[English] 日本語
 Yorodumi
Yorodumi- PDB-1tgj: HUMAN TRANSFORMING GROWTH FACTOR-BETA 3, CRYSTALLIZED FROM DIOXANE -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1tgj | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | HUMAN TRANSFORMING GROWTH FACTOR-BETA 3, CRYSTALLIZED FROM DIOXANE | |||||||||
 Components Components | TRANSFORMING GROWTH FACTOR-BETA 3 Transforming growth factor beta Transforming growth factor beta | |||||||||
 Keywords Keywords |  GROWTH FACTOR / GROWTH FACTOR /  MITOGEN / MITOGEN /  GLYCOPROTEIN GLYCOPROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationfrontal suture morphogenesis / uterine wall breakdown / detection of hypoxia / embryonic neurocranium morphogenesis / type III transforming growth factor beta receptor binding / positive regulation of tight junction disassembly / negative regulation of macrophage cytokine production /  secondary palate development / response to laminar fluid shear stress / type II transforming growth factor beta receptor binding ...frontal suture morphogenesis / uterine wall breakdown / detection of hypoxia / embryonic neurocranium morphogenesis / type III transforming growth factor beta receptor binding / positive regulation of tight junction disassembly / negative regulation of macrophage cytokine production / secondary palate development / response to laminar fluid shear stress / type II transforming growth factor beta receptor binding ...frontal suture morphogenesis / uterine wall breakdown / detection of hypoxia / embryonic neurocranium morphogenesis / type III transforming growth factor beta receptor binding / positive regulation of tight junction disassembly / negative regulation of macrophage cytokine production /  secondary palate development / response to laminar fluid shear stress / type II transforming growth factor beta receptor binding / type I transforming growth factor beta receptor binding / secondary palate development / response to laminar fluid shear stress / type II transforming growth factor beta receptor binding / type I transforming growth factor beta receptor binding /  mammary gland development / cell-cell junction organization / digestive tract development / mammary gland development / cell-cell junction organization / digestive tract development /  transforming growth factor beta binding / face morphogenesis / transforming growth factor beta binding / face morphogenesis /  odontogenesis / Molecules associated with elastic fibres / positive regulation of filopodium assembly / lung alveolus development / TGF-beta receptor signaling activates SMADs / inner ear development / negative regulation of vascular associated smooth muscle cell proliferation / positive regulation of SMAD protein signal transduction / positive regulation of cell division / ECM proteoglycans / positive regulation of collagen biosynthetic process / positive regulation of epithelial to mesenchymal transition / salivary gland morphogenesis / positive regulation of stress fiber assembly / odontogenesis / Molecules associated with elastic fibres / positive regulation of filopodium assembly / lung alveolus development / TGF-beta receptor signaling activates SMADs / inner ear development / negative regulation of vascular associated smooth muscle cell proliferation / positive regulation of SMAD protein signal transduction / positive regulation of cell division / ECM proteoglycans / positive regulation of collagen biosynthetic process / positive regulation of epithelial to mesenchymal transition / salivary gland morphogenesis / positive regulation of stress fiber assembly /  T-tubule / transforming growth factor beta receptor signaling pathway / platelet alpha granule lumen / response to progesterone / female pregnancy / T-tubule / transforming growth factor beta receptor signaling pathway / platelet alpha granule lumen / response to progesterone / female pregnancy /  cytokine activity / positive regulation of protein secretion / negative regulation of transforming growth factor beta receptor signaling pathway / cytokine activity / positive regulation of protein secretion / negative regulation of transforming growth factor beta receptor signaling pathway /  growth factor activity / response to estrogen / Platelet degranulation / regulation of cell population proliferation / collagen-containing extracellular matrix / in utero embryonic development / negative regulation of neuron apoptotic process / positive regulation of MAPK cascade / response to hypoxia / positive regulation of apoptotic process / negative regulation of cell population proliferation / intracellular membrane-bounded organelle / neuronal cell body / positive regulation of cell population proliferation / protein-containing complex binding / positive regulation of DNA-templated transcription / growth factor activity / response to estrogen / Platelet degranulation / regulation of cell population proliferation / collagen-containing extracellular