[English] 日本語
 Yorodumi
Yorodumi- PDB-6ak3: Crystal structure of the human prostaglandin E receptor EP3 bound... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6ak3 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Crystal structure of the human prostaglandin E receptor EP3 bound to prostaglandin E2 | |||||||||
 Components Components | Prostaglandin E2 receptor EP3 subtype,Soluble cytochrome b562 | |||||||||
 Keywords Keywords |  MEMBRANE PROTEIN / MEMBRANE PROTEIN /  GPCR / GPCR /  lipid / lipid /  signaling protein / signaling protein /  prostanoid receptor prostanoid receptor | |||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of gastric acid secretion / intestine smooth muscle contraction /  prostaglandin E receptor activity / Prostanoid ligand receptors / prostaglandin E receptor activity / Prostanoid ligand receptors /  cell death / positive regulation of fever generation / adenylate cyclase-activating G protein-coupled receptor signaling pathway / cell death / positive regulation of fever generation / adenylate cyclase-activating G protein-coupled receptor signaling pathway /  nuclear envelope / phospholipase C-activating G protein-coupled receptor signaling pathway / G alpha (i) signalling events ...negative regulation of gastric acid secretion / intestine smooth muscle contraction / nuclear envelope / phospholipase C-activating G protein-coupled receptor signaling pathway / G alpha (i) signalling events ...negative regulation of gastric acid secretion / intestine smooth muscle contraction /  prostaglandin E receptor activity / Prostanoid ligand receptors / prostaglandin E receptor activity / Prostanoid ligand receptors /  cell death / positive regulation of fever generation / adenylate cyclase-activating G protein-coupled receptor signaling pathway / cell death / positive regulation of fever generation / adenylate cyclase-activating G protein-coupled receptor signaling pathway /  nuclear envelope / phospholipase C-activating G protein-coupled receptor signaling pathway / G alpha (i) signalling events / positive regulation of cytosolic calcium ion concentration / nuclear envelope / phospholipase C-activating G protein-coupled receptor signaling pathway / G alpha (i) signalling events / positive regulation of cytosolic calcium ion concentration /  electron transfer activity / electron transfer activity /  periplasmic space / periplasmic space /  inflammatory response / iron ion binding / G protein-coupled receptor signaling pathway / inflammatory response / iron ion binding / G protein-coupled receptor signaling pathway /  heme binding / heme binding /  membrane / membrane /  plasma membrane plasma membraneSimilarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human)  Escherichia coli (E. coli) Escherichia coli (E. coli) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / Resolution: 2.9 Å SYNCHROTRON / Resolution: 2.9 Å | |||||||||
 Authors Authors | Morimoto, K. / Suno, R. / Iwata, S. / Kobayashi, T. | |||||||||
| Funding support |  Japan, 2items Japan, 2items
| |||||||||
 Citation Citation |  Journal: Nat. Chem. Biol. / Year: 2019 Journal: Nat. Chem. Biol. / Year: 2019Title: Crystal structure of the endogenous agonist-bound prostanoid receptor EP3. Authors: Morimoto, K. / Suno, R. / Hotta, Y. / Yamashita, K. / Hirata, K. / Yamamoto, M. / Narumiya, S. / Iwata, S. / Kobayashi, T. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6ak3.cif.gz 6ak3.cif.gz | 232.4 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6ak3.ent.gz pdb6ak3.ent.gz | 184.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6ak3.json.gz 6ak3.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ak/6ak3 https://data.pdbj.org/pub/pdb/validation_reports/ak/6ak3 ftp://data.pdbj.org/pub/pdb/validation_reports/ak/6ak3 ftp://data.pdbj.org/pub/pdb/validation_reports/ak/6ak3 | HTTPS FTP |
|---|
-Related structure data
| Similar structure data | |
|---|---|
| Experimental dataset #1 | Data reference:  10.11577/1482880 / Data set type: diffraction image data 10.11577/1482880 / Data set type: diffraction image data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
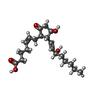
| #1: Protein | Mass: 46044.707 Da / Num. of mol.: 2 Mutation: A173I,V185S,N217Q,S258D,C289L,N308Q,M1007W,R1098I,H1102I Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human), (gene. exp.) Homo sapiens (human), (gene. exp.)   Escherichia coli (E. coli) Escherichia coli (E. coli)Gene: PTGER3, cybC / Production host:   Spodoptera frugiperda (fall armyworm) / References: UniProt: P43115, UniProt: P0ABE7 Spodoptera frugiperda (fall armyworm) / References: UniProt: P43115, UniProt: P0ABE7#2: Chemical |  Prostaglandin E2 Prostaglandin E2#3: Chemical |  POPC POPC |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.46 Å3/Da / Density % sol: 50.07 % |
|---|---|
Crystal grow | Temperature: 293 K / Method: lipidic cubic phase Details: buffer A (0.1 M MES-NaOH (pH 6.0), 35 % PEG300, 400 mM Na2SO4, 1% 1,2,3-heptanetriol, 0.2 mM ONO-AE3-240), buffer B (0.1 M MES-NaOH (pH 5.5-6.0), 30-40 % PEG300, 100 mM NaCl, 100 mM Li2SO4, ...Details: buffer A (0.1 M MES-NaOH (pH 6.0), 35 % PEG300, 400 mM Na2SO4, 1% 1,2,3-heptanetriol, 0.2 mM ONO-AE3-240), buffer B (0.1 M MES-NaOH (pH 5.5-6.0), 30-40 % PEG300, 100 mM NaCl, 100 mM Li2SO4, 1% 1,2,3-heptanetriol, 0.2 mM ONO-AE3-240), buffer C (0.1 M MES-NaOH (pH 6.1) or 0.1 M Tris-HCl (pH 7.5-8.0), 30 % PEG500MME, 200 mM (NH4)2SO4, 1% 1,2,3-heptanetriol, 0.2 mM ONO-AE3-240), buffer D (0.1 M MES-NaOH (pH 5.8-6.1), 30 % PEG300, 100 mM MgSO4, 1% 1,2,3-heptanetriol, 0.2 mM ONO-AE3-240), buffer E (0.1 M MES-NaOH (pH 5.5), 30 % PEG300, 100 mM K2SO4, 1% 1,2,3-heptanetriol, 0.2 mM ONO-AE3-240) |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SPring-8 SPring-8  / Beamline: BL32XU / Wavelength: 1 Å / Beamline: BL32XU / Wavelength: 1 Å |
| Detector | Type: DECTRIS EIGER X 9M / Detector: PIXEL / Date: Jan 22, 2018 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1 Å / Relative weight: 1 : 1 Å / Relative weight: 1 |
| Reflection | Resolution: 2.9→50 Å / Num. obs: 20206 / % possible obs: 99.9 % / Redundancy: 43.4 % / Net I/σ(I): 1.15 |
| Reflection shell | Resolution: 2.9→3.08 Å |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Resolution: 2.9→48.377 Å / SU ML: 0.37 / Cross valid method: FREE R-VALUE / σ(F): 1.34 / Phase error: 29.59 / Stereochemistry target values: ML
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.9→48.377 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj