[English] 日本語
 Yorodumi
Yorodumi- PDB-1f6a: Structure of the human ige-fc bound to its high affinity receptor... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1f6a | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
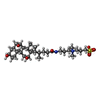
| Title | Structure of the human ige-fc bound to its high affinity receptor fc(epsilon)ri(alpha) | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords |  IMMUNE SYSTEM / IMMUNOGLOBULIN FOLD / IMMUNE SYSTEM / IMMUNOGLOBULIN FOLD /  GLYCOPROTEIN / GLYCOPROTEIN /  RECEPTOR / IGE-BINDING PROTEIN / IGE ANTIBODY / IGE-FC RECEPTOR / IGE-BINDING PROTEIN / IGE ANTIBODY / IGE-FC | |||||||||
| Function / homology |  Function and homology information Function and homology informationhigh-affinity IgE receptor activity / IgE B cell receptor complex / adaptive immune memory response / primary adaptive immune response /  type I hypersensitivity / B cell antigen processing and presentation / Fc receptor-mediated immune complex endocytosis / eosinophil degranulation / IgE immunoglobulin complex / type I hypersensitivity / B cell antigen processing and presentation / Fc receptor-mediated immune complex endocytosis / eosinophil degranulation / IgE immunoglobulin complex /  macrophage activation ...high-affinity IgE receptor activity / IgE B cell receptor complex / adaptive immune memory response / primary adaptive immune response / macrophage activation ...high-affinity IgE receptor activity / IgE B cell receptor complex / adaptive immune memory response / primary adaptive immune response /  type I hypersensitivity / B cell antigen processing and presentation / Fc receptor-mediated immune complex endocytosis / eosinophil degranulation / IgE immunoglobulin complex / type I hypersensitivity / B cell antigen processing and presentation / Fc receptor-mediated immune complex endocytosis / eosinophil degranulation / IgE immunoglobulin complex /  macrophage activation / IgE binding / type 2 immune response / Fc epsilon receptor (FCERI) signaling / macrophage activation / IgE binding / type 2 immune response / Fc epsilon receptor (FCERI) signaling /  antibody-dependent cellular cytotoxicity / mast cell degranulation / B cell proliferation / immunoglobulin complex, circulating / immunoglobulin receptor binding / macrophage differentiation / antibody-dependent cellular cytotoxicity / mast cell degranulation / B cell proliferation / immunoglobulin complex, circulating / immunoglobulin receptor binding / macrophage differentiation /  regulation of immune response / Role of LAT2/NTAL/LAB on calcium mobilization / regulation of immune response / Role of LAT2/NTAL/LAB on calcium mobilization /  complement activation, classical pathway / complement activation, classical pathway /  antigen binding / FCERI mediated Ca+2 mobilization / FCERI mediated MAPK activation / B cell receptor signaling pathway / FCERI mediated NF-kB activation / transmembrane signaling receptor activity / antibacterial humoral response / Interleukin-4 and Interleukin-13 signaling / antigen binding / FCERI mediated Ca+2 mobilization / FCERI mediated MAPK activation / B cell receptor signaling pathway / FCERI mediated NF-kB activation / transmembrane signaling receptor activity / antibacterial humoral response / Interleukin-4 and Interleukin-13 signaling /  adaptive immune response / cell surface receptor signaling pathway / adaptive immune response / cell surface receptor signaling pathway /  immune response / immune response /  inflammatory response / inflammatory response /  cell surface / cell surface /  extracellular space / extracellular region / extracellular space / extracellular region /  plasma membrane plasma membraneSimilarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / Resolution: 3.5 Å SYNCHROTRON / Resolution: 3.5 Å | |||||||||
 Authors Authors | Garman, S.C. / Wurzburg, B.A. / Tarchevskaya, S.S. / Kinet, J.P. / Jardetzky, T.S. | |||||||||
 Citation Citation |  Journal: Nature / Year: 2000 Journal: Nature / Year: 2000Title: Structure of the Fc fragment of human IgE bound to its high-affinity receptor Fc (epsilon) RI (alpha). Authors: Garman, S.C. / Wurzburg, B.A. / Tarchevskaya, S.S. / Kinet, J.P. / Jardetzky, T.S. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1f6a.cif.gz 1f6a.cif.gz | 143.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1f6a.ent.gz pdb1f6a.ent.gz | 119.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1f6a.json.gz 1f6a.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/f6/1f6a https://data.pdbj.org/pub/pdb/validation_reports/f6/1f6a ftp://data.pdbj.org/pub/pdb/validation_reports/f6/1f6a ftp://data.pdbj.org/pub/pdb/validation_reports/f6/1f6a | HTTPS FTP |
|---|
-Related structure data
| Related structure data | |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| Unit cell |
| |||||||||
| Noncrystallographic symmetry (NCS) | NCS domain:
| |||||||||
| Details | The biological assembly is the receptor chain A bound to the dimeric antibody chains B and D |
- Components
Components
-Protein , 2 types, 3 molecules ABD
