+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7z3k | ||||||
|---|---|---|---|---|---|---|---|
| Title | Autotaxin in complex with orthosteric site-binder CpdA | ||||||
 Components Components | Isoform 2 of Ectonucleotide pyrophosphatase/phosphodiesterase family member 2 | ||||||
 Keywords Keywords |  HYDROLASE / HYDROLASE /  ENPP2 / ENPP2 /  lysophosphatidic acid / lipid chaperone / / pulmonary fibrosis / lysophosphatidic acid / lipid chaperone / / pulmonary fibrosis /  GPCR / drug development / small-molecule inhibitor GPCR / drug development / small-molecule inhibitor | ||||||
| Function / homology |  Function and homology information Function and homology informationresponse to polycyclic arene /  alkylglycerophosphoethanolamine phosphodiesterase / sphingolipid catabolic process / phospholipid catabolic process / phosphatidylcholine catabolic process / alkylglycerophosphoethanolamine phosphodiesterase / sphingolipid catabolic process / phospholipid catabolic process / phosphatidylcholine catabolic process /  lysophospholipase activity / positive regulation of lamellipodium morphogenesis / lysophospholipase activity / positive regulation of lamellipodium morphogenesis /  phosphodiesterase I activity / scavenger receptor activity / phosphodiesterase I activity / scavenger receptor activity /  alkylglycerophosphoethanolamine phosphodiesterase activity ...response to polycyclic arene / alkylglycerophosphoethanolamine phosphodiesterase activity ...response to polycyclic arene /  alkylglycerophosphoethanolamine phosphodiesterase / sphingolipid catabolic process / phospholipid catabolic process / phosphatidylcholine catabolic process / alkylglycerophosphoethanolamine phosphodiesterase / sphingolipid catabolic process / phospholipid catabolic process / phosphatidylcholine catabolic process /  lysophospholipase activity / positive regulation of lamellipodium morphogenesis / lysophospholipase activity / positive regulation of lamellipodium morphogenesis /  phosphodiesterase I activity / scavenger receptor activity / phosphodiesterase I activity / scavenger receptor activity /  alkylglycerophosphoethanolamine phosphodiesterase activity / alkylglycerophosphoethanolamine phosphodiesterase activity /  polysaccharide binding / positive regulation of oligodendrocyte differentiation / negative regulation of cell-matrix adhesion / positive regulation of epithelial cell migration / positive regulation of focal adhesion assembly / polysaccharide binding / positive regulation of oligodendrocyte differentiation / negative regulation of cell-matrix adhesion / positive regulation of epithelial cell migration / positive regulation of focal adhesion assembly /  estrous cycle / phospholipid metabolic process / positive regulation of substrate adhesion-dependent cell spreading / estrous cycle / phospholipid metabolic process / positive regulation of substrate adhesion-dependent cell spreading /  regulation of cell migration / cellular response to cadmium ion / cell chemotaxis / cellular response to estradiol stimulus / positive regulation of peptidyl-tyrosine phosphorylation / regulation of cell migration / cellular response to cadmium ion / cell chemotaxis / cellular response to estradiol stimulus / positive regulation of peptidyl-tyrosine phosphorylation /  nucleic acid binding / nucleic acid binding /  immune response / immune response /  calcium ion binding / positive regulation of cell population proliferation / calcium ion binding / positive regulation of cell population proliferation /  extracellular space / zinc ion binding extracellular space / zinc ion bindingSimilarity search - Function | ||||||
| Biological species |   Rattus norvegicus (Norway rat) Rattus norvegicus (Norway rat) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2 Å MOLECULAR REPLACEMENT / Resolution: 2 Å | ||||||
 Authors Authors | Salgado-Polo, F. / Ford, P. / Heckmann, B. / Perrakis, A. | ||||||
| Funding support | 1items
| ||||||
 Citation Citation |  Journal: Cell Chem Biol / Year: 2023 Journal: Cell Chem Biol / Year: 2023Title: Autotaxin facilitates selective LPA receptor signaling. Authors: Salgado-Polo, F. / Borza, R. / Matsoukas, M.T. / Marsais, F. / Jagerschmidt, C. / Waeckel, L. / Moolenaar, W.H. / Ford, P. / Heckmann, B. / Perrakis, A. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7z3k.cif.gz 7z3k.cif.gz | 604.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7z3k.ent.gz pdb7z3k.ent.gz | Display |  PDB format PDB format | |
| PDBx/mmJSON format |  7z3k.json.gz 7z3k.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/z3/7z3k https://data.pdbj.org/pub/pdb/validation_reports/z3/7z3k ftp://data.pdbj.org/pub/pdb/validation_reports/z3/7z3k ftp://data.pdbj.org/pub/pdb/validation_reports/z3/7z3k | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7z3lC  2xr9S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 1 molecules AAA
| #1: Protein | Mass: 92590.125 Da / Num. of mol.: 1 / Mutation: N53A N410A N806A Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Rattus norvegicus (Norway rat) / Gene: Enpp2, Atx, Npps2 / Plasmid: pcDNA5.1 / Cell line (production host): HEK293T / Production host: Rattus norvegicus (Norway rat) / Gene: Enpp2, Atx, Npps2 / Plasmid: pcDNA5.1 / Cell line (production host): HEK293T / Production host:   Homo sapiens (human) Homo sapiens (human)References: UniProt: Q64610,  alkylglycerophosphoethanolamine phosphodiesterase alkylglycerophosphoethanolamine phosphodiesterase |
|---|
-Sugars , 2 types, 2 molecules 
| #2: Polysaccharide | beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose / Mass: 383.349 Da / Num. of mol.: 1 / Mass: 383.349 Da / Num. of mol.: 1Source method: isolated from a genetically manipulated source |
|---|---|
| #3: Sugar | ChemComp-NAG /  N-Acetylglucosamine N-Acetylglucosamine |
-Non-polymers , 9 types, 566 molecules 
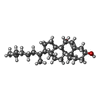





