[English] 日本語
 Yorodumi
Yorodumi- PDB-8h68: Crystal structure of Caenorhabditis elegans NMAD-1 in complex wit... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8h68 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Crystal structure of Caenorhabditis elegans NMAD-1 in complex with NOG and Mg(II) | |||||||||
 Components Components | Maltose/maltodextrin-binding periplasmic protein,DNA N6-methyl adenine demethylase | |||||||||
 Keywords Keywords | METAL BINDING PROTEIN / 2-OG/Fe dependent | |||||||||
| Function / homology |  Function and homology information Function and homology informationDNA N6-methyladenine demethylase activity / DNA N6-methyladenine demethylase / positive regulation of egg-laying behavior / positive regulation of fertilization / meiotic chromosome condensation /  contractile ring / positive regulation of chromosome organization / contractile ring / positive regulation of chromosome organization /  demethylation / positive regulation of meiosis I / broad specificity oxidative DNA demethylase activity ...DNA N6-methyladenine demethylase activity / DNA N6-methyladenine demethylase / positive regulation of egg-laying behavior / positive regulation of fertilization / meiotic chromosome condensation / demethylation / positive regulation of meiosis I / broad specificity oxidative DNA demethylase activity ...DNA N6-methyladenine demethylase activity / DNA N6-methyladenine demethylase / positive regulation of egg-laying behavior / positive regulation of fertilization / meiotic chromosome condensation /  contractile ring / positive regulation of chromosome organization / contractile ring / positive regulation of chromosome organization /  demethylation / positive regulation of meiosis I / broad specificity oxidative DNA demethylase activity / demethylation / positive regulation of meiosis I / broad specificity oxidative DNA demethylase activity /  demethylase activity / actomyosin structure organization / positive regulation of double-strand break repair / demethylase activity / actomyosin structure organization / positive regulation of double-strand break repair /  maltose binding / maltose transport / maltodextrin transmembrane transport / carbohydrate transmembrane transporter activity / ATP-binding cassette (ABC) transporter complex, substrate-binding subunit-containing / maltose binding / maltose transport / maltodextrin transmembrane transport / carbohydrate transmembrane transporter activity / ATP-binding cassette (ABC) transporter complex, substrate-binding subunit-containing /  actin binding / outer membrane-bounded periplasmic space / midbody / actin binding / outer membrane-bounded periplasmic space / midbody /  DNA replication / DNA replication /  oxidoreductase activity / oxidoreductase activity /  metal ion binding / metal ion binding /  nucleus nucleusSimilarity search - Function | |||||||||
| Biological species |   Escherichia coli (E. coli) Escherichia coli (E. coli)  Caenorhabditis elegans (invertebrata) Caenorhabditis elegans (invertebrata) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.2 Å MOLECULAR REPLACEMENT / Resolution: 2.2 Å | |||||||||
 Authors Authors | Shi, Y. / Ding, J. / Yang, H. | |||||||||
| Funding support | 2items
| |||||||||
 Citation Citation |  Journal: J Mol Cell Biol / Year: 2023 Journal: J Mol Cell Biol / Year: 2023Title: Caenorhabditis elegans NMAD-1 functions as a demethylase for actin. Authors: Shi, Y. / Yang, H. / Ding, J. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8h68.cif.gz 8h68.cif.gz | 275.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8h68.ent.gz pdb8h68.ent.gz | 216.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8h68.json.gz 8h68.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/h6/8h68 https://data.pdbj.org/pub/pdb/validation_reports/h6/8h68 ftp://data.pdbj.org/pub/pdb/validation_reports/h6/8h68 ftp://data.pdbj.org/pub/pdb/validation_reports/h6/8h68 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  3rzlS S: Starting model for refinement |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein / Sugars , 2 types, 2 molecules A
| #1: Protein | Mass: 70953.328 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Escherichia coli (E. coli), (gene. exp.) Escherichia coli (E. coli), (gene. exp.)   Caenorhabditis elegans (invertebrata) Caenorhabditis elegans (invertebrata)Gene: malE, Z5632, ECs5017, nmad-1, F09F7.7 / Production host:   Escherichia coli (E. coli) Escherichia coli (E. coli)References: UniProt: P0AEY0, UniProt: Q8MNT9, DNA N6-methyladenine demethylase |
|---|---|
| #2: Polysaccharide | alpha-D-glucopyranose-(1-4)-alpha-D-glucopyranose-(1-4)-alpha-D-glucopyranose-(1-4)-alpha-D-glucopyranose |
-Non-polymers , 7 types, 203 molecules 











| #3: Chemical | ChemComp-TRS /  Tris Tris |
|---|---|
| #4: Chemical | ChemComp-GLY /  Glycine Glycine |
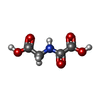
| #5: Chemical | ChemComp-OGA /  N-Oxalylglycine N-Oxalylglycine |
| #6: Chemical | ChemComp-MG / |
| #7: Chemical | ChemComp-EPE /  HEPES HEPES |
| #8: Chemical | ChemComp-PEG /  Diethylene glycol Diethylene glycol |
| #9: Water | ChemComp-HOH /  Water Water |
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.17 Å3/Da / Density % sol: 43.27 % |
|---|---|
Crystal grow | Temperature: 289 K / Method: vapor diffusion, hanging drop Details: 0.1 M HEPES, pH 7.5, 10% (w/v) PEG 6000, 10% (v/v) MPD |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SSRF SSRF  / Beamline: BL19U1 / Wavelength: 0.9791 Å / Beamline: BL19U1 / Wavelength: 0.9791 Å | ||||||||||||||||||||||||||||||
| Detector | Type: DECTRIS PILATUS3 S 6M / Detector: PIXEL / Date: Sep 20, 2021 | ||||||||||||||||||||||||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength : 0.9791 Å / Relative weight: 1 : 0.9791 Å / Relative weight: 1 | ||||||||||||||||||||||||||||||
| Reflection | Resolution: 2.01→75.64 Å / Num. obs: 31436 / % possible obs: 98.9 % / Redundancy: 3.2 % / Biso Wilson estimate: 38.35 Å2 / CC1/2: 0.995 / Rmerge(I) obs: 0.091 / Rpim(I) all: 0.059 / Rrim(I) all: 0.108 / Net I/σ(I): 8.8 | ||||||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1
|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 3RZL Resolution: 2.2→46.84 Å / SU ML: 0.29 / Cross valid method: THROUGHOUT / σ(F): 1.36 / Phase error: 25.47 / Stereochemistry target values: ML
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.2→46.84 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Origin x: 22.085 Å / Origin y: -32.3095 Å / Origin z: -19.4773 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group | Selection details: all |
 Movie
Movie Controller
Controller


 PDBj
PDBj