| sequence | D | |
| description | BINDING => ECO:0000250|UniProtKB:P23415 | |
| source | Swiss-Prot : SWS_FT_FI7 | |
| 40) | chain | A |
| residue | 231 | |
| type | BINDING | |
| sequence | H | |
| description | BINDING => ECO:0000250|UniProtKB:P23415 | |
| source | Swiss-Prot : SWS_FT_FI7 | |
| 41) | chain | B |
| residue | 208 | |
| type | BINDING | |
| sequence | E | |
| description | BINDING => ECO:0000250|UniProtKB:P23415 | |
| source | Swiss-Prot : SWS_FT_FI7 | |
| 42) | chain | B |
| residue | 210 | |
| type | BINDING | |
| sequence | D | |
| description | BINDING => ECO:0000250|UniProtKB:P23415 | |
| source | Swiss-Prot : SWS_FT_FI7 | |
| 43) | chain | B |
| residue | 231 | |
| type | BINDING | |
| sequence | H | |
| description | BINDING => ECO:0000250|UniProtKB:P23415 | |
| source | Swiss-Prot : SWS_FT_FI7 | |
| 44) | chain | C |
| residue | 208 | |
| type | BINDING | |
| sequence | E | |
| description | BINDING => ECO:0000250|UniProtKB:P23415 | |
| source | Swiss-Prot : SWS_FT_FI7 | |
| 45) | chain | C |
| residue | 210 | |
| type | BINDING | |
| sequence | D | |
| description | BINDING => ECO:0000250|UniProtKB:P23415 | |
| source | Swiss-Prot : SWS_FT_FI7 | |
| 46) | chain | C |
| residue | 231 | |
| type | BINDING | |
| sequence | H | |
| description | BINDING => ECO:0000250|UniProtKB:P23415 | |
| source | Swiss-Prot : SWS_FT_FI7 | |
| 47) | chain | A |
| residue | 218 | |
| type | BINDING | |
| sequence | Y | |
| description | BINDING => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI8 | |
| 48) | chain | B |
| residue | 218 | |
| type | BINDING | |
| sequence | Y | |
| description | BINDING => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI8 | |
| 49) | chain | C |
| residue | 218 | |
| type | BINDING | |
| sequence | Y | |
| description | BINDING => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI8 | |
| 50) | chain | D |
| residue | 218 | |
| type | BINDING | |
| sequence | Y | |
| description | BINDING => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI8 | |
| 51) | chain | E |
| residue | 218 | |
| type | BINDING | |
| sequence | Y | |
| description | BINDING => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI8 | |
| 52) | chain | A |
| residue | 277 | |
| type | SITE | |
| sequence | L | |
| description | Important for obstruction of the ion pore in the closed conformation => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI9 | |
| 53) | chain | B |
| residue | 277 | |
| type | SITE | |
| sequence | L | |
| description | Important for obstruction of the ion pore in the closed conformation => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI9 | |
| 54) | chain | C |
| residue | 277 | |
| type | SITE | |
| sequence | L | |
| description | Important for obstruction of the ion pore in the closed conformation => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI9 | |
| 55) | chain | D |
| residue | 277 | |
| type | SITE | |
| sequence | L | |
| description | Important for obstruction of the ion pore in the closed conformation => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI9 | |
| 56) | chain | E |
| residue | 277 | |
| type | SITE | |
| sequence | L | |
| description | Important for obstruction of the ion pore in the closed conformation => ECO:0000269|PubMed:26344198 | |
| source | Swiss-Prot : SWS_FT_FI9 | |