+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2gmd | ||||||
|---|---|---|---|---|---|---|---|
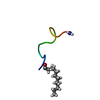
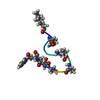
| Title | Structure of C12-LF11 bound to the SDS micelles | ||||||
 Components Components | LACTOFERRIN-BASED SYNTHETIC PEPTIDE C12-LF11 | ||||||
 Keywords Keywords |  ANTIMICROBIAL PROTEIN ANTIMICROBIAL PROTEIN | ||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation by host of viral process / membrane destabilizing activity / Mtb iron assimilation by chelation / phagocytic vesicle lumen / positive regulation of toll-like receptor 4 signaling pathway / Metal sequestration by antimicrobial proteins / negative regulation of viral process / negative regulation of cysteine-type endopeptidase activity / negative regulation of tumor necrosis factor (ligand) superfamily member 11 production / negative regulation of single-species biofilm formation in or on host organism ...negative regulation by host of viral process / membrane destabilizing activity / Mtb iron assimilation by chelation / phagocytic vesicle lumen / positive regulation of toll-like receptor 4 signaling pathway / Metal sequestration by antimicrobial proteins / negative regulation of viral process / negative regulation of cysteine-type endopeptidase activity / negative regulation of tumor necrosis factor (ligand) superfamily member 11 production / negative regulation of single-species biofilm formation in or on host organism / positive regulation of bone mineralization involved in bone maturation / negative regulation of lipopolysaccharide-mediated signaling pathway /  specific granule / negative regulation of osteoclast development / antifungal humoral response / positive regulation of chondrocyte proliferation / negative regulation of ATP-dependent activity / regulation of tumor necrosis factor production / bone morphogenesis / specific granule / negative regulation of osteoclast development / antifungal humoral response / positive regulation of chondrocyte proliferation / negative regulation of ATP-dependent activity / regulation of tumor necrosis factor production / bone morphogenesis /  Antimicrobial peptides / cysteine-type endopeptidase inhibitor activity / negative regulation of viral genome replication / positive regulation of osteoblast proliferation / Antimicrobial peptides / cysteine-type endopeptidase inhibitor activity / negative regulation of viral genome replication / positive regulation of osteoblast proliferation /  humoral immune response / humoral immune response /  Hydrolases; Acting on peptide bonds (peptidases); Serine endopeptidases / positive regulation of osteoblast differentiation / regulation of cytokine production / protein serine/threonine kinase activator activity / Hydrolases; Acting on peptide bonds (peptidases); Serine endopeptidases / positive regulation of osteoblast differentiation / regulation of cytokine production / protein serine/threonine kinase activator activity /  ossification / ossification /  innate immune response in mucosa / innate immune response in mucosa /  secretory granule / secretory granule /  lipopolysaccharide binding / positive regulation of protein serine/threonine kinase activity / recycling endosome / specific granule lumen / antimicrobial humoral immune response mediated by antimicrobial peptide / tertiary granule lumen / lipopolysaccharide binding / positive regulation of protein serine/threonine kinase activity / recycling endosome / specific granule lumen / antimicrobial humoral immune response mediated by antimicrobial peptide / tertiary granule lumen /  heparin binding / positive regulation of NF-kappaB transcription factor activity / antibacterial humoral response / iron ion transport / positive regulation of canonical NF-kappaB signal transduction / killing of cells of another organism / defense response to Gram-negative bacterium / heparin binding / positive regulation of NF-kappaB transcription factor activity / antibacterial humoral response / iron ion transport / positive regulation of canonical NF-kappaB signal transduction / killing of cells of another organism / defense response to Gram-negative bacterium /  early endosome / iron ion binding / Amyloid fiber formation / serine-type endopeptidase activity / Neutrophil degranulation / negative regulation of apoptotic process / early endosome / iron ion binding / Amyloid fiber formation / serine-type endopeptidase activity / Neutrophil degranulation / negative regulation of apoptotic process /  cell surface / protein-containing complex / cell surface / protein-containing complex /  proteolysis / proteolysis /  DNA binding / DNA binding /  extracellular space / extracellular exosome / extracellular region / extracellular space / extracellular exosome / extracellular region /  nucleus / nucleus /  plasma membrane / plasma membrane /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species | synthetic construct (others) | ||||||
| Method |  SOLUTION NMR / torsion angle dynamics SOLUTION NMR / torsion angle dynamics | ||||||
 Authors Authors | Japelj, B. | ||||||
 Citation Citation |  Journal: J.Am.Chem.Soc. / Year: 2007 Journal: J.Am.Chem.Soc. / Year: 2007Title: The Acyl Group as the Central Element of the Structural Organization of Antimicrobial Lipopeptide. Authors: Japelj, B. / Zorko, M. / Majerle, A. / Pristovsek, P. / Sanchez-Gomez, S. / Tejada, G.M. / Moriyon, I. / Blondelle, S.E. / Brandenburg, K. / Andra, J. / Lohner, K. / Jerala, R. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2gmd.cif.gz 2gmd.cif.gz | 92.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2gmd.ent.gz pdb2gmd.ent.gz | 74.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2gmd.json.gz 2gmd.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/gm/2gmd https://data.pdbj.org/pub/pdb/validation_reports/gm/2gmd ftp://data.pdbj.org/pub/pdb/validation_reports/gm/2gmd ftp://data.pdbj.org/pub/pdb/validation_reports/gm/2gmd | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein/peptide | Mass: 1532.817 Da / Num. of mol.: 1 / Source method: obtained synthetically / Source: (synth.) synthetic construct (others) |
|---|---|
| #2: Chemical | ChemComp-DAO /  Lauric acid Lauric acid |
-Experimental details
-Experiment
| Experiment | Method:  SOLUTION NMR SOLUTION NMR | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
|
- Sample preparation
Sample preparation
| Details | Contents: 1.3 mM C12-LF11, 227.1 mM d25-SDS (sodiumdodecylsulphate), 20 mM phosphate buffer Na, 7% D20, 93 % H20 Solvent system: 7% D20, 93 %H20 |
|---|---|
| Sample conditions | Ionic strength: 0.247 / pH: 5.5 / Pressure: ambient / Temperature: 303 K |
-NMR measurement
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M |
|---|---|
| Radiation wavelength | Relative weight: 1 |
| NMR spectrometer | Type: Varian UNITY / Manufacturer: Varian / Model : UNITY / Field strength: 600 MHz : UNITY / Field strength: 600 MHz |
- Processing
Processing
| NMR software |
| ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: torsion angle dynamics / Software ordinal: 1 | ||||||||||||||||
| NMR representative | Selection criteria: closest to the average | ||||||||||||||||
| NMR ensemble | Conformer selection criteria: all calculated structures submitted Conformers calculated total number: 20 / Conformers submitted total number: 20 |
 Movie
Movie Controller
Controller










 PDBj
PDBj