[English] 日本語
 Yorodumi
Yorodumi- PDB-4li8: TANKYRASE-1 complexed with small molecule inhibitor 2-[4-(4-fluor... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4li8 | ||||||
|---|---|---|---|---|---|---|---|
| Title | TANKYRASE-1 complexed with small molecule inhibitor 2-[4-(4-fluorobenzoyl)piperidin-1-yl]-N-[(4-oxo-3,5,7,8-tetrahydro-4H-pyrano[4,3-d]pyrimidin-2-yl)methyl]-N-(thiophen-2-ylmethyl)acetamide | ||||||
 Components Components | Tankyrase-1 | ||||||
 Keywords Keywords | TRANSFERASE/TRANSFERASE INHIBITOR / TRANSFERASE-TRANSFERASE INHIBITOR complex | ||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of telomeric DNA binding / negative regulation of maintenance of mitotic sister chromatid cohesion, telomeric / regulation of telomere maintenance via telomerase / XAV939 stabilizes AXIN /  NAD+ ADP-ribosyltransferase / negative regulation of telomere maintenance via telomere lengthening / protein auto-ADP-ribosylation / protein localization to chromosome, telomeric region / protein poly-ADP-ribosylation / NAD+ ADP-ribosyltransferase / negative regulation of telomere maintenance via telomere lengthening / protein auto-ADP-ribosylation / protein localization to chromosome, telomeric region / protein poly-ADP-ribosylation /  pericentriolar material ...negative regulation of telomeric DNA binding / negative regulation of maintenance of mitotic sister chromatid cohesion, telomeric / regulation of telomere maintenance via telomerase / XAV939 stabilizes AXIN / pericentriolar material ...negative regulation of telomeric DNA binding / negative regulation of maintenance of mitotic sister chromatid cohesion, telomeric / regulation of telomere maintenance via telomerase / XAV939 stabilizes AXIN /  NAD+ ADP-ribosyltransferase / negative regulation of telomere maintenance via telomere lengthening / protein auto-ADP-ribosylation / protein localization to chromosome, telomeric region / protein poly-ADP-ribosylation / NAD+ ADP-ribosyltransferase / negative regulation of telomere maintenance via telomere lengthening / protein auto-ADP-ribosylation / protein localization to chromosome, telomeric region / protein poly-ADP-ribosylation /  pericentriolar material / mitotic spindle pole / NAD+-protein ADP-ribosyltransferase activity / positive regulation of telomere capping / pericentriolar material / mitotic spindle pole / NAD+-protein ADP-ribosyltransferase activity / positive regulation of telomere capping /  Transferases; Glycosyltransferases; Pentosyltransferases / Transferases; Glycosyltransferases; Pentosyltransferases /  NAD+ ADP-ribosyltransferase activity / spindle assembly / mRNA transport / NAD+ ADP-ribosyltransferase activity / spindle assembly / mRNA transport /  nuclear pore / positive regulation of telomerase activity / positive regulation of telomere maintenance via telomerase / nuclear pore / positive regulation of telomerase activity / positive regulation of telomere maintenance via telomerase /  nucleotidyltransferase activity / mitotic spindle organization / TCF dependent signaling in response to WNT / Degradation of AXIN / peptidyl-threonine phosphorylation / nucleotidyltransferase activity / mitotic spindle organization / TCF dependent signaling in response to WNT / Degradation of AXIN / peptidyl-threonine phosphorylation /  Wnt signaling pathway / Regulation of PTEN stability and activity / protein polyubiquitination / positive regulation of canonical Wnt signaling pathway / Wnt signaling pathway / Regulation of PTEN stability and activity / protein polyubiquitination / positive regulation of canonical Wnt signaling pathway /  protein transport / protein transport /  histone binding / peptidyl-serine phosphorylation / histone binding / peptidyl-serine phosphorylation /  nuclear membrane / nuclear membrane /  chromosome, telomeric region / chromosome, telomeric region /  nuclear body / Ub-specific processing proteases / nuclear body / Ub-specific processing proteases /  cell division / cell division /  Golgi membrane / Golgi membrane /  Golgi apparatus / positive regulation of transcription by RNA polymerase II / zinc ion binding / Golgi apparatus / positive regulation of transcription by RNA polymerase II / zinc ion binding /  nucleoplasm / nucleoplasm /  nucleus / nucleus /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / FOURIER METHODS / Resolution: 2.521 Å SYNCHROTRON / FOURIER METHODS / Resolution: 2.521 Å | ||||||
 Authors Authors | Kirby, C.A. / Stams, T. | ||||||
