| Entry | Database: PDB / ID: 2ci9
|
|---|
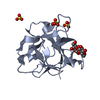
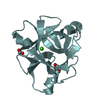
| Title | Nck1 SH2-domain in complex with a dodecaphosphopeptide from EPEC protein Tir |
|---|
 Components Components | - CYTOPLASMIC PROTEIN NCK1
 Cytoplasm Cytoplasm - TRANSLOCATED INTIMIN RECEPTOR
|
|---|
 Keywords Keywords |  PROTEIN BINDING / PROTEIN-BINDING / SH2-DOMAIN-COMPLEX PROTEIN BINDING / PROTEIN-BINDING / SH2-DOMAIN-COMPLEX |
|---|
| Function / homology |  Function and homology information Function and homology information
positive regulation of cap-dependent translational initiation / negative regulation of endoplasmic reticulum stress-induced eIF2 alpha phosphorylation / eukaryotic initiation factor eIF2 binding / positive regulation of peptidyl-serine dephosphorylation / protein phosphatase type 1 complex / positive regulation of translation in response to endoplasmic reticulum stress / positive regulation of cap-independent translational initiation / positive regulation of endoplasmic reticulum stress-induced intrinsic apoptotic signaling pathway / cytoskeletal anchor activity / substrate-dependent cell migration, cell extension ...positive regulation of cap-dependent translational initiation / negative regulation of endoplasmic reticulum stress-induced eIF2 alpha phosphorylation / eukaryotic initiation factor eIF2 binding / positive regulation of peptidyl-serine dephosphorylation / protein phosphatase type 1 complex / positive regulation of translation in response to endoplasmic reticulum stress / positive regulation of cap-independent translational initiation / positive regulation of endoplasmic reticulum stress-induced intrinsic apoptotic signaling pathway / cytoskeletal anchor activity / substrate-dependent cell migration, cell extension / vesicle membrane / signal complex assembly / Activation of RAC1 / Nephrin family interactions / DCC mediated attractive signaling /  lamellipodium assembly / RHOV GTPase cycle / positive regulation of actin filament polymerization / negative regulation of PERK-mediated unfolded protein response / lamellipodium assembly / RHOV GTPase cycle / positive regulation of actin filament polymerization / negative regulation of PERK-mediated unfolded protein response /  protein kinase inhibitor activity / antiviral innate immune response / RHOU GTPase cycle / Generation of second messenger molecules / RHO GTPases Activate WASPs and WAVEs / ephrin receptor signaling pathway / signaling adaptor activity / negative regulation of peptidyl-serine phosphorylation / positive regulation of T cell proliferation / protein kinase inhibitor activity / antiviral innate immune response / RHOU GTPase cycle / Generation of second messenger molecules / RHO GTPases Activate WASPs and WAVEs / ephrin receptor signaling pathway / signaling adaptor activity / negative regulation of peptidyl-serine phosphorylation / positive regulation of T cell proliferation /  regulation of cell migration / regulation of cell migration /  T cell activation / negative regulation of insulin receptor signaling pathway / response to endoplasmic reticulum stress / T cell activation / negative regulation of insulin receptor signaling pathway / response to endoplasmic reticulum stress /  ephrin receptor binding / Downstream signal transduction / Antigen activates B Cell Receptor (BCR) leading to generation of second messengers / molecular condensate scaffold activity / actin filament organization / FCGR3A-mediated phagocytosis / PKR-mediated signaling / ephrin receptor binding / Downstream signal transduction / Antigen activates B Cell Receptor (BCR) leading to generation of second messengers / molecular condensate scaffold activity / actin filament organization / FCGR3A-mediated phagocytosis / PKR-mediated signaling /  receptor tyrosine kinase binding / Regulation of actin dynamics for phagocytic cup formation / VEGFA-VEGFR2 Pathway / positive regulation of neuron projection development / cell-cell junction / signaling receptor complex adaptor activity / receptor tyrosine kinase binding / Regulation of actin dynamics for phagocytic cup formation / VEGFA-VEGFR2 Pathway / positive regulation of neuron projection development / cell-cell junction / signaling receptor complex adaptor activity /  cell migration / protein-macromolecule adaptor activity / Potential therapeutics for SARS / cell migration / protein-macromolecule adaptor activity / Potential therapeutics for SARS /  ribosome / ribosome /  cadherin binding / protein domain specific binding / cadherin binding / protein domain specific binding /  signaling receptor binding / host cell plasma membrane / negative regulation of transcription by RNA polymerase II / signaling receptor binding / host cell plasma membrane / negative regulation of transcription by RNA polymerase II /  endoplasmic reticulum / endoplasmic reticulum /  signal