[English] 日本語
 Yorodumi
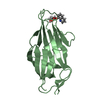
Yorodumi- PDB-1rlw: CALCIUM-PHOSPHOLIPID BINDING DOMAIN FROM CYTOSOLIC PHOSPHOLIPASE A2 -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1rlw | ||||||
|---|---|---|---|---|---|---|---|
| Title | CALCIUM-PHOSPHOLIPID BINDING DOMAIN FROM CYTOSOLIC PHOSPHOLIPASE A2 | ||||||
 Components Components | PHOSPHOLIPASE A2 | ||||||
 Keywords Keywords |  HYDROLASE / HYDROLASE /  C2 DOMAIN / CALB DOMAIN C2 DOMAIN / CALB DOMAIN | ||||||
| Function / homology |  Function and homology information Function and homology informationplatelet activating factor biosynthetic process / phosphatidylglycerol catabolic process /  Arachidonic acid metabolism / icosanoid metabolic process / Acyl chain remodeling of CL / monoacylglycerol biosynthetic process / glycerophospholipid catabolic process / Hydrolysis of LPC / phosphatidyl phospholipase B activity / Arachidonic acid metabolism / icosanoid metabolic process / Acyl chain remodeling of CL / monoacylglycerol biosynthetic process / glycerophospholipid catabolic process / Hydrolysis of LPC / phosphatidyl phospholipase B activity /  lysophospholipase ...platelet activating factor biosynthetic process / phosphatidylglycerol catabolic process / lysophospholipase ...platelet activating factor biosynthetic process / phosphatidylglycerol catabolic process /  Arachidonic acid metabolism / icosanoid metabolic process / Acyl chain remodeling of CL / monoacylglycerol biosynthetic process / glycerophospholipid catabolic process / Hydrolysis of LPC / phosphatidyl phospholipase B activity / Arachidonic acid metabolism / icosanoid metabolic process / Acyl chain remodeling of CL / monoacylglycerol biosynthetic process / glycerophospholipid catabolic process / Hydrolysis of LPC / phosphatidyl phospholipase B activity /  lysophospholipase / lysophospholipase /  O-acyltransferase activity / phosphatidylcholine acyl-chain remodeling / Acyl chain remodelling of PG / calcium-independent phospholipase A2 activity / Acyl chain remodelling of PC / Acyl chain remodelling of PI / Acyl chain remodelling of PS / Acyl chain remodelling of PE / ceramide 1-phosphate binding / Synthesis of PA / arachidonic acid metabolic process / glycerol metabolic process / positive regulation of T-helper 1 type immune response / phosphatidylcholine catabolic process / phosphatidylinositol-5-phosphate binding / positive regulation of prostaglandin secretion / O-acyltransferase activity / phosphatidylcholine acyl-chain remodeling / Acyl chain remodelling of PG / calcium-independent phospholipase A2 activity / Acyl chain remodelling of PC / Acyl chain remodelling of PI / Acyl chain remodelling of PS / Acyl chain remodelling of PE / ceramide 1-phosphate binding / Synthesis of PA / arachidonic acid metabolic process / glycerol metabolic process / positive regulation of T-helper 1 type immune response / phosphatidylcholine catabolic process / phosphatidylinositol-5-phosphate binding / positive regulation of prostaglandin secretion /  lysophospholipase activity / phospho-PLA2 pathway / lysophospholipase activity / phospho-PLA2 pathway /  phospholipase A2 activity / COPI-independent Golgi-to-ER retrograde traffic / phospholipase A2 activity / COPI-independent Golgi-to-ER retrograde traffic /  phosphatidylinositol-3-phosphate binding / leukotriene biosynthetic process / calcium-dependent phospholipid binding / calcium-dependent phospholipase A2 activity / positive regulation of macrophage activation / positive regulation of platelet activation / phosphatidylinositol-4-phosphate binding / phosphatidylinositol-3-phosphate binding / leukotriene biosynthetic process / calcium-dependent phospholipid binding / calcium-dependent phospholipase A2 activity / positive regulation of macrophage activation / positive regulation of platelet activation / phosphatidylinositol-4-phosphate binding /  phospholipase A2 / prostaglandin biosynthetic process / cellular response to antibiotic / arachidonic acid secretion / Platelet sensitization by LDL / establishment of localization in cell / ADP signalling through P2Y purinoceptor 1 / phospholipase A2 / prostaglandin biosynthetic process / cellular response to antibiotic / arachidonic acid secretion / Platelet