[English] 日本語
 Yorodumi
Yorodumi- PDB-6tvn: Crystal Structure of 5-bromoindoline-2,3-dione covalently bound t... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6tvn | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal Structure of 5-bromoindoline-2,3-dione covalently bound to the PH domain of Bruton's tyrosine kinase | ||||||
 Components Components | Tyrosine-protein kinase BTK | ||||||
 Keywords Keywords |  TRANSFERASE / BTK / Covalent fragments / surface entrophy reduction / TRANSFERASE / BTK / Covalent fragments / surface entrophy reduction /  crystal engineering crystal engineering | ||||||
| Function / homology |  Function and homology information Function and homology informationregulation of B cell cytokine production / proteoglycan catabolic process / monocyte proliferation / positive regulation of interleukin-17A production / regulation of B cell apoptotic process / eosinophil homeostasis / positive regulation of type III hypersensitivity / B cell affinity maturation / positive regulation of synoviocyte proliferation / histamine secretion by mast cell ...regulation of B cell cytokine production / proteoglycan catabolic process / monocyte proliferation / positive regulation of interleukin-17A production / regulation of B cell apoptotic process / eosinophil homeostasis / positive regulation of type III hypersensitivity / B cell affinity maturation / positive regulation of synoviocyte proliferation / histamine secretion by mast cell / neutrophil homeostasis / cellular response to molecule of fungal origin / positive regulation of type I hypersensitivity / cellular response to interleukin-7 / MyD88 deficiency (TLR2/4) / MyD88-dependent toll-like receptor signaling pathway / IRAK4 deficiency (TLR2/4) / MyD88:MAL(TIRAP) cascade initiated on plasma membrane / positive regulation of B cell differentiation / positive regulation of immunoglobulin production / positive regulation of NLRP3 inflammasome complex assembly / negative regulation of interleukin-10 production / phospholipase activator activity / B cell activation / negative regulation of B cell proliferation / Fc-epsilon receptor signaling pathway / mesoderm development / phosphatidylinositol-3,4,5-trisphosphate binding /  phospholipase binding / RHO GTPases Activate WASPs and WAVEs / positive regulation of phagocytosis / positive regulation of B cell proliferation / cell maturation / FCERI mediated Ca+2 mobilization / Antigen activates B Cell Receptor (BCR) leading to generation of second messengers / calcium-mediated signaling / apoptotic signaling pathway / FCGR3A-mediated phagocytosis / B cell receptor signaling pathway / phospholipase binding / RHO GTPases Activate WASPs and WAVEs / positive regulation of phagocytosis / positive regulation of B cell proliferation / cell maturation / FCERI mediated Ca+2 mobilization / Antigen activates B Cell Receptor (BCR) leading to generation of second messengers / calcium-mediated signaling / apoptotic signaling pathway / FCGR3A-mediated phagocytosis / B cell receptor signaling pathway /  non-specific protein-tyrosine kinase / non-membrane spanning protein tyrosine kinase activity / G beta:gamma signalling through BTK / Regulation of actin dynamics for phagocytic cup formation / cellular response to reactive oxygen species / peptidyl-tyrosine phosphorylation / positive regulation of interleukin-6 production / positive regulation of tumor necrosis factor production / G alpha (12/13) signalling events / DAP12 signaling / positive regulation of NF-kappaB transcription factor activity / T cell receptor signaling pathway / ER-Phagosome pathway / cytoplasmic vesicle / G alpha (q) signalling events / non-specific protein-tyrosine kinase / non-membrane spanning protein tyrosine kinase activity / G beta:gamma signalling through BTK / Regulation of actin dynamics for phagocytic cup formation / cellular response to reactive oxygen species / peptidyl-tyrosine phosphorylation / positive regulation of interleukin-6 production / positive regulation of tumor necrosis factor production / G alpha (12/13) signalling events / DAP12 signaling / positive regulation of NF-kappaB transcription factor activity / T cell receptor signaling pathway / ER-Phagosome pathway / cytoplasmic vesicle / G alpha (q) signalling events /  protein tyrosine kinase