Entry Database : PDB / ID : 4axbTitle Crystal structure of soman-aged human butyrylcholinesterase in complex with 2-PAM CHOLINESTERASE Keywords / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species HOMO SAPIENS (human)Method / / / Resolution : 2.4 Å Authors Wandhammer, M. / de Koning, M. / Noort, D. / Goeldner, M. / Nachon, F. Journal : Chem.Biol.Interact / Year : 2013Title : A Step Toward the Reactivation of Aged Cholinesterases -Crystal Structure of Ligands Binding to Aged Human ButyrylcholinesteraseAuthors : Wandhammer, M. / De Koning, M. / Van Grol, M. / Loiodice, M. / Saurel, L. / Noort, D. / Goeldner, M. / Nachon, F. History Deposition Jun 12, 2012 Deposition site / Processing site Revision 1.0 Aug 29, 2012 Provider / Type Revision 1.1 Jan 16, 2013 Group Revision 1.2 Apr 17, 2013 Group Revision 1.3 Sep 25, 2013 Group / Other / Source and taxonomyRevision 2.0 Jul 29, 2020 Group Advisory / Atomic model ... Advisory / Atomic model / Data collection / Derived calculations / Other / Structure summary Category atom_site / atom_site_anisotrop ... atom_site / atom_site_anisotrop / chem_comp / database_PDB_caveat / entity / pdbx_branch_scheme / pdbx_chem_comp_identifier / pdbx_database_status / pdbx_entity_branch / pdbx_entity_branch_descriptor / pdbx_entity_branch_link / pdbx_entity_branch_list / pdbx_entity_nonpoly / pdbx_nonpoly_scheme / pdbx_struct_assembly_gen / pdbx_struct_conn_angle / pdbx_validate_chiral / struct_asym / struct_conn / struct_site / struct_site_gen Item _atom_site.B_iso_or_equiv / _atom_site.Cartn_x ... _atom_site.B_iso_or_equiv / _atom_site.Cartn_x / _atom_site.Cartn_y / _atom_site.Cartn_z / _atom_site.auth_asym_id / _atom_site.auth_atom_id / _atom_site.auth_comp_id / _atom_site.auth_seq_id / _atom_site.label_asym_id / _atom_site.label_atom_id / _atom_site.label_comp_id / _atom_site.label_entity_id / _atom_site.type_symbol / _atom_site_anisotrop.pdbx_label_asym_id / _chem_comp.name / _chem_comp.type / _entity.formula_weight / _entity.pdbx_description / _entity.pdbx_number_of_molecules / _entity.src_method / _entity.type / _pdbx_database_status.status_code_sf / _pdbx_struct_assembly_gen.asym_id_list / _pdbx_struct_conn_angle.ptnr1_auth_comp_id / _pdbx_struct_conn_angle.ptnr1_auth_seq_id / _pdbx_struct_conn_angle.ptnr1_label_asym_id / _pdbx_struct_conn_angle.ptnr1_label_atom_id / _pdbx_struct_conn_angle.ptnr1_label_comp_id / _pdbx_struct_conn_angle.ptnr1_label_seq_id / _pdbx_struct_conn_angle.ptnr2_label_asym_id / _pdbx_struct_conn_angle.ptnr3_auth_comp_id / _pdbx_struct_conn_angle.ptnr3_auth_seq_id / _pdbx_struct_conn_angle.ptnr3_label_asym_id / _pdbx_struct_conn_angle.ptnr3_label_atom_id / _pdbx_struct_conn_angle.ptnr3_label_comp_id / _pdbx_struct_conn_angle.ptnr3_label_seq_id / _pdbx_struct_conn_angle.value / _pdbx_validate_chiral.auth_asym_id / _pdbx_validate_chiral.auth_seq_id / _struct_conn.conn_type_id / _struct_conn.id / _struct_conn.pdbx_dist_value / _struct_conn.pdbx_leaving_atom_flag / _struct_conn.pdbx_role / _struct_conn.ptnr1_auth_asym_id / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr2_auth_asym_id / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id / _struct_conn.ptnr2_label_seq_id Description / Provider / Type Revision 2.1 Dec 20, 2023 Group Data collection / Database references ... Data collection / Database references / Refinement description / Structure summary Category chem_comp / chem_comp_atom ... chem_comp / chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model Item / _database_2.pdbx_DOI / _database_2.pdbx_database_accession
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components
 Keywords
Keywords HYDROLASE /
HYDROLASE /  AGING
AGING Function and homology information
Function and homology information cholinesterase / cocaine metabolic process / neuroblast differentiation / Neurotransmitter clearance / choline metabolic process /
cholinesterase / cocaine metabolic process / neuroblast differentiation / Neurotransmitter clearance / choline metabolic process /  choline binding / response to folic acid / response to alkaloid / acetylcholine catabolic process / negative regulation of synaptic transmission ...
choline binding / response to folic acid / response to alkaloid / acetylcholine catabolic process / negative regulation of synaptic transmission ... cholinesterase / cocaine metabolic process / neuroblast differentiation / Neurotransmitter clearance / choline metabolic process /
cholinesterase / cocaine metabolic process / neuroblast differentiation / Neurotransmitter clearance / choline metabolic process /  choline binding / response to folic acid / response to alkaloid / acetylcholine catabolic process / negative regulation of synaptic transmission /
choline binding / response to folic acid / response to alkaloid / acetylcholine catabolic process / negative regulation of synaptic transmission /  cholinesterase activity / peptide hormone processing /
cholinesterase activity / peptide hormone processing /  acetylcholinesterase activity / hydrolase activity, acting on ester bonds / nuclear envelope lumen / Aspirin ADME / Synthesis of PC / Synthesis, secretion, and deacylation of Ghrelin /
acetylcholinesterase activity / hydrolase activity, acting on ester bonds / nuclear envelope lumen / Aspirin ADME / Synthesis of PC / Synthesis, secretion, and deacylation of Ghrelin /  catalytic activity / response to glucocorticoid / xenobiotic metabolic process /
catalytic activity / response to glucocorticoid / xenobiotic metabolic process /  learning /
learning /  amyloid-beta binding / blood microparticle / negative regulation of cell population proliferation /
amyloid-beta binding / blood microparticle / negative regulation of cell population proliferation /  endoplasmic reticulum lumen /
endoplasmic reticulum lumen /  enzyme binding /
enzyme binding /  extracellular space / extracellular region / identical protein binding /
extracellular space / extracellular region / identical protein binding /  plasma membrane
plasma membrane
 HOMO SAPIENS (human)
HOMO SAPIENS (human) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.4 Å
MOLECULAR REPLACEMENT / Resolution: 2.4 Å  Authors
Authors Citation
Citation Journal: Chem.Biol.Interact / Year: 2013
Journal: Chem.Biol.Interact / Year: 2013 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 4axb.cif.gz
4axb.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb4axb.ent.gz
pdb4axb.ent.gz PDB format
PDB format 4axb.json.gz
4axb.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/ax/4axb
https://data.pdbj.org/pub/pdb/validation_reports/ax/4axb ftp://data.pdbj.org/pub/pdb/validation_reports/ax/4axb
ftp://data.pdbj.org/pub/pdb/validation_reports/ax/4axb


