[English] 日本語
 Yorodumi
Yorodumi- PDB-7u2b: Cryo-electron microscopy structure of human mt-SerRS in complex w... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7u2b | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-electron microscopy structure of human mt-SerRS in complex with mt-tRNA(GCU-TL) | |||||||||||||||
 Components Components |
| |||||||||||||||
 Keywords Keywords | ligase/RNA /  tRNA / SerRS / ligase-RNA complex / tRNA / SerRS / ligase-RNA complex /  mitochondria / mitochondria /  aminoacylation aminoacylation | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationmitochondrial seryl-tRNA aminoacylation / Mitochondrial tRNA aminoacylation /  serine-tRNA ligase / serine-tRNA ligase /  serine-tRNA ligase activity / seryl-tRNA aminoacylation / serine-tRNA ligase activity / seryl-tRNA aminoacylation /  tRNA binding / tRNA binding /  mitochondrial matrix / mitochondrial matrix /  mitochondrion / mitochondrion /  RNA binding / RNA binding /  ATP binding ATP bindingSimilarity search - Function | |||||||||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 4.1 Å cryo EM / Resolution: 4.1 Å | |||||||||||||||
 Authors Authors | Hirschi, M. / Kuhle, B. / Doerfel, L. / Schimmel, P. / Lander, G. | |||||||||||||||
| Funding support |  United States, 4items United States, 4items
| |||||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2022 Journal: Nat Commun / Year: 2022Title: Structural basis for shape-selective recognition and aminoacylation of a D-armless human mitochondrial tRNA. Authors: Bernhard Kuhle / Marscha Hirschi / Lili K Doerfel / Gabriel C Lander / Paul Schimmel /  Abstract: Human mitochondrial gene expression relies on the specific recognition and aminoacylation of mitochondrial tRNAs (mtRNAs) by nuclear-encoded mitochondrial aminoacyl-tRNA synthetases (mt-aaRSs). ...Human mitochondrial gene expression relies on the specific recognition and aminoacylation of mitochondrial tRNAs (mtRNAs) by nuclear-encoded mitochondrial aminoacyl-tRNA synthetases (mt-aaRSs). Despite their essential role in cellular energy homeostasis, strong mutation pressure and genetic drift have led to an unparalleled sequence erosion of animal mtRNAs. The structural and functional consequences of this erosion are not understood. Here, we present cryo-EM structures of the human mitochondrial seryl-tRNA synthetase (mSerRS) in complex with mtRNA. These structures reveal a unique mechanism of substrate recognition and aminoacylation. The mtRNA is highly degenerated, having lost the entire D-arm, tertiary core, and stable L-shaped fold that define canonical tRNAs. Instead, mtRNA evolved unique structural innovations, including a radically altered T-arm topology that serves as critical identity determinant in an unusual shape-selective readout mechanism by mSerRS. Our results provide a molecular framework to understand the principles of mito-nuclear co-evolution and specialized mechanisms of tRNA recognition in mammalian mitochondrial gene expression. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7u2b.cif.gz 7u2b.cif.gz | 191.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7u2b.ent.gz pdb7u2b.ent.gz | 149.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7u2b.json.gz 7u2b.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/u2/7u2b https://data.pdbj.org/pub/pdb/validation_reports/u2/7u2b ftp://data.pdbj.org/pub/pdb/validation_reports/u2/7u2b ftp://data.pdbj.org/pub/pdb/validation_reports/u2/7u2b | HTTPS FTP |
|---|
-Related structure data
| Related structure data | 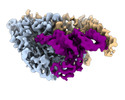 26311MC  7tzbC  7u2aC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: RNA chain | Mass: 16884.113 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Production host: Homo sapiens (human) / Production host:   Escherichia coli (E. coli) Escherichia coli (E. coli) | ||||
|---|---|---|---|---|---|
| #2: Protein | Mass: 58358.391 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: SARS2, SARSM / Production host: Homo sapiens (human) / Gene: SARS2, SARSM / Production host:   Escherichia coli (E. coli) / References: UniProt: Q9NP81, Escherichia coli (E. coli) / References: UniProt: Q9NP81,  serine-tRNA ligase serine-tRNA ligase#3: Chemical | Has ligand of interest | N | |
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Human mt-SerRS in complex with mt-tRNA(GCU-TL) and SerSA Type: COMPLEX / Entity ID: #1-#2 / Source: RECOMBINANT |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Source (recombinant) | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Buffer solution | pH: 7.5 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES : YES |
Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TECNAI ARCTICA |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 200 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 200 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 1200 nm / Nominal defocus min: 800 nm Bright-field microscopy / Nominal defocus max: 1200 nm / Nominal defocus min: 800 nm |
| Image recording | Electron dose: 66 e/Å2 / Film or detector model: GATAN K2 SUMMIT (4k x 4k) |
- Processing
Processing
CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION |
|---|---|
3D reconstruction | Resolution: 4.1 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 118269 / Symmetry type: POINT |
 Movie
Movie Controller
Controller


 PDBj
PDBj