+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8ew3 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Cryo EM structure of Vibrio cholerae NQR | ||||||
 Components Components | (Na(+)-translocating NADH-quinone reductase subunit ...) x 6 | ||||||
 Keywords Keywords |  ELECTRON TRANSPORT / ELECTRON TRANSPORT /  TRANSLOCASE / TRANSLOCASE /  complex complex | ||||||
| Function / homology |  Function and homology information Function and homology information NADH:ubiquinone reductase (Na+-transporting) / Gram-negative-bacterium-type cell wall / oxidoreductase activity, acting on NAD(P)H, quinone or similar compound as acceptor / sodium ion transport / respiratory electron transport chain / transmembrane transport / 2 iron, 2 sulfur cluster binding / FMN binding / NADH:ubiquinone reductase (Na+-transporting) / Gram-negative-bacterium-type cell wall / oxidoreductase activity, acting on NAD(P)H, quinone or similar compound as acceptor / sodium ion transport / respiratory electron transport chain / transmembrane transport / 2 iron, 2 sulfur cluster binding / FMN binding /  electron transfer activity / electron transfer activity /  metal ion binding / metal ion binding /  plasma membrane plasma membraneSimilarity search - Function | ||||||
| Biological species |   Vibrio cholerae O395 (bacteria) Vibrio cholerae O395 (bacteria) | ||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 2.65159 Å cryo EM / Resolution: 2.65159 Å | ||||||
 Authors Authors | Fuller, J.R. / Juarez, O. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: Novel cofactor binding motifs, electron transfer and ion pumping mechanisms of the respiratory complex NQR Authors: Juarez, O. / Fuller, J.R. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8ew3.cif.gz 8ew3.cif.gz | 379 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8ew3.ent.gz pdb8ew3.ent.gz | 245.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8ew3.json.gz 8ew3.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ew/8ew3 https://data.pdbj.org/pub/pdb/validation_reports/ew/8ew3 ftp://data.pdbj.org/pub/pdb/validation_reports/ew/8ew3 ftp://data.pdbj.org/pub/pdb/validation_reports/ew/8ew3 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  28641MC  8evuC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Na(+)-translocating NADH-quinone reductase subunit ... , 6 types, 6 molecules ABCDEF
| #1: Protein | Mass: 48680.734 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Vibrio cholerae O395 (bacteria) / Strain: O395N1 / Gene: nqrA, D6U24_04460, EYB64_17955, FLM02_04825 / Production host: Vibrio cholerae O395 (bacteria) / Strain: O395N1 / Gene: nqrA, D6U24_04460, EYB64_17955, FLM02_04825 / Production host:   Vibrio cholerae O395 (bacteria) / Strain (production host): O395N1 Vibrio cholerae O395 (bacteria) / Strain (production host): O395N1References: UniProt: A0A2D2BC37,  NADH:ubiquinone reductase (Na+-transporting) NADH:ubiquinone reductase (Na+-transporting) |
|---|---|
| #2: Protein | Mass: 45390.883 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Vibrio cholerae O395 (bacteria) / Strain: O395N1 Vibrio cholerae O395 (bacteria) / Strain: O395N1Gene: nqrB, D6U24_04465, ERS013186_02082, ERS013198_02508, ERS013199_02395, ERS013200_04117, ERS013202_01883, ERS013206_02987, ERS013207_01958, EYB64_17950, F0H40_10090, FLM02_04820, FLM12_12920, FXE67_12105 Production host:   Vibrio cholerae O395 (bacteria) / Strain (production host): O395N1 Vibrio cholerae O395 (bacteria) / Strain (production host): O395N1References: UniProt: A0A085SSI3,  NADH:ubiquinone reductase (Na+-transporting) NADH:ubiquinone reductase (Na+-transporting) |
| #3: Protein | Mass: 27652.270 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Vibrio cholerae O395 (bacteria) / Strain: O395N1 Vibrio cholerae O395 (bacteria) / Strain: O395N1Gene: nqrC, BC353_01370, D6U24_04470, ERS013165_00616, ERS013186_02083, ERS013198_02507, ERS013199_02396, ERS013200_04118, ERS013201_01110, ERS013202_01884, ERS013206_02988, ERS013207_01959, F0H40_ ...Gene: nqrC, BC353_01370, D6U24_04470, ERS013165_00616, ERS013186_02083, ERS013198_02507, ERS013199_02396, ERS013200_04118, ERS013201_01110, ERS013202_01884, ERS013206_02988, ERS013207_01959, F0H40_10095, FLM02_04815, FLM12_12915 Production host:   Vibrio cholerae O395 (bacteria) / Strain (production host): O395N1 Vibrio cholerae O395 (bacteria) / Strain (production host): O395N1References: UniProt: A0A085R7S2,  NADH:ubiquinone reductase (Na+-transporting) NADH:ubiquinone reductase (Na+-transporting) |
| #4: Protein | Mass: 22853.217 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Vibrio cholerae O395 (bacteria) / Strain: O395N1 / Gene: nqrD, VC0395_A1881, VC395_2408 / Production host: Vibrio cholerae O395 (bacteria) / Strain: O395N1 / Gene: nqrD, VC0395_A1881, VC395_2408 / Production host:   Vibrio cholerae O395 (bacteria) / Strain (production host): O395N1 Vibrio cholerae O395 (bacteria) / Strain (production host): O395N1References: UniProt: A5F5Y6,  NADH:ubiquinone reductase (Na+-transporting) NADH:ubiquinone reductase (Na+-transporting) |
