+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4icw | ||||||
|---|---|---|---|---|---|---|---|
| Title | N-terminal C2 domain of human CEP120 | ||||||
 Components Components | Centrosomal protein of 120 kDa Centrosome Centrosome | ||||||
 Keywords Keywords |  TRANSPORT PROTEIN / TRANSPORT PROTEIN /  C2 domain / C2 domain /  lipid binding lipid binding | ||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of centriole elongation / positive regulation of establishment of protein localization / positive regulation of centrosome duplication / interkinetic nuclear migration / astral microtubule organization / positive regulation of cilium assembly /  centrosome cycle / centrosome cycle /  centriole / centriole /  neurogenesis / cerebral cortex development ...positive regulation of centriole elongation / positive regulation of establishment of protein localization / positive regulation of centrosome duplication / interkinetic nuclear migration / astral microtubule organization / positive regulation of cilium assembly / neurogenesis / cerebral cortex development ...positive regulation of centriole elongation / positive regulation of establishment of protein localization / positive regulation of centrosome duplication / interkinetic nuclear migration / astral microtubule organization / positive regulation of cilium assembly /  centrosome cycle / centrosome cycle /  centriole / centriole /  neurogenesis / cerebral cortex development / neurogenesis / cerebral cortex development /  centrosome / centrosome /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.2 Å MOLECULAR REPLACEMENT / Resolution: 2.2 Å | ||||||
 Authors Authors | Janowski, R. / Guarin, N. / Coll, M. | ||||||
 Citation Citation |  Journal: To be Published Journal: To be PublishedTitle: N-terminal C2 domain tandem of human CEP120 shows lipid binding properties Authors: Janowski, R. / Guarin, N. / Speroni, S. / Serrano, L. / Coll, M. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4icw.cif.gz 4icw.cif.gz | 70.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4icw.ent.gz pdb4icw.ent.gz | 53.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4icw.json.gz 4icw.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ic/4icw https://data.pdbj.org/pub/pdb/validation_reports/ic/4icw ftp://data.pdbj.org/pub/pdb/validation_reports/ic/4icw ftp://data.pdbj.org/pub/pdb/validation_reports/ic/4icw | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein |  Centrosome / Cep120 / Coiled-coil domain-containing protein 100 Centrosome / Cep120 / Coiled-coil domain-containing protein 100Mass: 19407.268 Da / Num. of mol.: 1 / Fragment: C2 domain, UNP residues 1-151 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: CEP120, CCDC100 / Production host: Homo sapiens (human) / Gene: CEP120, CCDC100 / Production host:   Escherichia coli (E. coli) / Strain (production host): B834(DE3) / References: UniProt: Q8N960 Escherichia coli (E. coli) / Strain (production host): B834(DE3) / References: UniProt: Q8N960 |
|---|---|
| #2: Water | ChemComp-HOH /  Water Water |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
Crystal grow | Temperature: 292 K / Method: vapor diffusion, hanging drop / pH: 9.5 Details: 25% (v/w) PEG 3000 , pH 9.5, VAPOR DIFFUSION, HANGING DROP, temperature 292K |
|---|
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: ID29 / Wavelength: 0.9726 Å / Beamline: ID29 / Wavelength: 0.9726 Å |
| Detector | Type: PSI PILATUS 6M / Detector: PIXEL / Date: Apr 20, 2011 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 0.9726 Å / Relative weight: 1 : 0.9726 Å / Relative weight: 1 |
| Reflection | Resolution: 2.2→30 Å / Num. all: 6902 / Num. obs: 6902 / % possible obs: 98.2 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Redundancy: 3.4 % / Biso Wilson estimate: 58.8 Å2 / Rmerge(I) obs: 0.029 / Net I/σ(I): 25.8 |
| Reflection shell | Resolution: 2.2→2.26 Å / Rmerge(I) obs: 0.522 / Mean I/σ(I) obs: 3.06 / % possible all: 98.6 |
- Processing
Processing
| Software | Name: REFMAC / Version: 5.5.0102 / Classification: refinement | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT / Resolution: 2.2→30 Å / Cor.coef. Fo:Fc: 0.956 / Cor.coef. Fo:Fc free: 0.953 / SU B: 18.797 / SU ML: 0.206 / Cross valid method: THROUGHOUT / σ(F): 0 / σ(I): 0 / ESU R: 0.365 / ESU R Free: 0.219 / Stereochemistry target values: MAXIMUM LIKELIHOOD MOLECULAR REPLACEMENT / Resolution: 2.2→30 Å / Cor.coef. Fo:Fc: 0.956 / Cor.coef. Fo:Fc free: 0.953 / SU B: 18.797 / SU ML: 0.206 / Cross valid method: THROUGHOUT / σ(F): 0 / σ(I): 0 / ESU R: 0.365 / ESU R Free: 0.219 / Stereochemistry target values: MAXIMUM LIKELIHOOD
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.4 Å / Solvent model: BABINET MODEL WITH MASK | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 84.758 Å2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.2→30 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.2→2.318 Å / Total num. of bins used: 10
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Origin x: 11.3244 Å / Origin y: -8.0511 Å / Origin z: -13.9524 Å
|
 Movie
Movie Controller
Controller









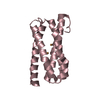




 PDBj
PDBj



