[English] 日本語
 Yorodumi
Yorodumi- PDB-2qqh: Structure of C8a-MACPF reveals mechanism of membrane attack in co... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2qqh | ||||||
|---|---|---|---|---|---|---|---|
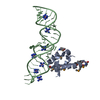
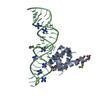
| Title | Structure of C8a-MACPF reveals mechanism of membrane attack in complement immune defense | ||||||
 Components Components | Complement component C8 alpha chain | ||||||
 Keywords Keywords |  IMMUNE SYSTEM / IMMUNE SYSTEM /  MEMBRANE PROTEIN / MEMBRANE PROTEIN /  MACPF / MEMBRANE PERFORATION / Cleavage on pair of basic residues / Complement alternate pathway / Complement pathway / MACPF / MEMBRANE PERFORATION / Cleavage on pair of basic residues / Complement alternate pathway / Complement pathway /  Cytolysis / Cytolysis /  EGF-like domain / EGF-like domain /  Glycoprotein / Glycoprotein /  Immune response / Immune response /  Innate immunity / Innate immunity /  Membrane attack complex / Polymorphism / Membrane attack complex / Polymorphism /  Secreted Secreted | ||||||
| Function / homology |  Function and homology information Function and homology informationTerminal pathway of complement /  membrane attack complex / complement binding / membrane attack complex / complement binding /  complement activation, alternative pathway / complement activation, alternative pathway /  complement activation / complement activation /  complement activation, classical pathway / complement activation, classical pathway /  Regulation of Complement cascade / positive regulation of immune response / blood microparticle / killing of cells of another organism ...Terminal pathway of complement / Regulation of Complement cascade / positive regulation of immune response / blood microparticle / killing of cells of another organism ...Terminal pathway of complement /  membrane attack complex / complement binding / membrane attack complex / complement binding /  complement activation, alternative pathway / complement activation, alternative pathway /  complement activation / complement activation /  complement activation, classical pathway / complement activation, classical pathway /  Regulation of Complement cascade / positive regulation of immune response / blood microparticle / killing of cells of another organism / Regulation of Complement cascade / positive regulation of immune response / blood microparticle / killing of cells of another organism /  immune response / protein-containing complex binding / immune response / protein-containing complex binding /  extracellular space / extracellular exosome / extracellular region / extracellular space / extracellular exosome / extracellular region /  membrane / membrane /  plasma membrane plasma membraneSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  SIRAS, SIRAS,  MAD / Resolution: 2.5 Å MAD / Resolution: 2.5 Å | ||||||
 Authors Authors | Hadders, M.A. / Gros, P. | ||||||
 Citation Citation |  Journal: Science / Year: 2007 Journal: Science / Year: 2007Title: Structure of C8alpha-MACPF reveals mechanism of membrane attack in complement immune defense. Authors: Hadders, M.A. / Beringer, D.X. / Gros, P. | ||||||
| History |
| ||||||
| Remark 999 | Sequence Residues 337 to 379 are identical to those of residues 367 to 409 in UniProt entry P07357, ...Sequence Residues 337 to 379 are identical to those of residues 367 to 409 in UniProt entry P07357, which are ITSRDITTCFGGSLGIQYEDKINVGGGLSGDHCKKFGGGKTER. However, only 12 residues were observed and the alignment between sequence and coordinates is unknown in this region. These 12 residues were originally modeled as ALA/GLY and have been changed to UNK during processing. |
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2qqh.cif.gz 2qqh.cif.gz | 70.9 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2qqh.ent.gz pdb2qqh.ent.gz | 56.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2qqh.json.gz 2qqh.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/qq/2qqh https://data.pdbj.org/pub/pdb/validation_reports/qq/2qqh ftp://data.pdbj.org/pub/pdb/validation_reports/qq/2qqh ftp://data.pdbj.org/pub/pdb/validation_reports/qq/2qqh | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein | Mass: 37543.215 Da / Num. of mol.: 1 / Mutation: C164S Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Gene: C8A / Plasmid: pET / Production host: Homo sapiens (human) / Gene: C8A / Plasmid: pET / Production host:   Escherichia coli (E. coli) / Strain (production host): Origami / References: UniProt: P07357 Escherichia coli (E. coli) / Strain (production host): Origami / References: UniProt: P07357 | ||||
|---|---|---|---|---|---|
| #2: Chemical |  Sulfate Sulfate#3: Chemical | ChemComp-NI / |  Nickel Nickel#4: Water | ChemComp-HOH / |  Water Water |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.98 Å3/Da / Density % sol: 69.08 % |
|---|---|
Crystal grow | Temperature: 303 K / Method: vapor diffusion, sitting drop / pH: 8 Details: 1M LiSO4, 5mM NiCl2, 100mM Tris, pH 8.0, VAPOR DIFFUSION, SITTING DROP, temperature 303K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: ID29 / Wavelength: 1.006 Å / Beamline: ID29 / Wavelength: 1.006 Å |
| Detector | Type: ADSC QUANTUM 315 / Detector: CCD / Date: Mar 10, 2007 |
| Radiation | Monochromator: Si 311 Channel / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.006 Å / Relative weight: 1 : 1.006 Å / Relative weight: 1 |
| Reflection | Resolution: 2.5→40 Å / Num. all: 21349 / Num. obs: 21349 / % possible obs: 100 % / Observed criterion σ(F): 1 / Observed criterion σ(I): 1 / Redundancy: 7.1 % / Biso Wilson estimate: 63.3 Å2 / Rmerge(I) obs: 0.08 / Rsym value: 0.08 / Net I/σ(I): 17.3 |
| Reflection shell | Resolution: 2.5→2.64 Å / Redundancy: 7.1 % / Rmerge(I) obs: 0.513 / Mean I/σ(I) obs: 3.2 / Num. unique all: 2888 / Rsym value: 0.513 / % possible all: 100 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  SIRAS, SIRAS,  MAD / Resolution: 2.5→40 Å / Cor.coef. Fo:Fc: 0.903 / Cor.coef. Fo:Fc free: 0.888 / SU B: 16.508 / SU ML: 0.198 / Isotropic thermal model: isotropic / Cross valid method: THROUGHOUT / ESU R: 0.315 / ESU R Free: 0.255 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS MAD / Resolution: 2.5→40 Å / Cor.coef. Fo:Fc: 0.903 / Cor.coef. Fo:Fc free: 0.888 / SU B: 16.508 / SU ML: 0.198 / Isotropic thermal model: isotropic / Cross valid method: THROUGHOUT / ESU R: 0.315 / ESU R Free: 0.255 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.4 Å / Solvent model: BABINET MODEL WITH MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 53.487 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.5→40 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.5→2.64 Å / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller










 PDBj
PDBj








