[English] 日本語
 Yorodumi
Yorodumi- PDB-1e8w: Structure determinants of phosphoinositide 3-kinase inhibition by... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1e8w | ||||||
|---|---|---|---|---|---|---|---|
| Title | Structure determinants of phosphoinositide 3-kinase inhibition by wortmannin, LY294002, quercetin, myricetin and staurosporine | ||||||
 Components Components | PHOSPHATIDYLINOSITOL 3-KINASE CATALYTIC SUBUNIT | ||||||
 Keywords Keywords | PHOSPHOINOSITIDE 3-KINASE GAMMA / SECONDARY MESSENGER GENERATION /  PI3K / PI 3K / PI3K / PI 3K /  QUERCETIN QUERCETIN | ||||||
| Function / homology |  Function and homology information Function and homology information phosphatidylinositol-4-phosphate 3-kinase / phosphatidylinositol 3-kinase complex, class IB / 1-phosphatidylinositol-4-phosphate 3-kinase activity / 1-phosphatidylinositol-4,5-bisphosphate 3-kinase activity / phosphatidylinositol-4-phosphate 3-kinase / phosphatidylinositol 3-kinase complex, class IB / 1-phosphatidylinositol-4-phosphate 3-kinase activity / 1-phosphatidylinositol-4,5-bisphosphate 3-kinase activity /  phosphatidylinositol-4,5-bisphosphate 3-kinase / phosphatidylinositol 3-kinase complex, class IA / phosphatidylinositol-4,5-bisphosphate 3-kinase / phosphatidylinositol 3-kinase complex, class IA /  phosphatidylinositol 3-kinase / phosphatidylinositol-3-phosphate biosynthetic process / 1-phosphatidylinositol-3-kinase activity / phosphatidylinositol 3-kinase / phosphatidylinositol-3-phosphate biosynthetic process / 1-phosphatidylinositol-3-kinase activity /  immune system process ... immune system process ... phosphatidylinositol-4-phosphate 3-kinase / phosphatidylinositol 3-kinase complex, class IB / 1-phosphatidylinositol-4-phosphate 3-kinase activity / 1-phosphatidylinositol-4,5-bisphosphate 3-kinase activity / phosphatidylinositol-4-phosphate 3-kinase / phosphatidylinositol 3-kinase complex, class IB / 1-phosphatidylinositol-4-phosphate 3-kinase activity / 1-phosphatidylinositol-4,5-bisphosphate 3-kinase activity /  phosphatidylinositol-4,5-bisphosphate 3-kinase / phosphatidylinositol 3-kinase complex, class IA / phosphatidylinositol-4,5-bisphosphate 3-kinase / phosphatidylinositol 3-kinase complex, class IA /  phosphatidylinositol 3-kinase / phosphatidylinositol-3-phosphate biosynthetic process / 1-phosphatidylinositol-3-kinase activity / phosphatidylinositol 3-kinase / phosphatidylinositol-3-phosphate biosynthetic process / 1-phosphatidylinositol-3-kinase activity /  immune system process / phosphatidylinositol phosphate biosynthetic process / phosphatidylinositol 3-kinase/protein kinase B signal transduction / immune system process / phosphatidylinositol phosphate biosynthetic process / phosphatidylinositol 3-kinase/protein kinase B signal transduction /  endocytosis / endocytosis /  chemotaxis / chemotaxis /  angiogenesis / angiogenesis /  non-specific serine/threonine protein kinase / non-specific serine/threonine protein kinase /  inflammatory response / inflammatory response /  phosphorylation / protein serine kinase activity / protein serine/threonine kinase activity / phosphorylation / protein serine kinase activity / protein serine/threonine kinase activity /  ATP binding / ATP binding /  plasma membrane / plasma membrane /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |   SUS SCROFA (pig) SUS SCROFA (pig) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.5 Å MOLECULAR REPLACEMENT / Resolution: 2.5 Å | ||||||
 Authors Authors | Walker, E.H. / Pacold, M.E. / Perisic, O. / Stephens, L. / Hawkins, P.T. / Wymann, M.P. / Williams, R.L. | ||||||
