+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-2166 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Architecture of human translation initiation factor 3 | |||||||||
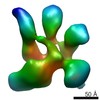
 Map data Map data | cryo-EM reconstruction of the eIF3 PCI-MPN core complex | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | translation initiation / eukaryotic initiation factor 3 / ribosome / proteasome / PCI-MPN domains | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / negative staining / Resolution: 15.0 Å | |||||||||
 Authors Authors | Querol-Audi J / Sun C / Vogan J / Cate J / Nogales E | |||||||||
 Citation Citation |  Journal: Structure / Year: 2013 Journal: Structure / Year: 2013Title: Architecture of human translation initiation factor 3. Authors: Jordi Querol-Audi / Chaomin Sun / Jacob M Vogan / M Duane Smith / Yu Gu / Jamie H D Cate / Eva Nogales /  Abstract: Eukaryotic translation initiation factor 3 (eIF3) plays a central role in protein synthesis by organizing the formation of the 43S preinitiation complex. Using genetic tag visualization by electron ...Eukaryotic translation initiation factor 3 (eIF3) plays a central role in protein synthesis by organizing the formation of the 43S preinitiation complex. Using genetic tag visualization by electron microscopy, we reveal the molecular organization of ten human eIF3 subunits, including an octameric core. The structure of eIF3 bears a close resemblance to that of the proteasome lid, with a conserved spatial organization of eight core subunits containing PCI and MPN domains that coordinate functional interactions in both complexes. We further show that eIF3 subunits a and c interact with initiation factors eIF1 and eIF1A, which control the stringency of start codon selection. Finally, we find that subunit j, which modulates messenger RNA interactions with the small ribosomal subunit, makes multiple independent interactions with the eIF3 octameric core. These results highlight the conserved architecture of eIF3 and how it scaffolds key factors that control translation initiation in higher eukaryotes, including humans. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_2166.map.gz emd_2166.map.gz | 9.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-2166-v30.xml emd-2166-v30.xml emd-2166.xml emd-2166.xml | 13.1 KB 13.1 KB | Display Display |  EMDB header EMDB header |
| Images |  eIF3_core.tif eIF3_core.tif | 763.3 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-2166 http://ftp.pdbj.org/pub/emdb/structures/EMD-2166 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2166 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2166 | HTTPS FTP |
-Validation report
| Summary document |  emd_2166_validation.pdf.gz emd_2166_validation.pdf.gz | 199.1 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_2166_full_validation.pdf.gz emd_2166_full_validation.pdf.gz | 198.3 KB | Display | |
| Data in XML |  emd_2166_validation.xml.gz emd_2166_validation.xml.gz | 5.9 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2166 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2166 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2166 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-2166 | HTTPS FTP |
-Related structure data
| Similar structure data |
|---|
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_2166.map.gz / Format: CCP4 / Size: 11.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_2166.map.gz / Format: CCP4 / Size: 11.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | cryo-EM reconstruction of the eIF3 PCI-MPN core complex | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 2.31 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : PCI MPN core of the human eukaryotic translation initiation facto...
| Entire | Name: PCI MPN core of the human eukaryotic translation initiation factor eIF3 |
|---|---|
| Components |
|
-Supramolecule #1000: PCI MPN core of the human eukaryotic translation initiation facto...
| Supramolecule | Name: PCI MPN core of the human eukaryotic translation initiation factor eIF3 type: sample / ID: 1000 / Details: The sample was monodisperse / Oligomeric state: Subcomplex containing 8 core subunits / Number unique components: 8 |
|---|---|
| Molecular weight | Theoretical: 400 KDa |
-Macromolecule #1: eIF3 a
| Macromolecule | Name: eIF3 a / type: protein_or_peptide / ID: 1 / Details: truncated version, contains residues 5-654 / Oligomeric state: Monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human / Cell: n/atruncated version, contains residues 302-913 Homo sapiens (human) / synonym: Human / Cell: n/atruncated version, contains residues 302-913 |
| Recombinant expression | Organism:  |
-Macromolecule #2: eIF3 c
| Macromolecule | Name: eIF3 c / type: protein_or_peptide / ID: 2 / Details: truncated version, contains residues 302-913 / Oligomeric state: Monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human Homo sapiens (human) / synonym: Human |
| Recombinant expression | Organism:  |
-Macromolecule #3: eIF3 e
| Macromolecule | Name: eIF3 e / type: protein_or_peptide / ID: 3 / Oligomeric state: Monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human Homo sapiens (human) / synonym: Human |
| Recombinant expression | Organism:  |
-Macromolecule #4: eIF3 f
| Macromolecule | Name: eIF3 f / type: protein_or_peptide / ID: 4 / Oligomeric state: Monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human Homo sapiens (human) / synonym: Human |
| Recombinant expression | Organism:  |
-Macromolecule #5: eIF3 h
| Macromolecule | Name: eIF3 h / type: protein_or_peptide / ID: 5 / Oligomeric state: Monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human Homo sapiens (human) / synonym: Human |
| Recombinant expression | Organism:  |
-Macromolecule #6: eIF3 k
| Macromolecule | Name: eIF3 k / type: protein_or_peptide / ID: 6 / Oligomeric state: Monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human Homo sapiens (human) / synonym: Human |
| Recombinant expression | Organism:  |
-Macromolecule #7: eIF3 l
| Macromolecule | Name: eIF3 l / type: protein_or_peptide / ID: 7 / Oligomeric state: Monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human Homo sapiens (human) / synonym: Human |
| Recombinant expression | Organism:  |
-Macromolecule #8: eIF3 m
| Macromolecule | Name: eIF3 m / type: protein_or_peptide / ID: 8 / Oligomeric state: Monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human Homo sapiens (human) / synonym: Human |
| Recombinant expression | Organism:  |
-Experimental details
-Structure determination
| Method | negative staining, cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.2 mg/mL |
|---|---|
| Buffer | pH: 7.4 Details: 20mM Hepes, 75mM KCl, 0.5mM EDTA, 1mM DTT, 2mM MgCl2, 3% trehalose |
| Staining | Type: NEGATIVE / Details: vitrified |
| Grid | Details: C-flat plasma cleaned with Solarus |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Instrument: FEI VITROBOT MARK II |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI F20 |
|---|---|
| Date | Oct 9, 2011 |
| Image recording | Number real images: 2100 / Average electron dose: 20 e/Å2 |
| Electron beam | Acceleration voltage: 120 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: SPOT SCAN / Imaging mode: BRIGHT FIELD / Nominal defocus max: 3.5 µm / Nominal defocus min: 1.4 µm / Nominal magnification: 100000 |
| Sample stage | Specimen holder model: GATAN LIQUID NITROGEN |
| Experimental equipment |  Model: Tecnai F20 / Image courtesy: FEI Company |
- Image processing
Image processing
| Details | automatic particle selection with template correlator |
|---|---|
| CTF correction | Details: each image |
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 15.0 Å / Resolution method: FSC 0.5 CUT-OFF / Software - Name: EMAN2 / Number images used: 172533 |
| Final two d classification | Number classes: 4000 |
 Movie
Movie Controller
Controller