[English] 日本語
 Yorodumi
Yorodumi- PDB-1n29: Crystal structure of the N1A mutant of human group IIA phospholip... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1n29 | ||||||
|---|---|---|---|---|---|---|---|
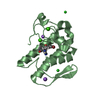
| Title | Crystal structure of the N1A mutant of human group IIA phospholipase A2 | ||||||
 Components Components | Phospholipase A2, membrane associated | ||||||
 Keywords Keywords |  HYDROLASE HYDROLASE | ||||||
| Function / homology |  Function and homology information Function and homology information regulation of neutrophil activation / phosphatidylethanolamine metabolic process / phosphatidic acid metabolic process / Acyl chain remodelling of PG / intestinal stem cell homeostasis / Acyl chain remodelling of PC / Acyl chain remodelling of PI / Acyl chain remodelling of PS / Acyl chain remodelling of PE / Synthesis of PA ... regulation of neutrophil activation / phosphatidylethanolamine metabolic process / phosphatidic acid metabolic process / Acyl chain remodelling of PG / intestinal stem cell homeostasis / Acyl chain remodelling of PC / Acyl chain remodelling of PI / Acyl chain remodelling of PS / Acyl chain remodelling of PE / Synthesis of PA ... regulation of neutrophil activation / phosphatidylethanolamine metabolic process / phosphatidic acid metabolic process / Acyl chain remodelling of PG / intestinal stem cell homeostasis / Acyl chain remodelling of PC / Acyl chain remodelling of PI / Acyl chain remodelling of PS / Acyl chain remodelling of PE / Synthesis of PA / calcium-dependent phospholipase A2 activity / positive regulation of macrophage derived foam cell differentiation / phosphatidylcholine metabolic process / low-density lipoprotein particle remodeling / regulation of neutrophil activation / phosphatidylethanolamine metabolic process / phosphatidic acid metabolic process / Acyl chain remodelling of PG / intestinal stem cell homeostasis / Acyl chain remodelling of PC / Acyl chain remodelling of PI / Acyl chain remodelling of PS / Acyl chain remodelling of PE / Synthesis of PA / calcium-dependent phospholipase A2 activity / positive regulation of macrophage derived foam cell differentiation / phosphatidylcholine metabolic process / low-density lipoprotein particle remodeling /  phospholipase A2 / phospholipase A2 /  phospholipase A2 activity / phospholipase A2 activity /  Antimicrobial peptides / arachidonic acid secretion / phospholipid metabolic process / lipid catabolic process / negative regulation of T cell proliferation / Antimicrobial peptides / arachidonic acid secretion / phospholipid metabolic process / lipid catabolic process / negative regulation of T cell proliferation /  secretory granule / secretory granule /  phospholipid binding / positive regulation of inflammatory response / killing of cells of another organism / mitochondrial outer membrane / positive regulation of ERK1 and ERK2 cascade / defense response to Gram-positive bacterium / phospholipid binding / positive regulation of inflammatory response / killing of cells of another organism / mitochondrial outer membrane / positive regulation of ERK1 and ERK2 cascade / defense response to Gram-positive bacterium /  inflammatory response / inflammatory response /  calcium ion binding / endoplasmic reticulum membrane / perinuclear region of cytoplasm / calcium ion binding / endoplasmic reticulum membrane / perinuclear region of cytoplasm /  endoplasmic reticulum / endoplasmic reticulum /  extracellular space / extracellular exosome / extracellular region / extracellular space / extracellular exosome / extracellular region /  plasma membrane plasma membraneSimilarity search - Function | ||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 2.6 Å MOLECULAR REPLACEMENT / Resolution: 2.6 Å | ||||||
 Authors Authors | Edwards, S.H. / Thompson, D. / Baker, S.F. / Wood, S.P. / Wilton, D.C. | ||||||
 Citation Citation |  Journal: Biochemistry / Year: 2002 Journal: Biochemistry / Year: 2002Title: The crystal structure of the H48Q active site mutant of human group IIA secreted phospholipase A2 at 1.5 A resolution provides an insight into the catalytic mechanism Authors: Edwards, S.H. / Thompson, D. / Baker, S.F. / Wood, S.P. / Wilton, D.C. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1n29.cif.gz 1n29.cif.gz | 34.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1n29.ent.gz pdb1n29.ent.gz | 26.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1n29.json.gz 1n29.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/n2/1n29 https://data.pdbj.org/pub/pdb/validation_reports/n2/1n29 ftp://data.pdbj.org/pub/pdb/validation_reports/n2/1n29 ftp://data.pdbj.org/pub/pdb/validation_reports/n2/1n29 | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein |  / Phosphatidylcholine 2-acylhydrolase / Group IIA phospholipase A2 / GIIC sPLA2 / Non-pancreatic ...Phosphatidylcholine 2-acylhydrolase / Group IIA phospholipase A2 / GIIC sPLA2 / Non-pancreatic secretory phospholipase A2 / NPS-PLA2 / Phosphatidylcholine 2-acylhydrolase / Group IIA phospholipase A2 / GIIC sPLA2 / Non-pancreatic ...Phosphatidylcholine 2-acylhydrolase / Group IIA phospholipase A2 / GIIC sPLA2 / Non-pancreatic secretory phospholipase A2 / NPS-PLA2Mass: 13901.986 Da / Num. of mol.: 1 / Mutation: N1A Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Plasmid: pET11a / Species (production host): Escherichia coli / Production host: Homo sapiens (human) / Plasmid: pET11a / Species (production host): Escherichia coli / Production host:   Escherichia coli BL21(DE3) (bacteria) / Strain (production host): BL21 DE3 / References: UniProt: P14555, Escherichia coli BL21(DE3) (bacteria) / Strain (production host): BL21 DE3 / References: UniProt: P14555,  phospholipase A2 phospholipase A2 | ||
|---|---|---|---|
| #2: Chemical | | #3: Water | ChemComp-HOH / |  Water Water |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.58 Å3/Da / Density % sol: 52.34 % | |||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Crystal grow | Temperature: 277 K / Method: vapor diffusion, hanging drop / pH: 7.5 Details: NaCl, CaCl2, pH 7.5, VAPOR DIFFUSION, HANGING DROP, temperature 277K | |||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 4 ℃ / Method: vapor diffusion | |||||||||||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: ENRAF-NONIUS / Wavelength: 1.5418 Å ROTATING ANODE / Type: ENRAF-NONIUS / Wavelength: 1.5418 Å |
| Detector | Type: MARRESEARCH / Detector: IMAGE PLATE / Date: Mar 21, 2000 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.5418 Å / Relative weight: 1 : 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 2.6→15 Å / Num. all: 4851 / Num. obs: 4635 / % possible obs: 90.4 % / Observed criterion σ(F): 2 / Observed criterion σ(I): 2 |
| Reflection shell | Resolution: 2.6→2.8 Å / % possible all: 99.8 |
| Reflection | *PLUS Num. obs: 4693 / % possible obs: 99.5 % / Redundancy: 3.4 % / Num. measured all: 31180 / Rmerge(I) obs: 0.066 |
- Processing
Processing
| Software |
| ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT / Resolution: 2.6→15 Å / Cross valid method: THROUGHOUT / σ(F): 2 / Stereochemistry target values: Engh & Huber MOLECULAR REPLACEMENT / Resolution: 2.6→15 Å / Cross valid method: THROUGHOUT / σ(F): 2 / Stereochemistry target values: Engh & Huber
| ||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.6→15 Å
|
 Movie
Movie Controller
Controller












 PDBj
PDBj