 Citation Citation |  Journal: Mol.Cell / Year: 2000 Journal: Mol.Cell / Year: 2000Title: Structural Determinations of Phosphoinositide 3-Kinase Inhibition by Wortmannin, Ly294002, Quercetin, Myricetin and Staurosporine Authors: Walker, E.H. / Pacold, M.E. / Perisic, O. / Stephens, L. / Hawkins, P.T. / Whymann, M.P. / Williams, R.L. #1: Journal: Nature / Year: 1999 Title: Structural Insights Into Phosphoinositide 3-Kinase Catalysis and Signalling Authors: Walker, E.H. / Perisic, O. / Ried, C. / Stephens, L. / Williams, R.L. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1e8w.cif.gz 1e8w.cif.gz | 185.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1e8w.ent.gz pdb1e8w.ent.gz | 144.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1e8w.json.gz 1e8w.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/e8/1e8w https://data.pdbj.org/pub/pdb/validation_reports/e8/1e8w ftp://data.pdbj.org/pub/pdb/validation_reports/e8/1e8w ftp://data.pdbj.org/pub/pdb/validation_reports/e8/1e8w | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1e7uC  1e7vC  1e8xC  1e8yC  1e8zC  1e90C  1qmm C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 109952.555 Da / Num. of mol.: 1 / Fragment: PI3-KINASE P110 SUBUNIT GAMMA / Mutation: YES Source method: isolated from a genetically manipulated source Details: QUERCETIN / Source: (gene. exp.)   SUS SCROFA (pig) / Cell: NEUTROPHIL / Gene: P120S144C / Plasmid: PACHLT-C / Gene (production host): P120S144C / Production host: SUS SCROFA (pig) / Cell: NEUTROPHIL / Gene: P120S144C / Plasmid: PACHLT-C / Gene (production host): P120S144C / Production host:   SPODOPTERA FRUGIPERDA (fall armyworm) / Strain (production host): Sf9 / References: UniProt: O02697, SPODOPTERA FRUGIPERDA (fall armyworm) / Strain (production host): Sf9 / References: UniProt: O02697,  phosphatidylinositol 3-kinase phosphatidylinositol 3-kinase |
|---|---|
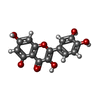
| #2: Chemical | ChemComp-QUE /  Quercetin Quercetin |
| Sequence details | THE NATIVE PROTEIN WAS MUTATED BY DELETION OF RESIDUES 1-143. THE RESIDUE 143 LISTED HERE IS FROM ...THE NATIVE PROTEIN WAS MUTATED BY DELETION OF RESIDUES 1-143. THE RESIDUE 143 LISTED HERE IS FROM THE HIS-TAG, AFTER THROMBIN CLEAVAGE. ALSO RESEQUENCI |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.829 Å3/Da / Density % sol: 54.85 % | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Crystal grow | pH: 7.25 / Details: 14% PEG 4000, 0.2 M LI2SO4, 0.1 M TRIS PH 7.25 | ||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 17 ℃ / pH: 7.2 / Method: vapor diffusion, hanging drop / Details: used hair seeding | ||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: ID14-2 / Wavelength: 0.93 / Beamline: ID14-2 / Wavelength: 0.93 |
| Detector | Type: MARRESEARCH / Detector: CCD / Date: Sep 15, 1999 / Details: BENT MIRROR |
| Radiation | Monochromator: DIAMOND C(111) / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 0.93 Å / Relative weight: 1 : 0.93 Å / Relative weight: 1 |
| Reflection | Resolution: 2.5→62.45 Å / Num. obs: 34614 / % possible obs: 97.5 % / Observed criterion σ(I): 0 / Redundancy: 3.37 % / Biso Wilson estimate: 57.3 Å2 / Rmerge(I) obs: 0.059 / Net I/σ(I): 22.55 |
| Reflection shell | Resolution: 2.5→2.64 Å / Redundancy: 2.9 % / Mean I/σ(I) obs: 2.51 / Rsym value: 0.397 / % possible all: 83.1 |
| Reflection | *PLUS Num. measured all: 116715 |
| Reflection shell | *PLUS % possible obs: 83.1 % |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1QMM  1qmm Resolution: 2.5→62.45 Å / Rfactor Rfree error: 0.008 / Data cutoff high absF: 1791131.55 / Isotropic thermal model: RESTRAINED / Cross valid method: THROUGHOUT / σ(F): 0
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Solvent model: FLAT MODEL / Bsol: 49.5134 Å2 / ksol: 0.337475 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 68.2 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.5→62.45 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.5→2.66 Å / Rfactor Rfree error: 0.028 / Total num. of bins used: 6
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xplor file |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name: CNS / Version: 1 / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Rfactor Rfree : 0.33 : 0.33 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller












 PDBj
PDBj