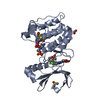
Entry Database : PDB / ID : 3kudTitle Complex of Ras-GDP with RafRBD(A85K) GTPase HRas RAF proto-oncogene serine/threonine-protein kinase Keywords / / / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Homo sapiens (human)Method / / / Resolution : 2.15 Å Authors Filchtinski, D. / Sharabi, O. / Rueppel, A. / Vetter, I.R. / Herrmann, C. / Shifman, J.M. Journal : J.Mol.Biol. / Year : 2010Title : What makes Ras an efficient molecular switch: a computational, biophysical, and structural study of Ras-GDP interactions with mutants of Raf.Authors : Filchtinski, D. / Sharabi, O. / Ruppel, A. / Vetter, I.R. / Herrmann, C. / Shifman, J.M. History Deposition Nov 27, 2009 Deposition site / Processing site Revision 1.0 Mar 23, 2010 Provider / Type Revision 1.1 Jul 13, 2011 Group / Version format complianceRevision 1.2 Nov 1, 2017 Group / Category / Item Revision 1.3 Oct 13, 2021 Group / Derived calculations / Category / struct_ref_seq_dif / struct_siteItem _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _struct_ref_seq_dif.details / _struct_site.pdbx_auth_asym_id / _struct_site.pdbx_auth_comp_id / _struct_site.pdbx_auth_seq_id Revision 1.4 Sep 6, 2023 Group / Refinement descriptionCategory / chem_comp_bond / pdbx_initial_refinement_model
Show all Show less
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Proto-oncogene /
Proto-oncogene /  Transferase / GTP BINDING PROTEIN-TRANSFERASE complex
Transferase / GTP BINDING PROTEIN-TRANSFERASE complex Function and homology information
Function and homology information death-inducing signaling complex assembly / intermediate filament cytoskeleton organization / type B pancreatic cell proliferation / regulation of Rho protein signal transduction / SHOC2 M1731 mutant abolishes MRAS complex function / Gain-of-function MRAS complexes activate RAF signaling / Rap1 signalling /
death-inducing signaling complex assembly / intermediate filament cytoskeleton organization / type B pancreatic cell proliferation / regulation of Rho protein signal transduction / SHOC2 M1731 mutant abolishes MRAS complex function / Gain-of-function MRAS complexes activate RAF signaling / Rap1 signalling /  regulation of cell motility /
regulation of cell motility /  GTPase complex / oncogene-induced cell senescence ...
GTPase complex / oncogene-induced cell senescence ... death-inducing signaling complex assembly / intermediate filament cytoskeleton organization / type B pancreatic cell proliferation / regulation of Rho protein signal transduction / SHOC2 M1731 mutant abolishes MRAS complex function / Gain-of-function MRAS complexes activate RAF signaling / Rap1 signalling /
death-inducing signaling complex assembly / intermediate filament cytoskeleton organization / type B pancreatic cell proliferation / regulation of Rho protein signal transduction / SHOC2 M1731 mutant abolishes MRAS complex function / Gain-of-function MRAS complexes activate RAF signaling / Rap1 signalling /  regulation of cell motility /
regulation of cell motility /  GTPase complex / oncogene-induced cell senescence / insulin secretion involved in cellular response to glucose stimulus / positive regulation of ruffle assembly / regulation of neurotransmitter receptor localization to postsynaptic specialization membrane / negative regulation of GTPase activity / positive regulation of miRNA metabolic process / Negative feedback regulation of MAPK pathway / T-helper 1 type immune response / IFNG signaling activates MAPKs / GP1b-IX-V activation signalling / positive regulation of wound healing / ERBB2-ERBB3 signaling pathway /
GTPase complex / oncogene-induced cell senescence / insulin secretion involved in cellular response to glucose stimulus / positive regulation of ruffle assembly / regulation of neurotransmitter receptor localization to postsynaptic specialization membrane / negative regulation of GTPase activity / positive regulation of miRNA metabolic process / Negative feedback regulation of MAPK pathway / T-helper 1 type immune response / IFNG signaling activates MAPKs / GP1b-IX-V activation signalling / positive regulation of wound healing / ERBB2-ERBB3 signaling pathway /  regulation of cell differentiation / defense response to protozoan / face development /
