+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7ul5 | ||||||
|---|---|---|---|---|---|---|---|
| Title | CryoEM Structure of Inactive SSTR2 bound to Nb6 | ||||||
 Components Components |
| ||||||
 Keywords Keywords | MEMBRANE PROTEIN / Antagonist / Complex | ||||||
| Function / homology |  Function and homology information Function and homology informationdynorphin receptor activity / response to acrylamide / regulation of saliva secretion / sensory perception of temperature stimulus / positive regulation of eating behavior / somatostatin receptor activity / adenylate cyclase-inhibiting opioid receptor signaling pathway / G protein-coupled opioid receptor activity / negative regulation of luteinizing hormone secretion / G protein-coupled opioid receptor signaling pathway ...dynorphin receptor activity / response to acrylamide / regulation of saliva secretion / sensory perception of temperature stimulus / positive regulation of eating behavior / somatostatin receptor activity / adenylate cyclase-inhibiting opioid receptor signaling pathway / G protein-coupled opioid receptor activity / negative regulation of luteinizing hormone secretion / G protein-coupled opioid receptor signaling pathway / peristalsis / positive regulation of dopamine secretion / sensory perception / positive regulation of potassium ion transmembrane transport / conditioned place preference / maternal behavior / receptor serine/threonine kinase binding / neuropeptide binding / cellular response to glucocorticoid stimulus / positive regulation of p38MAPK cascade / eating behavior / response to starvation / behavioral response to cocaine / G protein-coupled receptor signaling pathway, coupled to cyclic nucleotide second messenger / neuropeptide signaling pathway / estrous cycle / MECP2 regulates neuronal receptors and channels / axon terminus / forebrain development / sensory perception of pain / T-tubule / cellular response to estradiol stimulus / adenylate cyclase-inhibiting G protein-coupled receptor signaling pathway / Peptide ligand-binding receptors / cerebellum development / sarcoplasmic reticulum / locomotory behavior / PDZ domain binding / cellular response to glucose stimulus / response to nicotine / response to insulin / synaptic vesicle membrane / response to estrogen / presynaptic membrane / phospholipase C-activating G protein-coupled receptor signaling pathway / G alpha (i) signalling events / spermatogenesis / cellular response to lipopolysaccharide / chemical synaptic transmission / postsynaptic membrane / perikaryon / response to ethanol / defense response to virus / neuron projection / immune response / negative regulation of cell population proliferation / dendrite / mitochondrion / nucleoplasm / membrane / plasma membrane / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.1 Å | ||||||
 Authors Authors | Robertson, M.J. / Skiniotis, G. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2022 Journal: Nat Struct Mol Biol / Year: 2022Title: Structure determination of inactive-state GPCRs with a universal nanobody. Authors: Michael J Robertson / Makaía M Papasergi-Scott / Feng He / Alpay B Seven / Justin G Meyerowitz / Ouliana Panova / Maria Claudia Peroto / Tao Che / Georgios Skiniotis /  Abstract: Cryogenic electron microscopy (cryo-EM) has widened the field of structure-based drug discovery by allowing for routine determination of membrane protein structures previously intractable. Despite ...Cryogenic electron microscopy (cryo-EM) has widened the field of structure-based drug discovery by allowing for routine determination of membrane protein structures previously intractable. Despite representing one of the largest classes of therapeutic targets, most inactive-state G protein-coupled receptors (GPCRs) have remained inaccessible for cryo-EM because their small size and membrane-embedded nature impedes projection alignment for high-resolution map reconstructions. Here we demonstrate that the same single-chain camelid antibody (nanobody) recognizing a grafted intracellular loop can be used to obtain cryo-EM structures of inactive-state GPCRs at resolutions comparable or better than those obtained by X-ray crystallography. Using this approach, we obtained structures of neurotensin 1 receptor bound to antagonist SR48692, μ-opioid receptor bound to alvimopan, apo somatostatin receptor 2 and histamine receptor 2 bound to famotidine. We expect this rapid, straightforward approach to facilitate the broad exploration of GPCR inactive states without the need for extensive engineering and crystallization. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7ul5.cif.gz 7ul5.cif.gz | 74.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7ul5.ent.gz pdb7ul5.ent.gz | 54.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7ul5.json.gz 7ul5.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  7ul5_validation.pdf.gz 7ul5_validation.pdf.gz | 1.1 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  7ul5_full_validation.pdf.gz 7ul5_full_validation.pdf.gz | 1.1 MB | Display | |
| Data in XML |  7ul5_validation.xml.gz 7ul5_validation.xml.gz | 24.9 KB | Display | |
| Data in CIF |  7ul5_validation.cif.gz 7ul5_validation.cif.gz | 33 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ul/7ul5 https://data.pdbj.org/pub/pdb/validation_reports/ul/7ul5 ftp://data.pdbj.org/pub/pdb/validation_reports/ul/7ul5 ftp://data.pdbj.org/pub/pdb/validation_reports/ul/7ul5 | HTTPS FTP |
-Related structure data
| Related structure data | 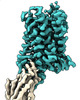 26592MC  7ul2C  7ul3C  7ul4C M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 46542.086 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: SSTR2, OPRK1, OPRK / Production host: Homo sapiens (human) / Gene: SSTR2, OPRK1, OPRK / Production host:  |
|---|---|
| #2: Antibody | Mass: 14730.255 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Source (natural) |
| ||||||||||||||||||||||||
| Source (recombinant) |
| ||||||||||||||||||||||||
| Buffer solution | pH: 7.5 | ||||||||||||||||||||||||
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | ||||||||||||||||||||||||
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: OTHER FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: OTHER |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 1800 nm / Nominal defocus min: 800 nm |
| Image recording | Electron dose: 68.93 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) |
- Processing
Processing
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION |
|---|---|
| 3D reconstruction | Resolution: 3.1 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 263630 / Symmetry type: POINT |
 Movie
Movie Controller
Controller





 PDBj
PDBj















