[English] 日本語
 Yorodumi
Yorodumi- PDB-8thm: Beta carbonic anhydrase from the carboxysome of Cyanobium PCC 7001 -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8thm | ||||||
|---|---|---|---|---|---|---|---|
| Title | Beta carbonic anhydrase from the carboxysome of Cyanobium PCC 7001 | ||||||
 Components Components | Carboxysome shell carbonic anhydrase | ||||||
 Keywords Keywords |  PHOTOSYNTHESIS / beta-carbonic anhydrase / PHOTOSYNTHESIS / beta-carbonic anhydrase /  carboxysome / carboxysome /  cyanobacteria / RuBP activated / cyanobacteria / RuBP activated /  LYASE LYASE | ||||||
| Function / homology |  Function and homology information Function and homology information carboxysome / carboxysome /  carbon fixation / carbon fixation /  carbonic anhydrase / carbonic anhydrase /  carbonate dehydratase activity / carbonate dehydratase activity /  metal ion binding metal ion bindingSimilarity search - Function | ||||||
| Biological species |  Cyanobium sp. PCC 7001 (bacteria) Cyanobium sp. PCC 7001 (bacteria) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.3 Å MOLECULAR REPLACEMENT / Resolution: 2.3 Å | ||||||
 Authors Authors | Pulsford, S.B. / Jackson, C.J. | ||||||
| Funding support |  Australia, 1items Australia, 1items
| ||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: Alpha-cyanobacterial carbonic anhydrase is allosterically regulated by the Rubisco substrate Ribulose 1,5-bisphosphate (RuBP) Authors: Pulsford, S.B. / Outram, M.A. / Forster, B. / Jackson, C.J. / Rhodes, T. / Williams, S.J. / Price, G.D. / Badger, M.R. / Long, B.M. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8thm.cif.gz 8thm.cif.gz | 562.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8thm.ent.gz pdb8thm.ent.gz | 464.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8thm.json.gz 8thm.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/th/8thm https://data.pdbj.org/pub/pdb/validation_reports/th/8thm ftp://data.pdbj.org/pub/pdb/validation_reports/th/8thm ftp://data.pdbj.org/pub/pdb/validation_reports/th/8thm | HTTPS FTP |
|---|
-Related structure data
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein / Sugars , 2 types, 12 molecules ABCDEF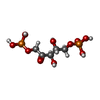

| #1: Protein | Mass: 51308.566 Da / Num. of mol.: 6 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Cyanobium sp. PCC 7001 (bacteria) / Gene: CPCC7001_23 / Production host: Cyanobium sp. PCC 7001 (bacteria) / Gene: CPCC7001_23 / Production host:   Escherichia coli BL21(DE3) (bacteria) / References: UniProt: B5ILN4, Escherichia coli BL21(DE3) (bacteria) / References: UniProt: B5ILN4,  carbonic anhydrase carbonic anhydrase#6: Sugar | ChemComp-RUB /  Ribulose 1,5-bisphosphate Ribulose 1,5-bisphosphate |
|---|
-Non-polymers , 7 types, 815 molecules 











| #2: Chemical | ChemComp-ZN / #3: Chemical |  Glycerol Glycerol#4: Chemical | ChemComp-SO4 /  Sulfate Sulfate#5: Chemical | ChemComp-EDO /  Ethylene glycol Ethylene glycol#7: Chemical |  Carbon dioxide Carbon dioxide#8: Chemical | ChemComp-BCT / |  Bicarbonate Bicarbonate#9: Water | ChemComp-HOH / |  Water Water |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.93 Å3/Da / Density % sol: 58.03 % |
|---|---|
Crystal grow | Temperature: 293.15 K / Method: vapor diffusion, hanging drop / pH: 6.5 Details: 0.2M Ammonium sulfate, 0.1M BIS-TRIS (pH 6.5), 21% PEG 3350, 15% Ethylene glycol |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  Australian Synchrotron Australian Synchrotron  / Beamline: MX2 / Wavelength: 0.9537 Å / Beamline: MX2 / Wavelength: 0.9537 Å |
| Detector | Type: DECTRIS EIGER X 16M / Detector: PIXEL / Date: Oct 29, 2021 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 0.9537 Å / Relative weight: 1 : 0.9537 Å / Relative weight: 1 |
| Reflection | Resolution: 2.3→48.48 Å / Num. obs: 160617 / % possible obs: 100 % / Redundancy: 12.3 % / CC1/2: 0.997 / Rmerge(I) obs: 0.26 / Rpim(I) all: 0.077 / Rrim(I) all: 0.271 / Χ2: 1.02 / Net I/σ(I): 8.1 / Num. measured all: 1970240 |
| Reflection shell | Resolution: 2.3→2.34 Å / % possible obs: 100 % / Redundancy: 12.7 % / Rmerge(I) obs: 5.093 / Num. measured all: 100213 / Num. unique obs: 7868 / CC1/2: 0.297 / Rpim(I) all: 1.468 / Rrim(I) all: 5.302 / Χ2: 1.04 / Net I/σ(I) obs: 0.7 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT / Resolution: 2.3→47.51 Å / SU ML: 0.38 / Cross valid method: FREE R-VALUE / σ(F): 1.33 / Phase error: 29.48 / Stereochemistry target values: ML MOLECULAR REPLACEMENT / Resolution: 2.3→47.51 Å / SU ML: 0.38 / Cross valid method: FREE R-VALUE / σ(F): 1.33 / Phase error: 29.48 / Stereochemistry target values: ML
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.3→47.51 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller


 PDBj
PDBj











