[English] 日本語
 Yorodumi
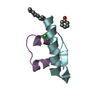
Yorodumi- PDB-1a4t: SOLUTION STRUCTURE OF PHAGE P22 N PEPTIDE-BOX B RNA COMPLEX, NMR,... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1a4t | ||||||
|---|---|---|---|---|---|---|---|
| Title | SOLUTION STRUCTURE OF PHAGE P22 N PEPTIDE-BOX B RNA COMPLEX, NMR, 20 STRUCTURES | ||||||
 Components Components |
| ||||||
 Keywords Keywords | TRANSCRIPTION/RNA / BACTERIOPHAGE TRANSCRIPTIONAL ANTITERMINATION / PEPTIDE-RNA RECOGNITION / GNRA LOOP / BENT ALPHA-HELICAL PEPTIDE /  TRANSCRIPTION REGULATION / TRANSCRIPTION-RNA COMPLEX TRANSCRIPTION REGULATION / TRANSCRIPTION-RNA COMPLEX | ||||||
| Function / homology | DNA-templated transcription termination /  RNA / RNA (> 10) / Probable regulatory protein N RNA / RNA (> 10) / Probable regulatory protein N Function and homology information Function and homology information | ||||||
| Biological species |   Enterobacteria phage P22 (virus) Enterobacteria phage P22 (virus) | ||||||
| Method |  SOLUTION NMR / distance geometry SOLUTION NMR / distance geometry | ||||||
 Authors Authors | Cai, Z. / Gorin, A.A. / Frederick, R. / Ye, X. / Hu, W. / Majumdar, A. / Kettani, A. / Patel, D.J. | ||||||
 Citation Citation |  Journal: Nat.Struct.Biol. / Year: 1998 Journal: Nat.Struct.Biol. / Year: 1998Title: Solution structure of P22 transcriptional antitermination N peptide-boxB RNA complex. Authors: Cai, Z. / Gorin, A. / Frederick, R. / Ye, X. / Hu, W. / Majumdar, A. / Kettani, A. / Patel, D.J. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1a4t.cif.gz 1a4t.cif.gz | 328.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1a4t.ent.gz pdb1a4t.ent.gz | 280.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1a4t.json.gz 1a4t.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/a4/1a4t https://data.pdbj.org/pub/pdb/validation_reports/a4/1a4t ftp://data.pdbj.org/pub/pdb/validation_reports/a4/1a4t ftp://data.pdbj.org/pub/pdb/validation_reports/a4/1a4t | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: RNA chain | Mass: 4829.969 Da / Num. of mol.: 1 / Source method: obtained synthetically |
|---|---|
| #2: Protein/peptide | Mass: 2415.781 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Enterobacteria phage P22 (virus) / Genus: P22-like viruses / References: UniProt: P04891 Enterobacteria phage P22 (virus) / Genus: P22-like viruses / References: UniProt: P04891 |
-Experimental details
-Experiment
| Experiment | Method:  SOLUTION NMR SOLUTION NMR |
|---|---|
| NMR experiment | Type : SEE BELOW : SEE BELOW |
- Sample preparation
Sample preparation
| Sample conditions | pH: 5.7 / Temperature: 297 K |
|---|
-NMR measurement
| NMR spectrometer |
|
|---|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR software |
| ||||||||||||||||||||||||||||
| Refinement | Method: distance geometry / Software ordinal: 1 Details: A TOTAL OF 100 INITIAL STRUCTURES OF THE COMPLEX WERE GENERATED USING THE X-PLOR METRIC MATRIX DISTANCE GEOMETRY PROTOCOL GUIDED BY THE NOE DISTANCE RESTRAINTS. ALL THE DISTANCE WERE ...Details: A TOTAL OF 100 INITIAL STRUCTURES OF THE COMPLEX WERE GENERATED USING THE X-PLOR METRIC MATRIX DISTANCE GEOMETRY PROTOCOL GUIDED BY THE NOE DISTANCE RESTRAINTS. ALL THE DISTANCE WERE SUBJECTED TO TWO ROUNDS OF MOLECULAR DYNAMICS REFINEMENT WITH THE NONBO NDED INTERACTIONS SUBJECTED TO A REPULSIVE FORCE FIELD IN THE INITIAL ROUND AND A CHARMM-22 FORCE FIELD CONTAINING VAN DER WAALS AND HYDROGEN BOND (WITH WELL DEPTH OF 3 KCAL/MOL) ENERGY TERM IN THE SUBSEQUENT ROUND. THE ELECTROSTATIC ENERGY TERM WAS TURNE D OFF DURING BOTH ROUNDS OF THE COMPUTATIONS. THE MOLECULAR DYNAMICS PROTOCOL DURING EACH ROUND OF THE COMPUTATIONS WAS AS FOLLOWS. THE STRUCTURES WERE HEATED TO 1000K AND EQUILIBRATED AT THIS TEMPERATURE FOR 0.8 PS, FOLLOWED BY GRADUAL COOLING TO 300 K O VER 48 PS AND SUBSEQUENT EQUILIBRATION AT THIS TEMPERATURE FOR 6 PS. THE RESULTING STRUCTURES WERE SUBJECTED TO 2,000 CYCLES OF ENERGY MINIMIZATION. THE FORCE CONSTANTS FOR NOE DISTANCE RESTRAINTS AND COUPLING CONSTANT BASED DIHEDRAL ANGLE RESTRAINTS WERE MAINTAINED AT VALUES 50 KCAL/MOL A A AND 25 KCAL/MOL RAD RAD, RESPECTIVELY THROUGHOUT THE CALCULATIONS. A FORCE CONSTANT OF 60 KCAL/MOL A A WAS APPLIED TO MAINTAIN HYDROGEN BONDING DISTANCE RESTRAINTS FOR WATSON-CRICK BASE PAIRS IN THE RNA STEM OF THE CO MPLEX. A SUBSET OF 27 REFINED STRUCTURES WERE SELECTED BASED ON THEIR LOWER VALUES OF TOTAL ENERGY, VAN DER WAALS ENERGY AND NOE VIOLATIONS ENERGY. THESE 27 REFINED STRUCTURES DEFINED A NOTICEABLE CONVERGED SUBSET (~10 KCAL/MOL VARIATION OF TOTAL ENERGY W ITHIN SUBSET) AND WERE SEPARATED BY A GAP OF ~120 KCAL/MOL OF TOTAL ENERGY FROM THE REST OF THE ENSEMBLE OF REFINED STRUCTURES. THE 20 STRUCTURES OF THE COMPLEX WITH THE LOWEST NUMBER OF NOE VIOLATIONS EXHIBITED PAIRWISE R.M.S.D. VALUE FOR ALL HEAVY ATOMS OF 1.27 A. | ||||||||||||||||||||||||||||
| NMR ensemble | Conformer selection criteria: DESCRIBED BELOW / Conformers calculated total number: 100 / Conformers submitted total number: 20 |
 Movie
Movie Controller
Controller










 PDBj
PDBj





























