+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8ihk | ||||||
|---|---|---|---|---|---|---|---|
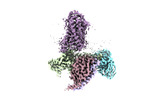
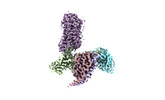
| Title | Cryo-EM structure of HCA3-Gi complex with acifran (local) | ||||||
 Components Components | Soluble cytochrome b562,Hydroxycarboxylic acid receptor 3 | ||||||
 Keywords Keywords |  SIGNALING PROTEIN / SIGNALING PROTEIN /  GPCR GPCR | ||||||
| Function / homology |  Function and homology information Function and homology information nicotinic acid receptor activity / Hydroxycarboxylic acid-binding receptors / G protein-coupled receptor activity / nicotinic acid receptor activity / Hydroxycarboxylic acid-binding receptors / G protein-coupled receptor activity /  cell junction / G alpha (i) signalling events / cell junction / G alpha (i) signalling events /  electron transfer activity / electron transfer activity /  periplasmic space / iron ion binding / G protein-coupled receptor signaling pathway / periplasmic space / iron ion binding / G protein-coupled receptor signaling pathway /  heme binding / heme binding /  plasma membrane plasma membraneSimilarity search - Function | ||||||
| Biological species |   Escherichia coli (E. coli) Escherichia coli (E. coli)  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.21 Å cryo EM / Resolution: 3.21 Å | ||||||
 Authors Authors | Suzuki, S. / Nishikawa, K. / Suzuki, H. / Fujiyoshi, Y. | ||||||
| Funding support |  Japan, 1items Japan, 1items
| ||||||
 Citation Citation |  Journal: Nat Commun / Year: 2023 Journal: Nat Commun / Year: 2023Title: Structural basis of hydroxycarboxylic acid receptor signaling mechanisms through ligand binding. Authors: Shota Suzuki / Kotaro Tanaka / Kouki Nishikawa / Hiroshi Suzuki / Atsunori Oshima / Yoshinori Fujiyoshi /  Abstract: Hydroxycarboxylic acid receptors (HCA) are expressed in various tissues and immune cells. HCA2 and its agonist are thus important targets for treating inflammatory and metabolic disorders. Only ...Hydroxycarboxylic acid receptors (HCA) are expressed in various tissues and immune cells. HCA2 and its agonist are thus important targets for treating inflammatory and metabolic disorders. Only limited information is available, however, on the active-state binding of HCAs with agonists. Here, we present cryo-EM structures of human HCA2-Gi and HCA3-Gi signaling complexes binding with multiple compounds bound. Agonists were revealed to form a salt bridge with arginine, which is conserved in the HCA family, to activate these receptors. Extracellular regions of the receptors form a lid-like structure that covers the ligand-binding pocket. Although transmembrane (TM) 6 in HCAs undergoes dynamic conformational changes, ligands do not directly interact with amino acids in TM6, suggesting that indirect signaling induces a slight shift in TM6 to activate Gi proteins. Structural analyses of agonist-bound HCA2 and HCA3 together with mutagenesis and molecular dynamics simulation provide molecular insights into HCA ligand recognition and activation mechanisms. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8ihk.cif.gz 8ihk.cif.gz | 77.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8ihk.ent.gz pdb8ihk.ent.gz | 51.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8ihk.json.gz 8ihk.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ih/8ihk https://data.pdbj.org/pub/pdb/validation_reports/ih/8ihk ftp://data.pdbj.org/pub/pdb/validation_reports/ih/8ihk ftp://data.pdbj.org/pub/pdb/validation_reports/ih/8ihk | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  35447MC  8ihbC  8ihfC  8ihhC  8ihiC  8ihjC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 60749.406 Da / Num. of mol.: 1 / Mutation: M29W,H124I Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Escherichia coli (E. coli), (gene. exp.) Escherichia coli (E. coli), (gene. exp.)   Homo sapiens (human) Homo sapiens (human)Gene: cybC, HCAR3, GPR109B, HCA3, HM74B, NIACR2 / Production host:   Spodoptera frugiperda (fall armyworm) / References: UniProt: P0ABE7, UniProt: P49019 Spodoptera frugiperda (fall armyworm) / References: UniProt: P0ABE7, UniProt: P49019 |
|---|---|
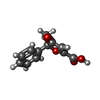
| #2: Chemical | ChemComp-P9X / ( |
| Has ligand of interest | Y |
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Multiprotein complex Protein complex / Type: COMPLEX / Entity ID: #1 / Source: RECOMBINANT Protein complex / Type: COMPLEX / Entity ID: #1 / Source: RECOMBINANT | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Experimental value: NO | ||||||||||||
| Source (natural) |
| ||||||||||||
| Source (recombinant) | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) | ||||||||||||
| Buffer solution | pH: 7.4 | ||||||||||||
| Specimen | Conc.: 15 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES / Details: This sample was monodisperse : YES / Details: This sample was monodisperse | ||||||||||||
Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 298 K |
- Electron microscopy imaging
Electron microscopy imaging
| Microscopy | Model: JEOL CRYO ARM 300 |
|---|---|
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 2000 nm / Nominal defocus min: 1000 nm Bright-field microscopy / Nominal defocus max: 2000 nm / Nominal defocus min: 1000 nm |
| Image recording | Electron dose: 49 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) |
- Processing
Processing
CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
3D reconstruction | Resolution: 3.21 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 95567 / Symmetry type: POINT | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller






 PDBj
PDBj