activity / protein tyrosine kinase activity /  adaptive immune response / Potential therapeutics for SARS / response to lipopolysaccharide / intracellular signal transduction / adaptive immune response / Potential therapeutics for SARS / response to lipopolysaccharide / intracellular signal transduction /  membrane raft / membrane raft /  protein phosphorylation / protein phosphorylation /  innate immune response / perinuclear region of cytoplasm / innate immune response / perinuclear region of cytoplasm /  ATP binding / identical protein binding / ATP binding / identical protein binding /  metal ion binding / metal ion binding /  nucleus / nucleus /  plasma membrane / plasma membrane /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.31 Å MOLECULAR REPLACEMENT / Resolution: 2.31 Å | ||||||
 Authors Authors | Brear, P. / Wagstaff, J. / Hyvonen, M. | ||||||
| Funding support |  United Kingdom, 1items United Kingdom, 1items
| ||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: Crystal Structure of 1-methylindoline-2,3-dione covalently bound to the PH domain of Bruton's tyrosine kinase mutant R28C Authors: Brear, P. / Fischer, G. / May, M. / Pantelejevs, T. / Mathieu, R. / Rossmann, M. / Wagstaff, J. / Blaszczyk, B. / Hyvonen, M. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6tvn.cif.gz 6tvn.cif.gz | 141.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6tvn.ent.gz pdb6tvn.ent.gz | 111.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6tvn.json.gz 6tvn.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/tv/6tvn https://data.pdbj.org/pub/pdb/validation_reports/tv/6tvn ftp://data.pdbj.org/pub/pdb/validation_reports/tv/6tvn ftp://data.pdbj.org/pub/pdb/validation_reports/tv/6tvn | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6tseC  6tt2C  6tuhC  1btkS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 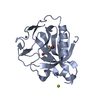
| ||||||||
| 2 | 
| ||||||||
| 3 | 
| ||||||||
| 4 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 19950.949 Da / Num. of mol.: 4 / Fragment: PH DOMAIN AND BTK MOTIF / Mutation: C145A Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: BTK, AGMX1, ATK, BPK / Plasmid: pBAT4 / Production host: Homo sapiens (human) / Gene: BTK, AGMX1, ATK, BPK / Plasmid: pBAT4 / Production host:   Escherichia coli BL21(DE3) (bacteria) Escherichia coli BL21(DE3) (bacteria)References: UniProt: Q06187,  non-specific protein-tyrosine kinase non-specific protein-tyrosine kinase#2: Chemical | ChemComp-NYQ / #3: Chemical | ChemComp-ZN / #4: Chemical | #5: Water | ChemComp-HOH / |  Water WaterHas ligand of interest | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.17 Å3/Da / Density % sol: 43.36 % / Mosaicity: 0 ° |
|---|---|
Crystal grow | Temperature: 298 K / Method: vapor diffusion, sitting drop / pH: 8.5 / Details: 0.1 M TRIS 8.5 pH, 32.5% w/v PEG 3350, 200mM MgCl2 |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  Diamond Diamond  / Beamline: I04 / Wavelength: 0.9795 Å / Beamline: I04 / Wavelength: 0.9795 Å | ||||||||||||||||||||||||||||||
| Detector | Type: DECTRIS PILATUS 6M / Detector: PIXEL / Date: Sep 13, 2017 | ||||||||||||||||||||||||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength : 0.9795 Å / Relative weight: 1 : 0.9795 Å / Relative weight: 1 | ||||||||||||||||||||||||||||||
| Reflection | Resolution: 2.31→66.45 Å / Num. obs: 30254 / % possible obs: 100 % / Redundancy: 6.7 % / Biso Wilson estimate: 50.62 Å2 / CC1/2: 0.998 / Rmerge(I) obs: 0.096 / Rpim(I) all: 0.04 / Rrim(I) all: 0.104 / Net I/σ(I): 11.7 / Num. measured all: 201455 / Scaling rejects: 301 | ||||||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1
|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1BTK Resolution: 2.31→66.45 Å / Cor.coef. Fo:Fc: 0.933 / Cor.coef. Fo:Fc free: 0.911 / SU R Cruickshank DPI: 0.393 / Cross valid method: THROUGHOUT / σ(F): 0 / SU R Blow DPI: 0.362 / SU Rfree Blow DPI: 0.238 / SU Rfree Cruickshank DPI: 0.247
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 136.23 Å2 / Biso mean: 53.75 Å2 / Biso min: 12.75 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.31→66.45 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.31→2.39 Å / Rfactor Rfree error: 0 / Total num. of bins used: 15
|
 Movie
Movie Controller
Controller












 PDBj
PDBj


















