[English] 日本語
 Yorodumi
Yorodumi- PDB-4cxg: Regulation of the mammalian elongation cycle by 40S subunit rolli... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4cxg | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Regulation of the mammalian elongation cycle by 40S subunit rolling: a eukaryotic-specific ribosome rearrangement | ||||||||||||
 Components Components |
| ||||||||||||
 Keywords Keywords |  TRANSLATION / MAMMALIAN 80S RIBOSOME / ELONGATION CYCLE / TRNA SELECTION / EUKARYOTIC TERNARY COMPLEX TRANSLATION / MAMMALIAN 80S RIBOSOME / ELONGATION CYCLE / TRNA SELECTION / EUKARYOTIC TERNARY COMPLEX | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationFormation of the ternary complex, and subsequently, the 43S complex / Ribosomal scanning and start codon recognition / Translation initiation complex formation / SARS-CoV-1 modulates host translation machinery /  Protein hydroxylation / Peptide chain elongation / Selenocysteine synthesis / Formation of a pool of free 40S subunits / Eukaryotic Translation Termination / Response of EIF2AK4 (GCN2) to amino acid deficiency ...Formation of the ternary complex, and subsequently, the 43S complex / Ribosomal scanning and start codon recognition / Translation initiation complex formation / SARS-CoV-1 modulates host translation machinery / Protein hydroxylation / Peptide chain elongation / Selenocysteine synthesis / Formation of a pool of free 40S subunits / Eukaryotic Translation Termination / Response of EIF2AK4 (GCN2) to amino acid deficiency ...Formation of the ternary complex, and subsequently, the 43S complex / Ribosomal scanning and start codon recognition / Translation initiation complex formation / SARS-CoV-1 modulates host translation machinery /  Protein hydroxylation / Peptide chain elongation / Selenocysteine synthesis / Formation of a pool of free 40S subunits / Eukaryotic Translation Termination / Response of EIF2AK4 (GCN2) to amino acid deficiency / SRP-dependent cotranslational protein targeting to membrane / Viral mRNA Translation / Nonsense Mediated Decay (NMD) independent of the Exon Junction Complex (EJC) / GTP hydrolysis and joining of the 60S ribosomal subunit / L13a-mediated translational silencing of Ceruloplasmin expression / Major pathway of rRNA processing in the nucleolus and cytosol / Protein hydroxylation / Peptide chain elongation / Selenocysteine synthesis / Formation of a pool of free 40S subunits / Eukaryotic Translation Termination / Response of EIF2AK4 (GCN2) to amino acid deficiency / SRP-dependent cotranslational protein targeting to membrane / Viral mRNA Translation / Nonsense Mediated Decay (NMD) independent of the Exon Junction Complex (EJC) / GTP hydrolysis and joining of the 60S ribosomal subunit / L13a-mediated translational silencing of Ceruloplasmin expression / Major pathway of rRNA processing in the nucleolus and cytosol /  rough endoplasmic reticulum / Nonsense Mediated Decay (NMD) enhanced by the Exon Junction Complex (EJC) / rough endoplasmic reticulum / Nonsense Mediated Decay (NMD) enhanced by the Exon Junction Complex (EJC) /  translation elongation factor activity / translation elongation factor activity /  stress granule assembly / small-subunit processome / cytosolic ribosome / maintenance of translational fidelity / stress granule assembly / small-subunit processome / cytosolic ribosome / maintenance of translational fidelity /  ribosomal small subunit biogenesis / Regulation of expression of SLITs and ROBOs / cytosolic small ribosomal subunit / cytoplasmic translation / SARS-CoV-2 modulates host translation machinery / ribosomal small subunit biogenesis / Regulation of expression of SLITs and ROBOs / cytosolic small ribosomal subunit / cytoplasmic translation / SARS-CoV-2 modulates host translation machinery /  ribosome / structural constituent of ribosome / ribosome / structural constituent of ribosome /  translation / translation /  GTPase activity / GTPase activity /  synapse / GTP binding / synapse / GTP binding /  nucleolus / nucleolus /  endoplasmic reticulum / endoplasmic reticulum /  RNA binding / RNA binding /  nucleoplasm / nucleoplasm /  membrane / membrane /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | ||||||||||||