sensitization by LDL / establishment of localization in cell / ADP signalling through P2Y purinoceptor 1 /  nuclear envelope / regulation of cell population proliferation / nuclear envelope / regulation of cell population proliferation /  mitochondrial inner membrane / mitochondrial inner membrane /  Golgi membrane / intracellular membrane-bounded organelle / Golgi membrane / intracellular membrane-bounded organelle /  calcium ion binding / endoplasmic reticulum membrane / calcium ion binding / endoplasmic reticulum membrane /  Golgi apparatus / Golgi apparatus /  endoplasmic reticulum / endoplasmic reticulum /  nucleus / nucleus /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MIR / Resolution: 2.4 Å MIR / Resolution: 2.4 Å | ||||||
 Authors Authors | Perisic, O. / Williams, R.L. | ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 1998 Journal: J.Biol.Chem. / Year: 1998Title: Crystal structure of a calcium-phospholipid binding domain from cytosolic phospholipase A2. Authors: Perisic, O. / Fong, S. / Lynch, D.E. / Bycroft, M. / Williams, R.L. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1rlw.cif.gz 1rlw.cif.gz | 43.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1rlw.ent.gz pdb1rlw.ent.gz | 29.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1rlw.json.gz 1rlw.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/rl/1rlw https://data.pdbj.org/pub/pdb/validation_reports/rl/1rlw ftp://data.pdbj.org/pub/pdb/validation_reports/rl/1rlw ftp://data.pdbj.org/pub/pdb/validation_reports/rl/1rlw | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein |  / CALB DOMAIN / CALB DOMAINMass: 14299.154 Da / Num. of mol.: 1 / Fragment: C2 DOMAIN, RESIDUES 17 - 141 FROM CPLA2 / Mutation: Y16S, C139A, C141S Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Cellular location: CYTOSOL Homo sapiens (human) / Cellular location: CYTOSOL / Plasmid: MINI-PRSETA / Production host: / Plasmid: MINI-PRSETA / Production host:   Escherichia coli (E. coli) / Strain (production host): BLR / Variant (production host): DE3 / References: UniProt: P47712, Escherichia coli (E. coli) / Strain (production host): BLR / Variant (production host): DE3 / References: UniProt: P47712,  phospholipase A2 phospholipase A2 | ||||
|---|---|---|---|---|---|
| #2: Chemical | | #3: Chemical | #4: Water | ChemComp-HOH / |  Water Water |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 5 Å3/Da / Density % sol: 70 % | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Crystal grow | pH: 7.5 Details: 5-10MG/ML PROTEIN, 5MM CACL2, 1M SODIUM ACETATE, 0.1M HEPES (PH 7.5), 50MM CDSO4 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 21 ℃ / Method: vapor diffusion, hanging drop | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  EMBL/DESY, HAMBURG EMBL/DESY, HAMBURG  / Beamline: BW7B / Wavelength: 0.8838 / Beamline: BW7B / Wavelength: 0.8838 |
| Detector | Type: MARRESEARCH / Detector: IMAGE PLATE / Date: Jul 7, 1997 / Details: BENT MIRROR |
| Radiation | Monochromator: SI(111) / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 0.8838 Å / Relative weight: 1 : 0.8838 Å / Relative weight: 1 |
| Reflection | Resolution: 2.3→31 Å / Num. obs: 11798 / % possible obs: 98.8 % / Observed criterion σ(I): 0 / Redundancy: 4.4 % / Biso Wilson estimate: 41 Å2 / Rmerge(I) obs: 0.086 / Rsym value: 0.086 / Net I/σ(I): 18 |
| Reflection shell | Resolution: 2.3→2.35 Å / Redundancy: 2.6 % / Rmerge(I) obs: 0.31 / Mean I/σ(I) obs: 3.4 / Rsym value: 0.31 / % possible all: 86 |
| Reflection | *PLUS Num. measured all: 52266 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MIR / Resolution: 2.4→15 Å / Cross valid method: THROUGHOUT / σ(F): 0 MIR / Resolution: 2.4→15 Å / Cross valid method: THROUGHOUT / σ(F): 0
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 25 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze | Luzzati d res low obs: 8 Å / Luzzati sigma a obs: 0.03 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.4→15 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name: REFMAC / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Num. reflection all: 10342 / Rfactor obs: 0.228 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS |
 Movie
Movie Controller
Controller











 PDBj
PDBj