transduction / positive regulation of transcription by RNA polymerase II / extracellular region / signal transduction / positive regulation of transcription by RNA polymerase II / extracellular region /  membrane / membrane /  nucleus / nucleus /  plasma membrane / plasma membrane /  cytosol / cytosol /  cytoplasmSimilarity search - Function cytoplasmSimilarity search - Function Translocated intimin receptor, central domain / Translocated intimin receptor, N-terminal / Translocated intimin receptor / Translocated intimin receptor, C-terminal / Translocated intimin receptor, central domain superfamily / Translocated intimin receptor (Tir) intimin-binding domain / Translocated intimin receptor (Tir) C-terminus / Translocated intimin receptor (Tir) N-terminus /  Nck1, SH3 domain 1 / Nck1, SH3 domain 1 /  Nck1, SH3 domain 2 ...Translocated intimin receptor, central domain / Translocated intimin receptor, N-terminal / Translocated intimin receptor / Translocated intimin receptor, C-terminal / Translocated intimin receptor, central domain superfamily / Translocated intimin receptor (Tir) intimin-binding domain / Translocated intimin receptor (Tir) C-terminus / Translocated intimin receptor (Tir) N-terminus / Nck1, SH3 domain 2 ...Translocated intimin receptor, central domain / Translocated intimin receptor, N-terminal / Translocated intimin receptor / Translocated intimin receptor, C-terminal / Translocated intimin receptor, central domain superfamily / Translocated intimin receptor (Tir) intimin-binding domain / Translocated intimin receptor (Tir) C-terminus / Translocated intimin receptor (Tir) N-terminus /  Nck1, SH3 domain 1 / Nck1, SH3 domain 1 /  Nck1, SH3 domain 2 / Nck1, SH3 domain 2 /  Nck1, SH3 domain 3 / Nck1, SH3 domain 3 /  Nck1, SH2 domain / Nck1, SH2 domain /  Cytoplasmic protein NCK / Cytoplasmic protein NCK /  SH2 domain / SHC Adaptor Protein / SH2 domain / SHC Adaptor Protein /  SH3 domain / SH3 domain /  SH2 domain / Src homology 2 (SH2) domain profile. / Src homology 2 domains / SH2 domain / Src homology 2 (SH2) domain profile. / Src homology 2 domains /  SH2 domain / Src homology 3 domains / SH2 domain superfamily / SH3-like domain superfamily / Src homology 3 (SH3) domain profile. / SH2 domain / Src homology 3 domains / SH2 domain superfamily / SH3-like domain superfamily / Src homology 3 (SH3) domain profile. /  SH3 domain / 2-Layer Sandwich / Alpha BetaSimilarity search - Domain/homology SH3 domain / 2-Layer Sandwich / Alpha BetaSimilarity search - Domain/homology |
|---|
| Biological species |   HOMO SAPIENS (human) HOMO SAPIENS (human)
   ESCHERICHIA COLI (E. coli) ESCHERICHIA COLI (E. coli) |
|---|
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.5 Å MOLECULAR REPLACEMENT / Resolution: 1.5 Å |
|---|
 Authors Authors | Frese, S. / Schubert, W.-D. / Findeis, A.C. / Marquardt, T. / Roske, Y.S. / Stradal, T.E.B. / Heinz, D.W. |
|---|
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2006 Journal: J.Biol.Chem. / Year: 2006
Title: The Phosphotyrosine Peptide Binding Specificity of Nck1 and Nck2 Src Homology 2 Domains.
Authors: Frese, S. / Schubert, W.-D. / Findeis, A.C. / Marquardt, T. / Roske, Y.S. / Stradal, T.E.B. / Heinz, D.W. |
|---|
| History | | Deposition | Mar 17, 2006 | Deposition site: PDBE / Processing site: PDBE |
|---|
| Revision 1.0 | Apr 24, 2006 | Provider: repository / Type: Initial release |
|---|
| Revision 1.1 | Jul 13, 2011 | Group: Refinement description / Version format compliance |
|---|
| Revision 1.2 | May 15, 2019 | Group: Data collection / Derived calculations ...Data collection / Derived calculations / Experimental preparation / Other
Category: exptl_crystal_grow / pdbx_database_proc ...exptl_crystal_grow / pdbx_database_proc / pdbx_database_status / struct_conn
Item: _exptl_crystal_grow.method / _pdbx_database_status.recvd_author_approval / _struct_conn.pdbx_leaving_atom_flag |
|---|
| Revision 1.3 | May 1, 2024 | Group: Data collection / Database references ...Data collection / Database references / Other / Refinement description
Category: chem_comp_atom / chem_comp_bond ...chem_comp_atom / chem_comp_bond / database_2 / pdbx_database_status / pdbx_initial_refinement_model
Item: _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _pdbx_database_status.status_code_sf |
|---|
|
|---|
| Remark 650 | HELIX DETERMINATION METHOD: AUTHOR PROVIDED. |
|---|
| Remark 700 | SHEET DETERMINATION METHOD: AUTHOR PROVIDED. |
|---|
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords PROTEIN BINDING / PROTEIN-BINDING / SH2-DOMAIN-COMPLEX
PROTEIN BINDING / PROTEIN-BINDING / SH2-DOMAIN-COMPLEX Function and homology information
Function and homology information lamellipodium assembly / RHOV GTPase cycle / positive regulation of actin filament polymerization / negative regulation of PERK-mediated unfolded protein response /