| Biological species |   ORYCTOLAGUS CUNICULUS (rabbit) ORYCTOLAGUS CUNICULUS (rabbit)  ESCHERICHIA COLI (E. coli) ESCHERICHIA COLI (E. coli) | ||||||||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 8.7 Å cryo EM / Resolution: 8.7 Å | ||||||||||||
 Authors Authors | Budkevich, T.V. / Giesebrecht, J. / Behrmann, E. / Loerke, J. / Ramrath, D.J.F. / Mielke, T. / Ismer, J. / Hildebrand, P. / Tung, C.-S. / Nierhaus, K.H. ...Budkevich, T.V. / Giesebrecht, J. / Behrmann, E. / Loerke, J. / Ramrath, D.J.F. / Mielke, T. / Ismer, J. / Hildebrand, P. / Tung, C.-S. / Nierhaus, K.H. / Sanbonmatsu, K.Y. / Spahn, C.M.T. | ||||||||||||
 Citation Citation |  Journal: Cell / Year: 2014 Journal: Cell / Year: 2014Title: Regulation of the mammalian elongation cycle by subunit rolling: a eukaryotic-specific ribosome rearrangement. Authors: Tatyana V Budkevich / Jan Giesebrecht / Elmar Behrmann / Justus Loerke / David J F Ramrath / Thorsten Mielke / Jochen Ismer / Peter W Hildebrand / Chang-Shung Tung / Knud H Nierhaus / ...Authors: Tatyana V Budkevich / Jan Giesebrecht / Elmar Behrmann / Justus Loerke / David J F Ramrath / Thorsten Mielke / Jochen Ismer / Peter W Hildebrand / Chang-Shung Tung / Knud H Nierhaus / Karissa Y Sanbonmatsu / Christian M T Spahn /    Abstract: The extent to which bacterial ribosomes and the significantly larger eukaryotic ribosomes share the same mechanisms of ribosomal elongation is unknown. Here, we present subnanometer resolution ...The extent to which bacterial ribosomes and the significantly larger eukaryotic ribosomes share the same mechanisms of ribosomal elongation is unknown. Here, we present subnanometer resolution cryoelectron microscopy maps of the mammalian 80S ribosome in the posttranslocational state and in complex with the eukaryotic eEF1A⋅Val-tRNA⋅GMPPNP ternary complex, revealing significant differences in the elongation mechanism between bacteria and mammals. Surprisingly, and in contrast to bacterial ribosomes, a rotation of the small subunit around its long axis and orthogonal to the well-known intersubunit rotation distinguishes the posttranslocational state from the classical pretranslocational state ribosome. We term this motion "subunit rolling." Correspondingly, a mammalian decoding complex visualized in substates before and after codon recognition reveals structural distinctions from the bacterial system. These findings suggest how codon recognition leads to GTPase activation in the mammalian system and demonstrate that in mammalia subunit rolling occurs during tRNA selection. | ||||||||||||
| History |
| ||||||||||||
| Remark 700 | SHEET DETERMINATION METHOD: DSSP THE SHEETS PRESENTED AS "XA" IN EACH CHAIN ON SHEET RECORDS BELOW ... SHEET DETERMINATION METHOD: DSSP THE SHEETS PRESENTED AS "XA" IN EACH CHAIN ON SHEET RECORDS BELOW IS ACTUALLY AN 5-STRANDED BARREL THIS IS REPRESENTED BY A 6-STRANDED SHEET IN WHICH THE FIRST AND LAST STRANDS ARE IDENTICAL. |
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4cxg.cif.gz 4cxg.cif.gz | 298.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4cxg.ent.gz pdb4cxg.ent.gz | 234.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4cxg.json.gz 4cxg.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/cx/4cxg https://data.pdbj.org/pub/pdb/validation_reports/cx/4cxg ftp://data.pdbj.org/pub/pdb/validation_reports/cx/4cxg ftp://data.pdbj.org/pub/pdb/validation_reports/cx/4cxg | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  2623MC  2620C  2621C  2622C  2624C  4cxhC  4ujeC C: citing same article ( M: map data used to model this data |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-RNA chain , 7 types, 7 molecules 12Yabcx
| #1: RNA chain | Mass: 43635.969 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   ORYCTOLAGUS CUNICULUS (rabbit) ORYCTOLAGUS CUNICULUS (rabbit) |
|---|---|
| #2: RNA chain | Mass: 15974.376 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   ORYCTOLAGUS CUNICULUS (rabbit) ORYCTOLAGUS CUNICULUS (rabbit) |
| #5: RNA chain |  Mass: 24686.006 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   ORYCTOLAGUS CUNICULUS (rabbit) ORYCTOLAGUS CUNICULUS (rabbit) |
