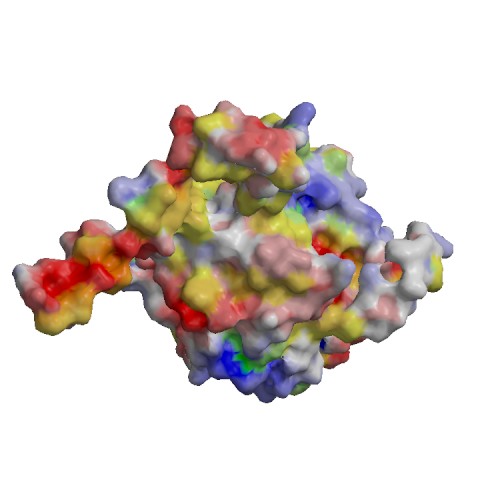
Functional site
| 1) | chain | B |
| residue | 46 | |
| type | catalytic | |
| sequence | S | |
| description | 271 | |
| source | MCSA : MCSA2 | |
| 2) | chain | B |
| residue | 123 | |
| type | catalytic | |
| sequence | S | |
| description | 271 | |
| source | MCSA : MCSA2 | |
| 3) | chain | B |
| residue | 159 | |
| type | catalytic | |
| sequence | R | |
| description | 271 | |
| source | MCSA : MCSA2 | |
| 4) | chain | B |
| residue | 190 | |
| type | catalytic | |
| sequence | H | |
| description | 271 | |
| source | MCSA : MCSA2 | |
| 5) | chain | B |
| residue | 47 | |
| type | catalytic | |
| sequence | G | |
| description | 271 | |
| source | MCSA : MCSA2 | |
| 6) | chain | B |
| residue | 48 | |
| type | catalytic | |
| sequence | L | |
| description | 271 | |
| source | MCSA : MCSA2 | |
| 7) | chain | B |
| residue | 58 | |
| type | catalytic | |
| sequence | D | |
| description | 271 | |
| source | MCSA : MCSA2 | |
| 8) | chain | B |
| residue | 85 | |
| type | catalytic | |
| sequence | D | |
| description | 271 | |
| source | MCSA : MCSA2 | |
| 9) | chain | B |
| residue | 87 | |
| type | catalytic | |
| sequence | D | |
| description | 271 | |
| source | MCSA : MCSA2 | |
| 10) | chain | B |
| residue | 114 | |
| type | catalytic | |
| sequence | E | |
| description | 271 | |
| source | MCSA : MCSA2 | |
| 11) | chain | B |
| residue | 120 | |
| type | catalytic | |
| sequence | K | |
| description | 271 | |
| source | MCSA : MCSA2 | |
| 12) | chain | B |
| residue | 122 | |
| type | catalytic | |
| sequence | N | |
| description | 271 | |
| source | MCSA : MCSA2 | |
| 13) | chain | B |
| residue | 58 | |
| type | ACT_SITE | |
| sequence | D | |
| description | Nucleophile => ECO:0000255 | |
| source | Swiss-Prot : SWS_FT_FI1 | |
| 14) | chain | B |
| residue | 58 | |
| type | BINDING | |
| sequence | D | |
| description | ||
| source | Swiss-Prot : SWS_FT_FI2 | |