regulation of cell differentiation / defense response to protozoan / face development /  pseudopodium / somatic stem cell population maintenance / Signaling by RAS GAP mutants / Signaling by RAS GTPase mutants / Activation of RAS in B cells / neurotrophin TRK receptor signaling pathway / thyroid gland development / RAS signaling downstream of NF1 loss-of-function variants / positive regulation of protein targeting to membrane / SOS-mediated signalling / Activated NTRK3 signals through RAS / Activated NTRK2 signals through RAS / extrinsic apoptotic signaling pathway via death domain receptors /
pseudopodium / somatic stem cell population maintenance / Signaling by RAS GAP mutants / Signaling by RAS GTPase mutants / Activation of RAS in B cells / neurotrophin TRK receptor signaling pathway / thyroid gland development / RAS signaling downstream of NF1 loss-of-function variants / positive regulation of protein targeting to membrane / SOS-mediated signalling / Activated NTRK3 signals through RAS / Activated NTRK2 signals through RAS / extrinsic apoptotic signaling pathway via death domain receptors /  MAP kinase kinase kinase activity / SHC1 events in ERBB4 signaling / Signalling to RAS / SHC-related events triggered by IGF1R / Activated NTRK2 signals through FRS2 and FRS3 / negative regulation of protein-containing complex assembly / adipose tissue development / Estrogen-stimulated signaling through PRKCZ / SHC-mediated cascade:FGFR3 / MET activates RAS signaling / : / PTK6 Regulates RHO GTPases, RAS GTPase and MAP kinases / Schwann cell development / Signaling by PDGFRA transmembrane, juxtamembrane and kinase domain mutants / Signaling by PDGFRA extracellular domain mutants / SHC-mediated cascade:FGFR2 / type II interferon-mediated signaling pathway / SHC-mediated cascade:FGFR4 / Signaling by FGFR4 in disease / SHC-mediated cascade:FGFR1 / Erythropoietin activates RAS / protein-membrane adaptor activity / negative regulation of extrinsic apoptotic signaling pathway via death domain receptors / FRS-mediated FGFR3 signaling / Signaling by FLT3 ITD and TKD mutants / FRS-mediated FGFR2 signaling / FRS-mediated FGFR4 signaling / Signaling by FGFR3 in disease / p38MAPK events / FRS-mediated FGFR1 signaling / Tie2 Signaling / Signaling by FGFR2 in disease / GRB2 events in EGFR signaling / EPHB-mediated forward signaling / SHC1 events in EGFR signaling / activation of adenylate cyclase activity / EGFR Transactivation by Gastrin / response to muscle stretch /
MAP kinase kinase kinase activity / SHC1 events in ERBB4 signaling / Signalling to RAS / SHC-related events triggered by IGF1R / Activated NTRK2 signals through FRS2 and FRS3 / negative regulation of protein-containing complex assembly / adipose tissue development / Estrogen-stimulated signaling through PRKCZ / SHC-mediated cascade:FGFR3 / MET activates RAS signaling / : / PTK6 Regulates RHO GTPases, RAS GTPase and MAP kinases / Schwann cell development / Signaling by PDGFRA transmembrane, juxtamembrane and kinase domain mutants / Signaling by PDGFRA extracellular domain mutants / SHC-mediated cascade:FGFR2 / type II interferon-mediated signaling pathway / SHC-mediated cascade:FGFR4 / Signaling by FGFR4 in disease / SHC-mediated cascade:FGFR1 / Erythropoietin activates RAS / protein-membrane adaptor activity / negative regulation of extrinsic apoptotic signaling pathway via death domain receptors / FRS-mediated FGFR3 signaling / Signaling by FLT3 ITD and TKD mutants / FRS-mediated FGFR2 signaling / FRS-mediated FGFR4 signaling / Signaling by FGFR3 in disease / p38MAPK events / FRS-mediated FGFR1 signaling / Tie2 Signaling / Signaling by FGFR2 in disease / GRB2 