| #6: RNA chain | Mass: 15424.328 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   ORYCTOLAGUS CUNICULUS (rabbit) ORYCTOLAGUS CUNICULUS (rabbit) |
| #7: RNA chain | Mass: 5445.253 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   ORYCTOLAGUS CUNICULUS (rabbit) ORYCTOLAGUS CUNICULUS (rabbit) |
| #8: RNA chain | Mass: 6140.738 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   ORYCTOLAGUS CUNICULUS (rabbit) ORYCTOLAGUS CUNICULUS (rabbit) |
| #9: RNA chain |  Mass: 8527.686 Da / Num. of mol.: 1 / Source method: obtained synthetically Details: FOR MODELING MRNA (CHAIN 1, PDB ID CODE 3I8G) WAS USED Source: (synth.)   ESCHERICHIA COLI (E. coli) ESCHERICHIA COLI (E. coli) |
-Protein , 2 types, 2 molecules AX
| #3: Protein | Mass: 48734.527 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   ORYCTOLAGUS CUNICULUS (rabbit) / References: UniProt: Q9YAV0*PLUS ORYCTOLAGUS CUNICULUS (rabbit) / References: UniProt: Q9YAV0*PLUS |
|---|---|
| #4: Protein |  / 40S RIBOSOMAL PROTEIN S23 / 40S RIBOSOMAL PROTEIN S23Mass: 15844.666 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   ORYCTOLAGUS CUNICULUS (rabbit) / References: UniProt: P62266*PLUS ORYCTOLAGUS CUNICULUS (rabbit) / References: UniProt: P62266*PLUS |
-Non-polymers , 1 types, 1 molecules 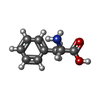
| #10: Chemical | ChemComp-PHE /  Phenylalanine Phenylalanine |
|---|
-Details
| Sequence details | AUTHORS HAVE USED SEQUENCE FOR MODEL BUILDING, WITH FOLLOWING PDB-GENBANK OR UNIPROT RESIDUE ...AUTHORS HAVE USED SEQUENCE FOR MODEL BUILDING, WITH FOLLOWING PDB-GENBANK OR UNIPROT RESIDUE MAPPING: CHAIN A 1- 437 UNP Q9YAV0 1- 437 CHAIN X 1- 143 UNP P62266 1- 143 CHAIN Y 1- 76 GB X00655 1- 76 CHAIN 1 1701-1835 GB X03205 1701-1835 CHAIN 2 4359-4408 GB M11167 4359-4408 CHAIN a 458- 505 GB X03205 458- 505 CHAIN b 151- 167 GB X03205 151- 167 CHAIN c 4553-4571 GB M11167 4553-4571 |
|---|
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: INITIAL CODON SAMPLING STATE OF 80S MAMMALIAN RIBOSOME Type: RIBOSOME |
|---|---|
| Buffer solution | Name: 20 MM HEPES-KOH, 5 MM MGCL2, 100 MM NH4CL, 6 MM MERCAPROETHANOL, 0.8 MM SPERMIDINE, 0.6 MM SPERMINE pH: 7.5 Details: 20 MM HEPES-KOH, 5 MM MGCL2, 100 MM NH4CL, 6 MM MERCAPROETHANOL, 0.8 MM SPERMIDINE, 0.6 MM SPERMINE |
| Specimen | Conc.: 1.3 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES : YES |
| Specimen support | Details: HOLEY CARBON |
Vitrification | Instrument: FEI VITROBOT MARK II / Cryogen name: ETHANE Details: VITRIFICATION 1 -- CRYOGEN ETHANE, HUMIDITY 80, INSTRUMENT - FEI VITROBOT MARK II, METHOD - BLOT FOR 2 SEC BEFORE PLUNGING |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Tecnai F30 / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TECNAI F30 / Date: Sep 22, 2008 / Details: MINIMAL DOSE SYSTEM |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Nominal magnification: 39000 X / Calibrated magnification: 65520 X / Nominal defocus max: 4000 nm / Nominal defocus min: 2000 nm / Cs Bright-field microscopy / Nominal magnification: 39000 X / Calibrated magnification: 65520 X / Nominal defocus max: 4000 nm / Nominal defocus min: 2000 nm / Cs : 2 mm : 2 mm |
| Image recording | Electron dose: 20 e/Å2 / Film or detector model: KODAK SO-163 FILM |
| Image scans | Num. digital images: 230 |
- Processing
Processing
| EM software |
| |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
CTF correction | Details: DEFOCUS GROUP | |||||||||||||||
| Symmetry | Point symmetry : C1 (asymmetric) : C1 (asymmetric) | |||||||||||||||
3D reconstruction | Method: MULTI-REFERENCE TEMPLATE MATCHING / Resolution: 8.7 Å / Num. of particles: 79705 / Nominal pixel size: 1.26 Å / Actual pixel size: 1.26 Å Magnification calibration: CROSS- -CORRELATION WITH KNOWN STRUCTURE Details: SUBMISSION BASED ON EXPERIMENTAL DATA FROM EMDB EMD-2623. (DEPOSITION ID: 12411). Symmetry type: POINT | |||||||||||||||
| Atomic model building | Protocol: RIGID BODY FIT / Space: REAL / Target criteria: Cross-correlation coefficient / Details: METHOD--RIGID BODY | |||||||||||||||
| Refinement | Highest resolution: 8.7 Å | |||||||||||||||
| Refinement step | Cycle: LAST / Highest resolution: 8.7 Å
|
 Movie
Movie Controller
Controller










 PDBj
PDBj





