| #5: Protein | Mass: 21481.678 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Vibrio cholerae O395 (bacteria) / Strain: O395N1 Vibrio cholerae O395 (bacteria) / Strain: O395N1Gene: nqrE, BC353_01360, D6U24_04480, ERS013165_00614, ERS013186_02085, ERS013198_02505, ERS013199_02398, ERS013200_04120, ERS013201_01112, ERS013202_01886, ERS013206_02990, ERS013207_01961, EYB64_ ...Gene: nqrE, BC353_01360, D6U24_04480, ERS013165_00614, ERS013186_02085, ERS013198_02505, ERS013199_02398, ERS013200_04120, ERS013201_01112, ERS013202_01886, ERS013206_02990, ERS013207_01961, EYB64_17935, F0315_08350, F0H40_10105, F0M16_14025, FLM02_04805, FLM12_12905 Production host:   Vibrio cholerae O395 (bacteria) / Strain (production host): O395N1 Vibrio cholerae O395 (bacteria) / Strain (production host): O395N1References: UniProt: A0A085QWM0,  NADH:ubiquinone reductase (Na+-transporting) NADH:ubiquinone reductase (Na+-transporting) |
| #6: Protein | Mass: 45113.492 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Vibrio cholerae O395 (bacteria) / Strain: O395N1 Vibrio cholerae O395 (bacteria) / Strain: O395N1Gene: nqrF, D6U24_04485, ERS013198_02504, ERS013199_02399, ERS013201_01113, ERS013202_01887, ERS013206_02991, EYB64_17930, FLM12_12900 Production host:   Vibrio cholerae O395 (bacteria) / Strain (production host): O395N1 Vibrio cholerae O395 (bacteria) / Strain (production host): O395N1References: UniProt: A0A085ST13,  NADH:ubiquinone reductase (Na+-transporting) NADH:ubiquinone reductase (Na+-transporting) |
-Non-polymers , 4 types, 5 molecules 
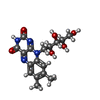
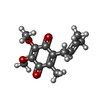




| #7: Chemical |  Flavin mononucleotide Flavin mononucleotide#8: Chemical | ChemComp-RBF / |  Riboflavin Riboflavin#9: Chemical | ChemComp-UQ1 / | #10: Chemical | ChemComp-FES / |  Iron–sulfur cluster Iron–sulfur cluster |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Na(+)-translocating NADH-quinone reductase / Type: COMPLEX / Entity ID: #1-#6 / Source: RECOMBINANT | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Experimental value: NO | ||||||||||||||||||||
| Source (natural) | Organism:   Vibrio cholerae O395 (bacteria) / Strain: O395N1 Vibrio cholerae O395 (bacteria) / Strain: O395N1 | ||||||||||||||||||||
| Source (recombinant) | Organism:   Vibrio cholerae O395 (bacteria) / Strain: O395N1 Vibrio cholerae O395 (bacteria) / Strain: O395N1 | ||||||||||||||||||||
| Buffer solution | pH: 7 / Details: 100 mM KCl, 1 mM EDTA, 50 mM HEPES, pH 7.0 | ||||||||||||||||||||
| Buffer component |
| ||||||||||||||||||||
| Specimen | Conc.: 2 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES : YES | ||||||||||||||||||||
| Specimen support | Details: Gatan Solarus plasma cleaner operated at 20W and using ambient/room air Grid material: COPPER / Grid mesh size: 200 divisions/in. / Grid type: Quantifoil R1.2/1.3 | ||||||||||||||||||||
Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 295 K Details: A 3.5 uL droplet of sample was applied to the grid surface and blotted for 4 s before plunging into liquid ethane. |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Nominal magnification: 81000 X / Nominal defocus max: 3200 nm / Nominal defocus min: 800 nm / Calibrated defocus min: 1200 nm / Calibrated defocus max: 3600 nm / Cs Bright-field microscopy / Nominal magnification: 81000 X / Nominal defocus max: 3200 nm / Nominal defocus min: 800 nm / Calibrated defocus min: 1200 nm / Calibrated defocus max: 3600 nm / Cs : 2.7 mm / C2 aperture diameter: 70 µm / Alignment procedure: ZEMLIN TABLEAU : 2.7 mm / C2 aperture diameter: 70 µm / Alignment procedure: ZEMLIN TABLEAU |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Average exposure time: 4.66 sec. / Electron dose: 50 e/Å2 / Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Num. of grids imaged: 1 / Num. of real images: 3159 / Details: 50 e-/A2 fractionated over 40 movie frames |
| EM imaging optics | Energyfilter name : GIF Bioquantum / Energyfilter slit width: 20 eV : GIF Bioquantum / Energyfilter slit width: 20 eV |
| Image scans | Width: 5760 / Height: 4092 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| ||||||||||||||||||||||||||||||||||||||||||||||||
| Image processing | Details: Micrograph movies were summed and dose-weighted using the motion correction algorithm implemented in RELION. | ||||||||||||||||||||||||||||||||||||||||||||||||
CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 304335 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Point symmetry : C1 (asymmetric) : C1 (asymmetric) | ||||||||||||||||||||||||||||||||||||||||||||||||
3D reconstruction | Resolution: 2.65159 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 73763 / Symmetry type: POINT | ||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | Protocol: FLEXIBLE FIT / Space: REAL / Target criteria: Cross-correlation Details: The model was refined by iterating between manual edits in Coot, molecular dynamics-guided edits using the ISOLDE ChimeraX package, and automated real-space refinement in Phenix. Models that ...Details: The model was refined by iterating between manual edits in Coot, molecular dynamics-guided edits using the ISOLDE ChimeraX package, and automated real-space refinement in Phenix. Models that had been automatically rebuilt using Rosetta were used as guides during manual refinement. | ||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | Cross valid method: NONE Stereochemistry target values: GeoStd + Monomer Library + CDL v1.2 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 70.1 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller




 PDBj
PDBj