| #1: Protein | Mass: 20318.611 Da / Num. of mol.: 1 / Fragment: EXTRACELLULAR DOMAIN / Mutation: N74A,N135A,T142A,V143A Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Plasmid: PVL1392 / Production host: HI-5 INSECT CELLS / References: UniProt: P12319 Homo sapiens (human) / Plasmid: PVL1392 / Production host: HI-5 INSECT CELLS / References: UniProt: P12319 |
|---|---|
| #2: Protein | Mass: 24821.018 Da / Num. of mol.: 2 / Fragment: C(EPSILON)3-C(EPSILON)4 DOMAINS Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human) / Plasmid: PVL1392 / Production host: HI-5 INSECT CELLS / References: UniProt: P01854 Homo sapiens (human) / Plasmid: PVL1392 / Production host: HI-5 INSECT CELLS / References: UniProt: P01854 |
-Sugars , 5 types, 5 molecules
| #3: Polysaccharide | alpha-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-[alpha-L-fucopyranose-(1- ...alpha-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-[alpha-L-fucopyranose-(1-6)]2-acetamido-2-deoxy-beta-D-glucopyranose / Mass: 732.682 Da / Num. of mol.: 1 / Mass: 732.682 Da / Num. of mol.: 1Source method: isolated from a genetically manipulated source |
|---|---|
| #4: Polysaccharide | alpha-D-mannopyranose-(1-3)-[alpha-D-mannopyranose-(1-6)]alpha-D-mannopyranose-(1-4)-2-acetamido-2- ...alpha-D-mannopyranose-(1-3)-[alpha-D-mannopyranose-(1-6)]alpha-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose / Mass: 910.823 Da / Num. of mol.: 1 / Mass: 910.823 Da / Num. of mol.: 1Source method: isolated from a genetically manipulated source |
| #5: Polysaccharide | 2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-[alpha-L-fucopyranose-(1-6)]2-acetamido-2-deoxy-beta- ...2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-[alpha-L-fucopyranose-(1-6)]2-acetamido-2-deoxy-beta-D-glucopyranose / Mass: 570.542 Da / Num. of mol.: 1 / Mass: 570.542 Da / Num. of mol.: 1Source method: isolated from a genetically manipulated source |
| #6: Polysaccharide | alpha-D-mannopyranose-(1-2)-alpha-D-mannopyranose-(1-6)-[alpha-D-mannopyranose-(1-3)]alpha-D- ...alpha-D-mannopyranose-(1-2)-alpha-D-mannopyranose-(1-6)-[alpha-D-mannopyranose-(1-3)]alpha-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose / Mass: 1072.964 Da / Num. of mol.: 1 / Mass: 1072.964 Da / Num. of mol.: 1Source method: isolated from a genetically manipulated source |
| #7: Polysaccharide | alpha-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta- ...alpha-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose / Mass: 586.542 Da / Num. of mol.: 1 / Mass: 586.542 Da / Num. of mol.: 1Source method: isolated from a genetically manipulated source |
-Non-polymers , 2 types, 10 molecules 

| #8: Chemical | ChemComp-SO4 /  Sulfate Sulfate#9: Chemical | ChemComp-CPS /  CHAPS detergent CHAPS detergent |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 7.28 Å3/Da / Density % sol: 80 % | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Crystal grow | Temperature: 296 K / Method: vapor diffusion, hanging drop / pH: 8.5 Details: Ammonium Sulfate, Tris, CHAPS, pH 8.5, VAPOR DIFFUSION, HANGING DROP, temperature 296K | |||||||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 23 ℃ | |||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction |
| |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source |
| |||||||||||||||
| Detector |
| |||||||||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | |||||||||||||||
| Radiation wavelength |
| |||||||||||||||
| Reflection | Resolution: 3.5→40 Å / Num. all: 27510 / Num. obs: 27411 / % possible obs: 99.8 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Redundancy: 3.1 % / Biso Wilson estimate: 100.5 Å2 / Rmerge(I) obs: 0.091 / Net I/σ(I): 11 | |||||||||||||||
| Reflection shell | Resolution: 3.5→3.63 Å / Redundancy: 3.1 % / Rmerge(I) obs: 0.461 / Num. unique all: 2704 / % possible all: 99.7 | |||||||||||||||
| Reflection | *PLUS Num. obs: 27484 / Num. measured all: 84873 | |||||||||||||||
| Reflection shell | *PLUS % possible obs: 99.7 % / Mean I/σ(I) obs: 2.7 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Resolution: 3.5→36.87 Å / Rfactor Rfree error: 0.007 / Data cutoff high absF: 5879235.76 / Data cutoff low absF: 0 / Isotropic thermal model: RESTRAINED / Cross valid method: THROUGHOUT / σ(F): 0 / σ(I): 0 / Stereochemistry target values: Engh & Huber
| ||||||||||||||||||||||||||||||||||||
| Solvent computation | Solvent model: FLAT MODEL / Bsol: 72.83 Å2 / ksol: 0.26 e/Å3 | ||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 89.2 Å2
| ||||||||||||||||||||||||||||||||||||
| Refine analyze |
| ||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 3.5→36.87 Å
| ||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||
| Refine LS restraints NCS |
| ||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 3.5→3.72 Å / Rfactor Rfree error: 0.021 / Total num. of bins used: 6
| ||||||||||||||||||||||||||||||||||||
| Xplor file |
| ||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name: CNS / Version: 0.9 / Classification: refinement | ||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller












 PDBj
PDBj