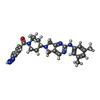









| #4: Chemical | | #5: Chemical | ChemComp-5JK / |  7α-Hydroxycholesterol 7α-Hydroxycholesterol#6: Chemical | ChemComp-CA / | #7: Chemical | ChemComp-IOD /  Iodide Iodide#8: Chemical | ChemComp-SCN /  Thiocyanate Thiocyanate#9: Chemical | #10: Chemical | ChemComp-GOL /  Glycerol Glycerol#11: Chemical | ChemComp-IA0 / [( | #12: Water | ChemComp-HOH / |  Water Water |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.24 Å3/Da / Density % sol: 45 % |
|---|---|
Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop Details: ATX was incubated with each screened compound at a 1:10 (protein:compound) ratio for at least 30 minutes. Crystals were grown for at least 7 days in a 24-well optimization screen: 18 to 20% ...Details: ATX was incubated with each screened compound at a 1:10 (protein:compound) ratio for at least 30 minutes. Crystals were grown for at least 7 days in a 24-well optimization screen: 18 to 20% PEG 3350, 0.1 to 0.4 M NaSCN, and 0.1 to 0.4 M NH4I. |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SLS SLS  / Beamline: X06DA / Wavelength: 0.999995 Å / Beamline: X06DA / Wavelength: 0.999995 Å | ||||||||||||||||||||||||
| Detector | Type: DECTRIS PILATUS 2M-F / Detector: PIXEL / Date: Jul 5, 2020 | ||||||||||||||||||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||||||||||||||
| Radiation wavelength | Wavelength : 0.999995 Å / Relative weight: 1 : 0.999995 Å / Relative weight: 1 | ||||||||||||||||||||||||
| Reflection | Resolution: 2→43.68 Å / Num. obs: 52303 / % possible obs: 98 % / Redundancy: 3.2 % / CC1/2: 0.985 / Rmerge(I) obs: 0.137 / Rpim(I) all: 0.137 / Rrim(I) all: 0.194 / Net I/σ(I): 5.6 | ||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1
|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 2xr9 Resolution: 2→43.68 Å / Cor.coef. Fo:Fc: 0.942 / Cor.coef. Fo:Fc free: 0.912 / SU B: 12.667 / SU ML: 0.173 / Cross valid method: FREE R-VALUE / ESU R: 0.24 / ESU R Free: 0.188 Details: Hydrogens have been added in their riding positions
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.7 Å / Shrinkage radii: 0.7 Å / VDW probe radii: 1 Å / Solvent model: MASK BULK SOLVENT | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 26.899 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2→43.68 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Origin x: 0.0101 Å / Origin y: -0.0737 Å / Origin z: 0.1439 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller



 PDBj
PDBj