lamellipodium assembly / RHOV GTPase cycle / positive regulation of actin filament polymerization / negative regulation of PERK-mediated unfolded protein response /  protein kinase inhibitor activity / antiviral innate immune response / RHOU GTPase cycle / Generation of second messenger molecules / RHO GTPases Activate WASPs and WAVEs / ephrin receptor signaling pathway / signaling adaptor activity / negative regulation of peptidyl-serine phosphorylation / positive regulation of T cell proliferation /
protein kinase inhibitor activity / antiviral innate immune response / RHOU GTPase cycle / Generation of second messenger molecules / RHO GTPases Activate WASPs and WAVEs / ephrin receptor signaling pathway / signaling adaptor activity / negative regulation of peptidyl-serine phosphorylation / positive regulation of T cell proliferation /  regulation of cell migration /
regulation of cell migration /  T cell activation / negative regulation of insulin receptor signaling pathway / response to endoplasmic reticulum stress /
T cell activation / negative regulation of insulin receptor signaling pathway / response to endoplasmic reticulum stress /  ephrin receptor binding / Downstream signal transduction / Antigen activates B Cell Receptor (BCR) leading to generation of second messengers / molecular condensate scaffold activity / actin filament organization / FCGR3A-mediated phagocytosis / PKR-mediated signaling /
ephrin receptor binding / Downstream signal transduction / Antigen activates B Cell Receptor (BCR) leading to generation of second messengers / molecular condensate scaffold activity / actin filament organization / FCGR3A-mediated phagocytosis / PKR-mediated signaling /  receptor tyrosine kinase binding / Regulation of actin dynamics for phagocytic cup formation / VEGFA-VEGFR2 Pathway / positive regulation of neuron projection development / cell-cell junction / signaling receptor complex adaptor activity /
receptor tyrosine kinase binding / Regulation of actin dynamics for phagocytic cup formation / VEGFA-VEGFR2 Pathway / positive regulation of neuron projection development / cell-cell junction / signaling receptor complex adaptor activity /  cell migration / protein-macromolecule adaptor activity / Potential therapeutics for SARS /
cell migration / protein-macromolecule adaptor activity / Potential therapeutics for SARS /  ribosome /
ribosome /  cadherin binding / protein domain specific binding /
cadherin binding / protein domain specific binding /  signaling receptor binding / host cell plasma membrane / negative regulation of transcription by RNA polymerase II /
signaling receptor binding / host cell plasma membrane / negative regulation of transcription by RNA polymerase II /  endoplasmic reticulum /
endoplasmic reticulum /  signal transduction / positive regulation of transcription by RNA polymerase II / extracellular region /
signal transduction / positive regulation of transcription by RNA polymerase II / extracellular region /  membrane /
membrane /  nucleus /
nucleus /  plasma membrane /
plasma membrane /  cytosol /
cytosol /  cytoplasm
cytoplasm
 HOMO SAPIENS (human)
HOMO SAPIENS (human)
 ESCHERICHIA COLI (E. coli)
ESCHERICHIA COLI (E. coli) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.5 Å
MOLECULAR REPLACEMENT / Resolution: 1.5 Å  Authors
Authors Citation
Citation Journal: J.Biol.Chem. / Year: 2006
Journal: J.Biol.Chem. / Year: 2006 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 2ci9.cif.gz
2ci9.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb2ci9.ent.gz
pdb2ci9.ent.gz PDB format
PDB format 2ci9.json.gz
2ci9.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/ci/2ci9
https://data.pdbj.org/pub/pdb/validation_reports/ci/2ci9 ftp://data.pdbj.org/pub/pdb/validation_reports/ci/2ci9
ftp://data.pdbj.org/pub/pdb/validation_reports/ci/2ci9 Links
Links Assembly
Assembly


 Components
Components Cytoplasm / NCK ADAPTOR PROTEIN 1 / SH2/SH3 ADAPTOR PROTEIN NCK-ALPHA
Cytoplasm / NCK ADAPTOR PROTEIN 1 / SH2/SH3 ADAPTOR PROTEIN NCK-ALPHA
 HOMO SAPIENS (human) / Plasmid: PGEX-6P-1 (AMERSHAM BIOSCIENCES) / Production host:
HOMO SAPIENS (human) / Plasmid: PGEX-6P-1 (AMERSHAM BIOSCIENCES) / Production host: 
 ESCHERICHIA COLI (E. coli) / Strain (production host): BL21 / References: UniProt: P16333
ESCHERICHIA COLI (E. coli) / Strain (production host): BL21 / References: UniProt: P16333
 ESCHERICHIA COLI (E. coli) / References: UniProt: O50190, UniProt: B7UM99*PLUS
ESCHERICHIA COLI (E. coli) / References: UniProt: O50190, UniProt: B7UM99*PLUS Water
Water X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation
 SYNCHROTRON / Site: MPG/DESY, HAMBURG
SYNCHROTRON / Site: MPG/DESY, HAMBURG  / Beamline: BW6 / Wavelength: 1.05
/ Beamline: BW6 / Wavelength: 1.05  : 1.05 Å / Relative weight: 1
: 1.05 Å / Relative weight: 1  Processing
Processing :
:  MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller













 PDBj
PDBj

