 Citation Citation |  Journal: J.Med.Chem. / Year: 2013 Journal: J.Med.Chem. / Year: 2013Title: Identification of NVP-TNKS656: The Use of Structure-Efficiency Relationships To Generate a Highly Potent, Selective, and Orally Active Tankyrase Inhibitor. Authors: Shultz, M.D. / Cheung, A.K. / Kirby, C.A. / Firestone, B. / Fan, J. / Chen, C.H. / Chen, Z. / Chin, D.N. / Dipietro, L. / Fazal, A. / Feng, Y. / Fortin, P.D. / Gould, T. / Lagu, B. / Lei, H. ...Authors: Shultz, M.D. / Cheung, A.K. / Kirby, C.A. / Firestone, B. / Fan, J. / Chen, C.H. / Chen, Z. / Chin, D.N. / Dipietro, L. / Fazal, A. / Feng, Y. / Fortin, P.D. / Gould, T. / Lagu, B. / Lei, H. / Lenoir, F. / Majumdar, D. / Ochala, E. / Palermo, M.G. / Pham, L. / Pu, M. / Smith, T. / Stams, T. / Tomlinson, R.C. / Toure, B.B. / Visser, M. / Wang, R.M. / Waters, N.J. / Shao, W. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4li8.cif.gz 4li8.cif.gz | 98.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4li8.ent.gz pdb4li8.ent.gz | 75.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4li8.json.gz 4li8.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/li/4li8 https://data.pdbj.org/pub/pdb/validation_reports/li/4li8 ftp://data.pdbj.org/pub/pdb/validation_reports/li/4li8 ftp://data.pdbj.org/pub/pdb/validation_reports/li/4li8 | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 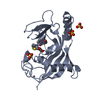
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein |  / TANK1 / ADP-ribosyltransferase diphtheria toxin-like 5 / ARTD5 / Poly [ADP-ribose] polymerase 5A / ...TANK1 / ADP-ribosyltransferase diphtheria toxin-like 5 / ARTD5 / Poly [ADP-ribose] polymerase 5A / TNKS-1 / TRF1-interacting ankyrin-related ADP-ribose polymerase / Tankyrase I / TANK1 / ADP-ribosyltransferase diphtheria toxin-like 5 / ARTD5 / Poly [ADP-ribose] polymerase 5A / ...TANK1 / ADP-ribosyltransferase diphtheria toxin-like 5 / ARTD5 / Poly [ADP-ribose] polymerase 5A / TNKS-1 / TRF1-interacting ankyrin-related ADP-ribose polymerase / Tankyrase IMass: 25409.711 Da / Num. of mol.: 2 / Fragment: CATALYTIC DOMAIN, UNP residues 1105-1325 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: TNKS, PARP5A, PARPL, TIN1, TINF1, TNKS1 / Production host: Homo sapiens (human) / Gene: TNKS, PARP5A, PARPL, TIN1, TINF1, TNKS1 / Production host:   Escherichia coli (E. coli) / References: UniProt: O95271, Escherichia coli (E. coli) / References: UniProt: O95271,  NAD+ ADP-ribosyltransferase NAD+ ADP-ribosyltransferase#2: Chemical | #3: Chemical | #4: Chemical | ChemComp-SO4 /  Sulfate Sulfate#5: Water | ChemComp-HOH / |  Water WaterSequence details | SEQUENCE CONFLICT M1266I PRESENT IN UNP ENTRY O95271 | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.33 Å3/Da / Density % sol: 47.18 % |
|---|---|
Crystal grow | Temperature: 298 K / Method: vapor diffusion, hanging drop / pH: 5.8 Details: EQUAL VOLUMES (2 UL) OF RESERVIOR SOLUTION AND PROTEIN SOLUTION WERE MIXED AND SET AS HANGING DROPS. 15% PEG3350, 100MM BIS-TRIS PH5.8, 310MM AMMONIUM SULFATE. PROTEIN SOLUTION: 20 MG/ML IN ...Details: EQUAL VOLUMES (2 UL) OF RESERVIOR SOLUTION AND PROTEIN SOLUTION WERE MIXED AND SET AS HANGING DROPS. 15% PEG3350, 100MM BIS-TRIS PH5.8, 310MM AMMONIUM SULFATE. PROTEIN SOLUTION: 20 MG/ML IN 25 MM TRIS PH8, 200MM NACL, 1MM TCEP, 1MM PJ34. CRYSTALS WERE THEN MOVED INTO A SOAKING SOLUTION CONTAINING 18% PEG 3350, 320MM AMMONIUM SULFATE, 100MM BIS- TRIS PH 5.8, 0.2 MM inhibitor. , VAPOR DIFFUSION, HANGING DROP, temperature 298K |
-Data collection
| Diffraction | Mean temperature: 100 K | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SLS SLS  / Beamline: X06SA / Wavelength: 1 / Wavelength: 1 Å / Beamline: X06SA / Wavelength: 1 / Wavelength: 1 Å | |||||||||
| Detector | Type: PSI PILATUS 6M / Detector: PIXEL / Date: Feb 1, 2009 | |||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | |||||||||
| Radiation wavelength |
| |||||||||
| Reflection | Resolution: 2.521→50 Å / Num. obs: 15321 / % possible obs: 98.2 % / Redundancy: 3.17 % / Rmerge(I) obs: 0.105 / Net I/σ(I): 9.41 | |||||||||
| Reflection shell | Resolution: 2.521→2.67 Å / Redundancy: 3.25 % / Rmerge(I) obs: 0.353 / Mean I/σ(I) obs: 3.57 / % possible all: 98.7 |
- Processing
Processing
| Software |
| ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : FOURIER METHODS / Resolution: 2.521→44.039 Å / σ(F): 1.36 : FOURIER METHODS / Resolution: 2.521→44.039 Å / σ(F): 1.36
| ||||||||||||||||||
| Displacement parameters | Biso mean: 35.17 Å2
| ||||||||||||||||||
| Refine analyze | Luzzati coordinate error obs: 0.37 Å | ||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.521→44.039 Å
| ||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||
| LS refinement shell | Resolution: 2.521→2.7156 Å
|
 Movie
Movie Controller
Controller























 PDBj
PDBj










