[English] 日本語
 Yorodumi
Yorodumi- PDB-7bu6: Structure of human beta1 adrenergic receptor bound to norepinephr... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7bu6 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Structure of human beta1 adrenergic receptor bound to norepinephrine and nanobody 6B9 | ||||||
 Components Components |
| ||||||
 Keywords Keywords |  MEMBRANE PROTEIN / MEMBRANE PROTEIN /  G protein coupled receptor G protein coupled receptor | ||||||
| Function / homology |  Function and homology information Function and homology informationviral release from host cell by cytolysis / peptidoglycan catabolic process / cell wall macromolecule catabolic process /  lysozyme / lysozyme /  lysozyme activity / host cell cytoplasm / defense response to bacterium lysozyme activity / host cell cytoplasm / defense response to bacteriumSimilarity search - Function | ||||||
| Biological species |   Enterobacteria phage T4 (virus) Enterobacteria phage T4 (virus)  Homo sapiens (human) Homo sapiens (human) Camelidae mixed library (mammal) Camelidae mixed library (mammal) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.7 Å MOLECULAR REPLACEMENT / Resolution: 2.7 Å | ||||||
 Authors Authors | Xu, X. / Kaindl, J. / Clark, M. / Hubner, H. / Hirata, K. / Sunahara, R. / Gmeiner, P. / Kobilka, B.K. / Liu, X. | ||||||
 Citation Citation |  Journal: Cell Res. / Year: 2021 Journal: Cell Res. / Year: 2021Title: Binding pathway determines norepinephrine selectivity for the human beta 1 AR over beta 2 AR. Authors: Xu, X. / Kaindl, J. / Clark, M.J. / Hubner, H. / Hirata, K. / Sunahara, R.K. / Gmeiner, P. / Kobilka, B.K. / Liu, X. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7bu6.cif.gz 7bu6.cif.gz | 145.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7bu6.ent.gz pdb7bu6.ent.gz | 98.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7bu6.json.gz 7bu6.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/bu/7bu6 https://data.pdbj.org/pub/pdb/validation_reports/bu/7bu6 ftp://data.pdbj.org/pub/pdb/validation_reports/bu/7bu6 ftp://data.pdbj.org/pub/pdb/validation_reports/bu/7bu6 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7btsC  7bu7C  7bvqC  4ldeS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||||||
| Unit cell |
|
- Components
Components
-Protein / Antibody / Sugars , 3 types, 3 molecules AB
| #1: Protein | Mass: 57625.109 Da / Num. of mol.: 1 / Mutation: C944T,C987A Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Enterobacteria phage T4 (virus), (gene. exp.) Enterobacteria phage T4 (virus), (gene. exp.)   Homo sapiens (human) Homo sapiens (human)Gene: e, T4Tp126, ADRB1, ADRB1R, B1AR / Production host:   Spodoptera frugiperda (fall armyworm) / References: UniProt: D9IEF7, Spodoptera frugiperda (fall armyworm) / References: UniProt: D9IEF7,  lysozyme lysozyme |
|---|---|
| #2: Antibody | Mass: 14020.527 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Camelidae mixed library (mammal) Camelidae mixed library (mammal)Production host:   Escherichia coli 'BL21-Gold(DE3)pLysS AG' (bacteria) Escherichia coli 'BL21-Gold(DE3)pLysS AG' (bacteria) |
| #3: Polysaccharide | alpha-D-glucopyranose-(1-4)-alpha-D-glucopyranose / alpha-maltose |
-Non-polymers , 8 types, 16 molecules 



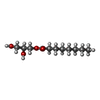










| #4: Chemical | ChemComp-E5E /  Norepinephrine Norepinephrine | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| #5: Chemical | ChemComp-SO4 /  Sulfate Sulfate#6: Chemical | ChemComp-NA / | #7: Chemical | ChemComp-EPE / |  HEPES HEPES#8: Chemical | ChemComp-480 / ( | #9: Chemical | ChemComp-CLR / |  Cholesterol Cholesterol#10: Chemical | ChemComp-PG4 / |  Polyethylene glycol Polyethylene glycol#11: Water | ChemComp-HOH / |  Water Water |
-Details
| Has ligand of interest | Y |
|---|---|
| Sequence details | THE GENEBANK ENTRY NP_000675 IS A REFERENCE SEQUENCE FOR THE RESIDUES FROM 171TH to 462TH OF CHAIN A. |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 4.14 Å3/Da / Density % sol: 73 % |
|---|---|
Crystal grow | Temperature: 293 K / Method: lipidic cubic phase Details: 100 mM HEPES, pH 7.5, 100-250 mM Sodium Sulfate, 39-43% PEG300, 10mM norepinephrine |
-Data collection
| Diffraction | Mean temperature: 80 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SPring-8 SPring-8  / Beamline: BL32XU / Wavelength: 1 Å / Beamline: BL32XU / Wavelength: 1 Å |
| Detector | Type: DECTRIS EIGER X 9M / Detector: PIXEL / Date: Dec 12, 2018 |
| Radiation | Monochromator: liquid nitrogen-cooled double crystal Si(111) Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1 Å / Relative weight: 1 : 1 Å / Relative weight: 1 |
| Reflection | Resolution: 2.7→50 Å / Num. obs: 32544 / % possible obs: 100 % / Redundancy: 41.6 % / Biso Wilson estimate: 56.67 Å2 / CC1/2: 0.995 / Net I/σ(I): 10.32 |
| Reflection shell | Resolution: 2.7→2.8 Å / Mean I/σ(I) obs: 0.99 / Num. unique obs: 3316 / CC1/2: 0.518 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 4lde Resolution: 2.7→19.98 Å / SU ML: 0.4112 / Cross valid method: FREE R-VALUE / σ(F): 1.33 / Phase error: 27.68 Stereochemistry target values: GeoStd + Monomer Library + CDL v1.2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 68.86 Å2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.7→19.98 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj



