matrix / in utero embryonic development / negative regulation of neuron apoptotic process / positive regulation of MAPK cascade / response to hypoxia / positive regulation of apoptotic process / negative regulation of cell population proliferation / intracellular membrane-bounded organelle / neuronal cell body / positive regulation of cell population proliferation / protein-containing complex binding / positive regulation of DNA-templated transcription /  cell surface / positive regulation of transcription by RNA polymerase II / cell surface / positive regulation of transcription by RNA polymerase II /  extracellular space / extracellular region / identical protein binding / extracellular space / extracellular region / identical protein binding /  plasma membrane plasma membraneSimilarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  molecular replacement / Resolution: 2 Å molecular replacement / Resolution: 2 Å | |||||||||
 Authors Authors | Mittl, P.R.E. / Priestle, J.P. / Gruetter, M.G. | |||||||||
 Citation Citation |  Journal: Protein Sci. / Year: 1996 Journal: Protein Sci. / Year: 1996Title: The crystal structure of TGF-beta 3 and comparison to TGF-beta 2: implications for receptor binding. Authors: Mittl, P.R. / Priestle, J.P. / Cox, D.A. / McMaster, G. / Cerletti, N. / Grutter, M.G. #1:  Journal: J.Mol.Biol. / Year: 1993 Journal: J.Mol.Biol. / Year: 1993Title: Refined Crystal Structure of Human Transforming Growth Factor Beta 2 at 1.95 A Resolution Authors: Schlunegger, M.P. / Grutter, M.G. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1tgj.cif.gz 1tgj.cif.gz | 37.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1tgj.ent.gz pdb1tgj.ent.gz | 24.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1tgj.json.gz 1tgj.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/tg/1tgj https://data.pdbj.org/pub/pdb/validation_reports/tg/1tgj ftp://data.pdbj.org/pub/pdb/validation_reports/tg/1tgj ftp://data.pdbj.org/pub/pdb/validation_reports/tg/1tgj | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| |||||||||
| Unit cell |
| |||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein |  Transforming growth factor beta / TGF-BETA3 Transforming growth factor beta / TGF-BETA3Mass: 12734.504 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Details: ONE DIOXANE MOLECULE BOUND / Source: (gene. exp.)   Homo sapiens (human) Homo sapiens (human)Description: STRAIN DEPOSITED AT DEUTSCHE SAMMLUNG VON MIKROORGANISMEN, MASCHENRODER WEG 1B, 3300 BRAUNSCHWEIG, GERMANY, ACCESSION NUMBER IS DSM 5658 Gene: HTGF-BETA3 / Plasmid: PPLMU / Gene (production host): HTGF-BETA3 / Production host:   Escherichia coli (E. coli) / Strain (production host): LC137 / References: UniProt: P10600 Escherichia coli (E. coli) / Strain (production host): LC137 / References: UniProt: P10600 |
|---|---|
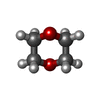
| #2: Chemical | ChemComp-DIO /  1,4-Dioxane 1,4-Dioxane |
| #3: Water | ChemComp-HOH /  Water Water |
| Nonpolymer details | THE PROTEIN WAS CRYSTALLIZED FROM A SOLUTION CONTAINING 15 % DIOXANE. ONE DIOXANE MOLECULE IS BOUND ...THE PROTEIN WAS CRYSTALLIZ |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.25 Å3/Da / Density % sol: 43.37 % | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Crystal grow | pH: 5 / Details: 15% (V/V) DIOXANE, 100 MM SODIUM ACETATE, PH 5.0 | ||||||||||||||||||||||||||||||||||||
| Crystal | *PLUS | ||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Method: vapor diffusion | ||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 293 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: ENRAF-NONIUS FR571 / Wavelength: 1.5418 ROTATING ANODE / Type: ENRAF-NONIUS FR571 / Wavelength: 1.5418 |
| Detector | Type: ENRAF-NONIUS FAST / Detector: DIFFRACTOMETER / Date: Dec 16, 1994 |
| Radiation | Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.5418 Å / Relative weight: 1 : 1.5418 Å / Relative weight: 1 |
| Reflection | Num. obs: 6323 / % possible obs: 76 % / Observed criterion σ(I): 0 / Redundancy: 3.7 % / Rmerge(I) obs: 0.07 |
| Reflection shell | Resolution: 2→2.1 Å / Rmerge(I) obs: 0.353 / % possible all: 37.4 |
| Reflection | *PLUS Highest resolution: 2 Å / Lowest resolution: 10 Å |
| Reflection shell | *PLUS % possible obs: 37.4 % |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  molecular replacement molecular replacementStarting model: TGF-BETA2 (SCHLUNEGGER & GRUETTER) Resolution: 2→7 Å / σ(F): 0
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 29.6 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze | Luzzati coordinate error obs: 0.23 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2→7 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2→2.1 Å / % reflection obs: 37.4 % | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name:  X-PLOR / Version: 3.1 / Classification: refinement X-PLOR / Version: 3.1 / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller









 PDBj
PDBj