[English] 日本語
 Yorodumi
Yorodumi- PDB-4rak: Crystal structure of nuclear receptor subfamily 1, group h, membe... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4rak | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of nuclear receptor subfamily 1, group h, member 2 (lxrb) complexed with partial agonist | ||||||
 Components Components | Oxysterols receptor LXR-beta | ||||||
 Keywords Keywords | TRANSCRIPTION/AGONIST / NHR / NR1H2 / LXR-B /  LXRB / UNR / NER-I / RIP15 / NER / LXRB / UNR / NER-I / RIP15 / NER /  Ligand Binding Domain / Ligand Binding Domain /  Nuclear Hormone Receptor / RXR / TRANSCRIPTION-AGONIST complex Nuclear Hormone Receptor / RXR / TRANSCRIPTION-AGONIST complex | ||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of secretion of lysosomal enzymes / positive regulation of high-density lipoprotein particle assembly / positive regulation of pancreatic juice secretion / phosphatidylcholine acyl-chain remodeling / positive regulation of cholesterol transport / negative regulation of response to endoplasmic reticulum stress / negative regulation of pinocytosis / positive regulation of lipoprotein lipase activity / apolipoprotein A-I receptor binding / positive regulation of triglyceride biosynthetic process ...positive regulation of secretion of lysosomal enzymes / positive regulation of high-density lipoprotein particle assembly / positive regulation of pancreatic juice secretion / phosphatidylcholine acyl-chain remodeling / positive regulation of cholesterol transport / negative regulation of response to endoplasmic reticulum stress / negative regulation of pinocytosis / positive regulation of lipoprotein lipase activity / apolipoprotein A-I receptor binding / positive regulation of triglyceride biosynthetic process / NR1H2 & NR1H3 regulate gene expression linked to triglyceride lipolysis in adipose / NR1H2 & NR1H3 regulate gene expression to limit cholesterol uptake / positive regulation of lipid storage / NR1H2 & NR1H3 regulate gene expression linked to lipogenesis / positive regulation of fatty acid biosynthetic process / negative regulation of lipid transport / negative regulation of cold-induced thermogenesis / NR1H2 & NR1H3 regulate gene expression to control bile acid homeostasis / negative regulation of type II interferon-mediated signaling pathway / negative regulation of cholesterol storage / negative regulation of macrophage derived foam cell differentiation / positive regulation of cholesterol efflux / nuclear retinoid X receptor binding / NR1H3 & NR1H2 regulate gene expression linked to cholesterol transport and efflux / VLDLR internalisation and degradation / positive regulation of protein metabolic process / hormone-mediated signaling pathway / cholesterol homeostasis / negative regulation of proteolysis / SUMOylation of intracellular receptors / mRNA transcription by RNA polymerase II / PPARA activates gene expression / chromatin DNA binding / positive regulation of miRNA transcription / Nuclear Receptor transcription pathway / RNA polymerase II transcription regulator complex /  nuclear receptor activity / nuclear receptor activity /  ATPase binding / DNA-binding transcription activator activity, RNA polymerase II-specific / ATPase binding / DNA-binding transcription activator activity, RNA polymerase II-specific /  cell differentiation / DNA-binding transcription factor activity, RNA polymerase II-specific / RNA polymerase II cis-regulatory region sequence-specific DNA binding / negative regulation of DNA-templated transcription / cell differentiation / DNA-binding transcription factor activity, RNA polymerase II-specific / RNA polymerase II cis-regulatory region sequence-specific DNA binding / negative regulation of DNA-templated transcription /  chromatin / positive regulation of DNA-templated transcription / negative regulation of transcription by RNA polymerase II / positive regulation of transcription by RNA polymerase II / chromatin / positive regulation of DNA-templated transcription / negative regulation of transcription by RNA polymerase II / positive regulation of transcription by RNA polymerase II /  DNA binding / zinc ion binding / DNA binding / zinc ion binding /  nucleoplasm / nucleoplasm /  nucleus / nucleus /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.04 Å MOLECULAR REPLACEMENT / Resolution: 2.04 Å | ||||||
 Authors Authors | Nanao, M. | ||||||
 Citation Citation |  Journal: Bioorg.Med.Chem.Lett. / Year: 2015 Journal: Bioorg.Med.Chem.Lett. / Year: 2015Title: Liver X Receptor (LXR) partial agonists: Biaryl pyrazoles and imidazoles displaying a preference for LXR beta. Authors: Kick, E. / Martin, R. / Xie, Y. / Flatt, B. / Schweiger, E. / Wang, T.L. / Busch, B. / Nyman, M. / Gu, X.H. / Yan, G. / Wagner, B. / Nanao, M. / Nguyen, L. / Stout, T. / Plonowski, A. / ...Authors: Kick, E. / Martin, R. / Xie, Y. / Flatt, B. / Schweiger, E. / Wang, T.L. / Busch, B. / Nyman, M. / Gu, X.H. / Yan, G. / Wagner, B. / Nanao, M. / Nguyen, L. / Stout, T. / Plonowski, A. / Schulman, I. / Ostrowski, J. / Kirchgessner, T. / Wexler, R. / Mohan, R. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4rak.cif.gz 4rak.cif.gz | 115.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4rak.ent.gz pdb4rak.ent.gz | 89 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4rak.json.gz 4rak.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ra/4rak https://data.pdbj.org/pub/pdb/validation_reports/ra/4rak ftp://data.pdbj.org/pub/pdb/validation_reports/ra/4rak ftp://data.pdbj.org/pub/pdb/validation_reports/ra/4rak | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
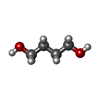
| #1: Protein | Mass: 30542.854 Da / Num. of mol.: 2 / Fragment: UNP residues 213-460 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: NR1H2, LXRB, NER, UNR / References: UniProt: P55055 Homo sapiens (human) / Gene: NR1H2, LXRB, NER, UNR / References: UniProt: P55055#2: Chemical |  1,4-Butanediol 1,4-Butanediol#3: Chemical | #4: Water | ChemComp-HOH / |  Water Water |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.43 Å3/Da / Density % sol: 49.35 % |
|---|
-Data collection
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SSRL SSRL  / Beamline: BL9-1 / Wavelength: 0.979 Å / Beamline: BL9-1 / Wavelength: 0.979 Å |
|---|---|
| Detector | Type: ADSC QUANTUM 315r / Detector: CCD |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 0.979 Å / Relative weight: 1 : 0.979 Å / Relative weight: 1 |
| Reflection | Resolution: 1.95→50 Å / Num. obs: 38284 / % possible obs: 87.2 % / Redundancy: 6.4 % |
- Processing
Processing
| Software | Name: REFMAC / Version: 5.2.0019 / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT / Resolution: 2.04→53.21 Å / Cor.coef. Fo:Fc: 0.953 / Cor.coef. Fo:Fc free: 0.938 / SU B: 5.595 / SU ML: 0.151 / Cross valid method: THROUGHOUT / ESU R: 0.217 / ESU R Free: 0.189 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS MOLECULAR REPLACEMENT / Resolution: 2.04→53.21 Å / Cor.coef. Fo:Fc: 0.953 / Cor.coef. Fo:Fc free: 0.938 / SU B: 5.595 / SU ML: 0.151 / Cross valid method: THROUGHOUT / ESU R: 0.217 / ESU R Free: 0.189 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 57.366 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze | Luzzati coordinate error obs: 0.304 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.04→53.21 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj