+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2n7e | ||||||
|---|---|---|---|---|---|---|---|
| Title | Solution structure of the UBL domain of yeast Ddi1 | ||||||
 Components Components | DNA damage-inducible protein 1 | ||||||
 Keywords Keywords | UNKNOWN FUNCTION /  DDI1 DDI1 | ||||||
| Function / homology |  Function and homology information Function and homology informationproteasome regulatory particle binding / protein transport to vacuole involved in ubiquitin-dependent protein catabolic process via the multivesicular body sorting pathway /  Hydrolases; Acting on peptide bonds (peptidases); Aspartic endopeptidases / Hydrolases; Acting on peptide bonds (peptidases); Aspartic endopeptidases /  protein secretion / polyubiquitin modification-dependent protein binding / vesicle-mediated transport / protein secretion / polyubiquitin modification-dependent protein binding / vesicle-mediated transport /  SNARE binding / positive regulation of DNA replication / SNARE binding / positive regulation of DNA replication /  ubiquitin binding / protein-macromolecule adaptor activity ...proteasome regulatory particle binding / protein transport to vacuole involved in ubiquitin-dependent protein catabolic process via the multivesicular body sorting pathway / ubiquitin binding / protein-macromolecule adaptor activity ...proteasome regulatory particle binding / protein transport to vacuole involved in ubiquitin-dependent protein catabolic process via the multivesicular body sorting pathway /  Hydrolases; Acting on peptide bonds (peptidases); Aspartic endopeptidases / Hydrolases; Acting on peptide bonds (peptidases); Aspartic endopeptidases /  protein secretion / polyubiquitin modification-dependent protein binding / vesicle-mediated transport / protein secretion / polyubiquitin modification-dependent protein binding / vesicle-mediated transport /  SNARE binding / positive regulation of DNA replication / SNARE binding / positive regulation of DNA replication /  ubiquitin binding / protein-macromolecule adaptor activity / ubiquitin-dependent protein catabolic process / aspartic-type endopeptidase activity / ubiquitin binding / protein-macromolecule adaptor activity / ubiquitin-dependent protein catabolic process / aspartic-type endopeptidase activity /  plasma membrane / plasma membrane /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |   Saccharomyces cerevisiae (brewer's yeast) Saccharomyces cerevisiae (brewer's yeast) | ||||||
| Method |  SOLUTION NMR / torsion angle dynamics, SOLUTION NMR / torsion angle dynamics,  molecular dynamics molecular dynamics | ||||||
 Authors Authors | Siva, M. / Grantz Saskova, K. / Veverka, V. | ||||||
 Citation Citation |  Journal: Sci Rep / Year: 2016 Journal: Sci Rep / Year: 2016Title: Structural studies of the yeast DNA damage-inducible protein Ddi1 reveal domain architecture of this eukaryotic protein family. Authors: Trempe, J.F. / Saskova, K.G. / Siva, M. / Ratcliffe, C.D. / Veverka, V. / Hoegl, A. / Menade, M. / Feng, X. / Shenker, S. / Svoboda, M. / Kozisek, M. / Konvalinka, J. / Gehring, K. | ||||||
| History |
|
- Structure visualization
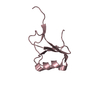
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2n7e.cif.gz 2n7e.cif.gz | 1.1 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2n7e.ent.gz pdb2n7e.ent.gz | 962.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2n7e.json.gz 2n7e.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/n7/2n7e https://data.pdbj.org/pub/pdb/validation_reports/n7/2n7e ftp://data.pdbj.org/pub/pdb/validation_reports/n7/2n7e ftp://data.pdbj.org/pub/pdb/validation_reports/n7/2n7e | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 9691.938 Da / Num. of mol.: 1 / Fragment: UBL domain (UNP residues 1-80) Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Saccharomyces cerevisiae (brewer's yeast) Saccharomyces cerevisiae (brewer's yeast)Strain: ATCC 204508 / S288c / Gene: DDI1, VSM1, YER143W / Production host:   Escherichia coli (E. coli) / References: UniProt: P40087 Escherichia coli (E. coli) / References: UniProt: P40087 |
|---|
-Experimental details
-Experiment
| Experiment | Method:  SOLUTION NMR SOLUTION NMR | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
|
- Sample preparation
Sample preparation
| Details | Contents: 25 mM sodium phosphate, 100 mM sodium chloride, 0.4 mM [U-13C; U-15N] protein, 95% H2O/5% D2O Solvent system: 95% H2O/5% D2O | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample |
| ||||||||||||||||
| Sample conditions | Ionic strength: 0.1 / pH: 6.5 / Pressure: ambient / Temperature: 298 K |
-NMR measurement
| NMR spectrometer | Type: Bruker Avance / Manufacturer: Bruker / Model : AVANCE / Field strength: 600 MHz : AVANCE / Field strength: 600 MHz |
|---|
- Processing
Processing
| NMR software |
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: torsion angle dynamics,  molecular dynamics / Software ordinal: 1 molecular dynamics / Software ordinal: 1 | ||||||||||||
| NMR representative | Selection criteria: lowest energy | ||||||||||||
| NMR ensemble | Conformer selection criteria: structures with the least restraint violations Conformers calculated total number: 100 / Conformers submitted total number: 43 |
 Movie
Movie Controller
Controller















 PDBj
PDBj

