+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4j09 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal Structure of LpxA bound to RJPXD33 | ||||||
 Components Components |
| ||||||
 Keywords Keywords |  TRANSFERASE / TRANSFERASE /  acyltransferase / left-handed beta helix acyltransferase / left-handed beta helix | ||||||
| Function / homology |  Function and homology information Function and homology informationcarboxylic acid transport / acyl-[acyl-carrier-protein]-UDP-N-acetylglucosamine O-acyltransferase / acyl-[acyl-carrier-protein]-UDP-N-acetylglucosamine O-acyltransferase activity / carboxylic acid transmembrane transporter activity / lipid A biosynthetic process / membrane => GO:0016020 / identical protein binding /  plasma membrane / plasma membrane /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |   Escherichia coli (E. coli) Escherichia coli (E. coli) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / MOLECULAR REPLACEMENT /  molecular replacement / Resolution: 1.9 Å molecular replacement / Resolution: 1.9 Å | ||||||
 Authors Authors | Jenkins, R.J. / Meagher, J.L. / Stuckey, J.A. / Dotson, G.D. | ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2014 Journal: J.Biol.Chem. / Year: 2014Title: Structural Basis for the Recognition of Peptide RJPXD33 by Acyltransferases in Lipid A Biosynthesis. Authors: Jenkins, R.J. / Heslip, K.A. / Meagher, J.L. / Stuckey, J.A. / Dotson, G.D. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4j09.cif.gz 4j09.cif.gz | 117.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4j09.ent.gz pdb4j09.ent.gz | 91.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4j09.json.gz 4j09.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/j0/4j09 https://data.pdbj.org/pub/pdb/validation_reports/j0/4j09 ftp://data.pdbj.org/pub/pdb/validation_reports/j0/4j09 ftp://data.pdbj.org/pub/pdb/validation_reports/j0/4j09 | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 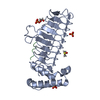
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 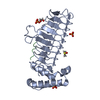
| ||||||||
| Unit cell |
|
- Components
Components
-Protein / Protein/peptide , 2 types, 2 molecules AB
| #1: Protein | Mass: 28117.018 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Escherichia coli (E. coli) / Strain: K-12 / Gene: b0181, JW0176, lpxA / Plasmid: pET24a / Production host: Escherichia coli (E. coli) / Strain: K-12 / Gene: b0181, JW0176, lpxA / Plasmid: pET24a / Production host:   Escherichia coli (E. coli) / Strain (production host): BL21(AI) Escherichia coli (E. coli) / Strain (production host): BL21(AI)References: UniProt: P0A722, acyl-[acyl-carrier-protein]-UDP-N-acetylglucosamine O-acyltransferase |
|---|---|
| #2: Protein/peptide | Mass: 753.907 Da / Num. of mol.: 1 / Source method: obtained synthetically / Details: peptide synthesis / Source: (synth.)   Escherichia coli (E. coli) / References: UniProt: P39352 Escherichia coli (E. coli) / References: UniProt: P39352 |
-Non-polymers , 4 types, 209 molecules 






| #3: Chemical |  Phosphate Phosphate#4: Chemical | ChemComp-DMS / |  Dimethyl sulfoxide Dimethyl sulfoxide#5: Chemical |  Ethylene glycol Ethylene glycol#6: Water | ChemComp-HOH / |  Water Water |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.59 Å3/Da / Density % sol: 52.43 % |
|---|---|
Crystal grow | Temperature: 293.15 K / Method: vapor diffusion / pH: 6.5 Details: 1.4 M Potassium Phosphate, 33% DMSO, pH 6.5, VAPOR DIFFUSION, temperature 293.15K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 21-ID-F / Wavelength: 0.97872 Å / Beamline: 21-ID-F / Wavelength: 0.97872 Å |
| Detector | Type: Marmosaic 300 / Detector: CCD / Date: Apr 26, 2011 / Details: mirrors |
| Radiation | Monochromator: Diamond (111) / Protocol: SINGLE WAVELENGTH / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 0.97872 Å / Relative weight: 1 : 0.97872 Å / Relative weight: 1 |
| Reflection | Resolution: 1.9→48.2 Å / Num. all: 23834 / Num. obs: 23751 / % possible obs: 99.65 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Biso Wilson estimate: 27.87 Å2 |
-Phasing
Phasing | Method:  molecular replacement molecular replacement |
|---|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT / Resolution: 1.9→21.46 Å / Cor.coef. Fo:Fc: 0.9588 / Cor.coef. Fo:Fc free: 0.9565 / Occupancy max: 1 / Occupancy min: 1 / SU R Cruickshank DPI: 0.131 / Cross valid method: THROUGHOUT / σ(F): 0 / Stereochemistry target values: Engh & Huber MOLECULAR REPLACEMENT / Resolution: 1.9→21.46 Å / Cor.coef. Fo:Fc: 0.9588 / Cor.coef. Fo:Fc free: 0.9565 / Occupancy max: 1 / Occupancy min: 1 / SU R Cruickshank DPI: 0.131 / Cross valid method: THROUGHOUT / σ(F): 0 / Stereochemistry target values: Engh & Huber
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 30.46 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.9→21.46 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.9→1.98 Å / Total num. of bins used: 12
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj







