+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3j7z | ||||||
|---|---|---|---|---|---|---|---|
| Title | Structure of the E. coli 50S subunit with ErmCL nascent chain | ||||||
 Components Components |
| ||||||
 Keywords Keywords | RIBOSOME/ANTIBIOTIC / erythromycin / stalling / RIBOSOME-ANTIBIOTIC complex | ||||||
| Function / homology |  Function and homology information Function and homology informationstringent response / transcriptional attenuation / endoribonuclease inhibitor activity / RNA-binding transcription regulator activity / positive regulation of ribosome biogenesis / negative regulation of cytoplasmic translation / translational termination / DnaA-L2 complex / translation repressor activity / negative regulation of DNA-templated DNA replication initiation ...stringent response / transcriptional attenuation / endoribonuclease inhibitor activity / RNA-binding transcription regulator activity / positive regulation of ribosome biogenesis / negative regulation of cytoplasmic translation / translational termination / DnaA-L2 complex / translation repressor activity / negative regulation of DNA-templated DNA replication initiation / mRNA regulatory element binding translation repressor activity / ribosome assembly / assembly of large subunit precursor of preribosome / cytosolic ribosome assembly / ribosomal large subunit assembly / response to reactive oxygen species / regulation of cell growth / DNA-templated transcription termination / response to radiation / mRNA 5'-UTR binding / large ribosomal subunit / ribosome binding / transferase activity / 5S rRNA binding / large ribosomal subunit rRNA binding / cytosolic large ribosomal subunit / cytoplasmic translation / tRNA binding / rRNA binding / negative regulation of translation / ribosome / structural constituent of ribosome / translation / response to antibiotic / negative regulation of DNA-templated transcription / mRNA binding / protein homodimerization activity / DNA binding / RNA binding / zinc ion binding / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |   | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.9 Å | ||||||
 Authors Authors | Arenz, S. / Meydan, S. / Starosta, A.L. / Berninghausen, O. / Beckmann, R. / Vazquez-Laslop, N. / Wilson, D.N. | ||||||
 Citation Citation |  Journal: Mol Cell / Year: 2014 Journal: Mol Cell / Year: 2014Title: Drug sensing by the ribosome induces translational arrest via active site perturbation. Authors: Stefan Arenz / Sezen Meydan / Agata L Starosta / Otto Berninghausen / Roland Beckmann / Nora Vázquez-Laslop / Daniel N Wilson /   Abstract: During protein synthesis, nascent polypeptide chains within the ribosomal tunnel can act in cis to induce ribosome stalling and regulate expression of downstream genes. The Staphylococcus aureus ...During protein synthesis, nascent polypeptide chains within the ribosomal tunnel can act in cis to induce ribosome stalling and regulate expression of downstream genes. The Staphylococcus aureus ErmCL leader peptide induces stalling in the presence of clinically important macrolide antibiotics, such as erythromycin, leading to the induction of the downstream macrolide resistance methyltransferase ErmC. Here, we present a cryo-electron microscopy (EM) structure of the erythromycin-dependent ErmCL-stalled ribosome at 3.9 Å resolution. The structure reveals how the ErmCL nascent chain directly senses the presence of the tunnel-bound drug and thereby induces allosteric conformational rearrangements at the peptidyltransferase center (PTC) of the ribosome. ErmCL-induced perturbations of the PTC prevent stable binding and accommodation of the aminoacyl-tRNA at the A-site, leading to inhibition of peptide bond formation and translation arrest. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3j7z.cif.gz 3j7z.cif.gz | 2.2 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3j7z.ent.gz pdb3j7z.ent.gz | 1.7 MB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3j7z.json.gz 3j7z.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  3j7z_validation.pdf.gz 3j7z_validation.pdf.gz | 1.2 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  3j7z_full_validation.pdf.gz 3j7z_full_validation.pdf.gz | 1.4 MB | Display | |
| Data in XML |  3j7z_validation.xml.gz 3j7z_validation.xml.gz | 158.6 KB | Display | |
| Data in CIF |  3j7z_validation.cif.gz 3j7z_validation.cif.gz | 261 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/j7/3j7z https://data.pdbj.org/pub/pdb/validation_reports/j7/3j7z ftp://data.pdbj.org/pub/pdb/validation_reports/j7/3j7z ftp://data.pdbj.org/pub/pdb/validation_reports/j7/3j7z | HTTPS FTP |
-Related structure data
| Related structure data |  6057MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
+50S ribosomal protein ... , 31 types, 31 molecules 0123456CDEFGHIJKLMNOPQRSTUVWXYZ
-RNA chain , 3 types, 3 molecules 7AB
| #8: RNA chain | Mass: 894.612 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
|---|---|
| #9: RNA chain | Mass: 941306.188 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #10: RNA chain | Mass: 38177.762 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
-Protein/peptide / Non-polymers , 2 types, 2 molecules a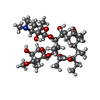

| #35: Protein/peptide | Mass: 2212.631 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
|---|---|
| #36: Chemical | ChemComp-ERY / |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component |
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer solution | pH: 7.4 | ||||||||||||
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | ||||||||||||
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Details: Plunged into liquid ethane (FEI VITROBOT MARK IV). |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS / Date: Mar 12, 2014 |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN |
| Electron lens | Mode: BRIGHT FIELD / Nominal magnification: 125085 X / Cs: 0 mm / Camera length: 0 mm |
| Specimen holder | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Film or detector model: FEI FALCON II (4k x 4k) |
| Image scans | Num. digital images: 6118 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Relative weight: 1 |
- Processing
Processing
| EM software |
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Details: defocus groups | ||||||||||||
| Symmetry | Point symmetry: C1 (asymmetric) | ||||||||||||
| 3D reconstruction | Resolution: 3.9 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 269163 Details: Since the microscopy images were processed in the absence of spatial frequencies higher than 8 A, a FSC cut-off value of 0.143 was used for average resolution determination of 3.9 A (Scheres ...Details: Since the microscopy images were processed in the absence of spatial frequencies higher than 8 A, a FSC cut-off value of 0.143 was used for average resolution determination of 3.9 A (Scheres and Chen, 2012). (Single particle--Applied symmetry: C1) Symmetry type: POINT | ||||||||||||
| Atomic model building | Protocol: RIGID BODY FIT / Space: REAL Details: REFINEMENT PROTOCOL--rigid body DETAILS--The crystal structure of the E. coli 50S subunit was fitted as a rigid body. | ||||||||||||
| Atomic model building | PDB-ID: 4KIX 4kix | ||||||||||||
| Refinement step | Cycle: LAST
|
 Movie
Movie Controller
Controller













 PDBj
PDBj