events in EGFR signaling / EPHB-mediated forward signaling / SHC1 events in EGFR signaling / activation of adenylate cyclase activity / EGFR Transactivation by Gastrin / response to muscle stretch /  myelination / Signaling by FLT3 fusion proteins / FLT3 Signaling / Ras activation upon Ca2+ influx through NMDA receptor / GRB2 events in ERBB2 signaling / Signaling by FGFR1 in disease / NCAM signaling for neurite out-growth / CD209 (DC-SIGN) signaling / SHC1 events in ERBB2 signaling / Downstream signal transduction / Constitutive Signaling by Overexpressed ERBB2 / Insulin receptor signalling cascade / intrinsic apoptotic signaling pathway /
myelination / Signaling by FLT3 fusion proteins / FLT3 Signaling / Ras activation upon Ca2+ influx through NMDA receptor / GRB2 events in ERBB2 signaling / Signaling by FGFR1 in disease / NCAM signaling for neurite out-growth / CD209 (DC-SIGN) signaling / SHC1 events in ERBB2 signaling / Downstream signal transduction / Constitutive Signaling by Overexpressed ERBB2 / Insulin receptor signalling cascade / intrinsic apoptotic signaling pathway /  small monomeric GTPase / insulin-like growth factor receptor signaling pathway / G protein activity / Signaling by phosphorylated juxtamembrane, extracellular and kinase domain KIT mutants / thymus development / VEGFR2 mediated cell proliferation / positive regulation of epithelial cell proliferation / regulation of actin cytoskeleton organization / FCERI mediated MAPK activation / animal organ morphogenesis / Signaling by ERBB2 TMD/JMD mutants / positive regulation of JNK cascade
small monomeric GTPase / insulin-like growth factor receptor signaling pathway / G protein activity / Signaling by phosphorylated juxtamembrane, extracellular and kinase domain KIT mutants / thymus development / VEGFR2 mediated cell proliferation / positive regulation of epithelial cell proliferation / regulation of actin cytoskeleton organization / FCERI mediated MAPK activation / animal organ morphogenesis / Signaling by ERBB2 TMD/JMD mutants / positive regulation of JNK cascade
 Homo sapiens (human)
Homo sapiens (human) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.15 Å
MOLECULAR REPLACEMENT / Resolution: 2.15 Å  Authors
Authors Citation
Citation Journal: J.Mol.Biol. / Year: 2010
Journal: J.Mol.Biol. / Year: 2010 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 3kud.cif.gz
3kud.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb3kud.ent.gz
pdb3kud.ent.gz PDB format
PDB format 3kud.json.gz
3kud.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/ku/3kud
https://data.pdbj.org/pub/pdb/validation_reports/ku/3kud ftp://data.pdbj.org/pub/pdb/validation_reports/ku/3kud
ftp://data.pdbj.org/pub/pdb/validation_reports/ku/3kud

 Links
Links Assembly
Assembly
 Components
Components HRAS / Transforming protein p21 / p21ras / H-Ras-1 / c-H-ras / Ha-Ras / GTPase HRas / N-terminally processed
HRAS / Transforming protein p21 / p21ras / H-Ras-1 / c-H-ras / Ha-Ras / GTPase HRas / N-terminally processed
 Homo sapiens (human) / Gene: HRAS, HRAS1 / Production host:
Homo sapiens (human) / Gene: HRAS, HRAS1 / Production host: 
 Escherichia coli (E. coli) / References: UniProt: P01112
Escherichia coli (E. coli) / References: UniProt: P01112
 Homo sapiens (human) / Gene: RAF1, RAF / Production host:
Homo sapiens (human) / Gene: RAF1, RAF / Production host: 
 Escherichia coli (E. coli)
Escherichia coli (E. coli) non-specific serine/threonine protein kinase
non-specific serine/threonine protein kinase Guanosine diphosphate
Guanosine diphosphate Water
Water X-RAY DIFFRACTION
X-RAY DIFFRACTION Sample preparation
Sample preparation
 SYNCHROTRON / Site:
SYNCHROTRON / Site:  SLS
SLS  / Beamline: X10SA / Wavelength: 0.97935 Å
/ Beamline: X10SA / Wavelength: 0.97935 Å : 0.97935 Å / Relative weight: 1
: 0.97935 Å / Relative weight: 1  Processing
Processing :
:  MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller












 PDBj
PDBj






