 Links
Links Assembly
Assembly

 Components
Components / ACYLCHOLINE ACYLHYDROLASE / BUTYRYLCHOLINE ESTERASE / CHOLINE ESTERASE II / PSEUDOCHOLINESTERASE
/ ACYLCHOLINE ACYLHYDROLASE / BUTYRYLCHOLINE ESTERASE / CHOLINE ESTERASE II / PSEUDOCHOLINESTERASE
 HOMO SAPIENS (human) / Plasmid: PGS / Cell line (production host): CHO K1 / Production host:
HOMO SAPIENS (human) / Plasmid: PGS / Cell line (production host): CHO K1 / Production host: 
 CRICETULUS GRISEUS (Chinese hamster) / References: UniProt: P06276,
CRICETULUS GRISEUS (Chinese hamster) / References: UniProt: P06276,  cholinesterase
cholinesterase
 / Mass: 570.542 Da / Num. of mol.: 2
/ Mass: 570.542 Da / Num. of mol.: 2 N-Acetylglucosamine
N-Acetylglucosamine



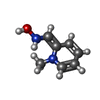






 Sulfate
Sulfate Glycerol
Glycerol Chloride
Chloride Water
Water X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation
 SYNCHROTRON / Site:
SYNCHROTRON / Site:  ESRF
ESRF  / Beamline: ID23-2 / Wavelength: 0.8726
/ Beamline: ID23-2 / Wavelength: 0.8726  : 0.8726 Å / Relative weight: 1
: 0.8726 Å / Relative weight: 1  Processing
Processing :
:  MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller



















 PDBj
PDBj













